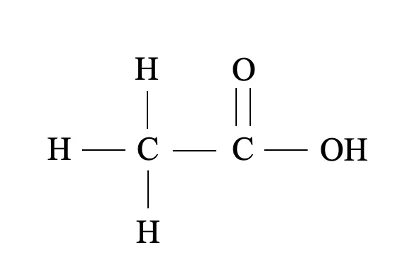
Carboxylic acids
All contain the -COOH functional group, e.g:
Physical properties
Short chained acids are soluble in water (form hydrogen bonds with water). Solubility decreases as chain length increases.
Melting and boiling points are higher than corresponding alkanes due to hydrogen bonds
Must be in water to dissociate/ionise. Carboxylic acids are weak acids, e.g.
CH3COOH(aq) ⇌ CH3COO- + H+
Carboxylate ion - relatively stable, delocalises the negative charge
Equilibrium constant still on the left
The carbon that contains the functional group is carbon no. 1 when naming
Chemical reactions
React with;
Metals → Salt+ hydrogen
Alkalis/oxides → Salt + water
Carbonates → Salt + water + CO2
Test for carboxylic acids
E.g. ethanoic acid + calcium carbonate → calcium ethanoate + water + carbon dioxide
2CH3COOH + CaCO3 → Ca(CH3COO)2 + H2O + CO2
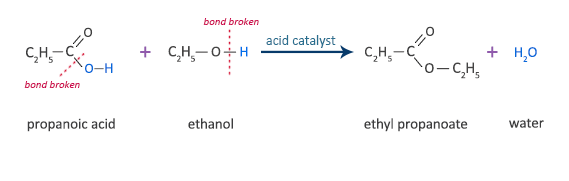
React with alcohols to form esters
E.g.