L4 - Valence Bond and Molecular Orbital Theory
Lewis Theory of Covalent Bonding
A Lewis structure is a topological map of the arrangement of bonds, and rarely portrays the actual geometrical structure of a molecule.
Valence Shell Electron Pair Repulsion (VSEPR) Model
Basic idea: Electron pairs around the central atom maximize their separation.
Example: If an atom has 4 electron pairs in its valence shell, the pairs adopt a tetrahedral arrangement.
Valence Bond (VB) Theory and Molecular Orbital (MO) Theory
VB theory is easier to apply and provides a good understanding of bonding in a molecule’s ground state.
MO theory is more useful in understanding molecules in excited states (e.g., light absorption properties).
Valence Bond Theory
Orbitals overlap between 2 atoms.
A maximum of 2 electrons of opposite spin are present in the overlapping orbitals (generally one electron supplied by each of the 2 bonded atoms, but not always).
An electron in one atomic orbital pairs its spin with an electron in another atom which allows for bonding.
Why Does a Bond Form?
When two atoms are at their bonding distance, an electron from atom A may be found on A or B.
Valence Bond (VB) theory suggests adding the wave functions (i.e., no electron-electron interaction assumed) because it is equally likely.
Why Bonding Occurs
As two atoms approach each other, there is an accumulation of charge between the two nuclei, which lowers the potential energy of the system.
Electrons can migrate from one to other atoms lowering their kinetic energy.
Sigma Bonds
Overlap of 1s orbitals of hydrogen atoms creates an H-H σ bond.
Overlap of 1s orbital of hydrogen and 2p orbital of fluorine creates an H-F sigma (σ) bond.
Overlap of 2p orbitals of fluorine atoms creates an F-F sigma (σ) bond.
Electron density is greatest along the axis of the bond.
Methane
Lewis structure and VSEPR theory predict 4 equivalent bonding pairs at 109.5°.
The valence shell electron configuration of C is 2s, 2p_x, 2p_y, 2p_z. Unpaired electrons are in atomic p orbitals at 90° to one another, but there are only 2 of them. Does this mean only 2 bonds should be formed?
Hybrid Atomic Orbitals
Created by mixing together atomic orbitals to form an equal number of new hybrid atomic orbitals.
In methane, an isolated C atom undergoes hybridization to form 4 sp^3 hybrid orbitals, resulting in tetrahedral geometry.
Hybridization
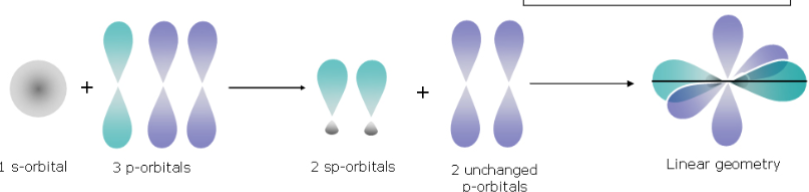
BeF_2: linear geometry, 180°.
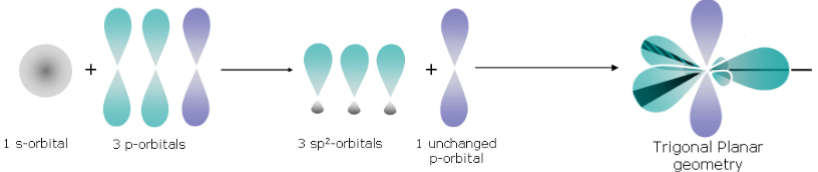
BF_3: trigonal planar geometry, 120°.
PF_5: trigonal bipyramidal geometry, 90° and 120°.
SF_6: octahedral geometry, 90°.
BeF_2 – Hybridisation (Linear Geometry)
2s 2px 2py 2pz
Isolated Be atom - Pair of electrons in 2s orbital, no electrons in p orbitals.
Formation of 2 sp hybrid orbitals, while the other two p orbitals remain unchanged.
BF_3 – Hybridisation (Trigonal Planar Geometry)
2s 2p_x 2p_y 2p_z
Isolated B atom - Pair of electrons in 2s orbital, single unpaired electron in 2p_x orbital.
Formation of 3 sp hybrid orbitals while the other p orbital remains unchanged
PF_5 – Hybridisation (Trigonal Bipyramidal Geometry)
3s3p_x3p_y3p_z + 5 3d orbitals
Isolated P atom - Pair of electrons in 3s orbital, unpaired electrons filling 3p orbitals in all orientations.
Formation of 5 sp3d hybrid orbitals while the other four 3d orbitals remain unchanged
SF_6 – Hybridisation (Octahedral Geometry)
3s 3p_x 3p_y 3p_z + 5 3d orbitals
Isolated S atom - Pair of electrons in 3s and 3px orbitals, unpaired electrons in remaining p orbitals
Formation of 5 sp3d2 hybrid orbitals while other three 3w orbitals remain unchanged
Hybridisation - Summary
Remember the number of hybrid atomic orbitals formed is equal to the number of atomic orbitals used to form them.
Multiple Bonds: Pi Bonds - Double Bond
Ethene: C=C
Electron density is above and below the internuclear axis
P orbitals merge together to form pi bond
Bonding in Ethene
2s 2p_x 2p_y 2p_z
Isolated C atom - Pair of electrons in 2s, unpaired electrons in 2px and 2py with 2pz being empty.
Formation of 3 sp2 hybrid orbitals with 1 unchanged p orbital containing an unpaired electron.
The unchanged p orbital is used for \pi bonding to other C. The C \sigma bonds are used for s bonding to other C and H atoms.
Pi Bonds – Triple Bond
Ethyne: H-C≡C-H
Hybrid Orbitals
Number | Shape | Hybridization |
|---|---|---|
2 | Linear | sp |
3 | Trigonal planar | sp² |
4 | Tetrahedral | sp³ |
5 | Trigonal bipyramidal | sp³d |
6 | Octahedral | sp³d2 |
Delocalisation – Nitrate Ion (NO3-)
Laboratory analyses show all 3 bonds in the nitrate ion to be the same strength and the same length.
The behaviour of the bonds suggests they are longer than double bonds and shorter than single bonds.
This fact is despite being drawn as
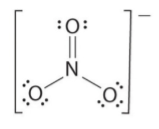
What’s actually happening is that the extra bond in the top double bond is delocalised and is shared between all 3 bonds
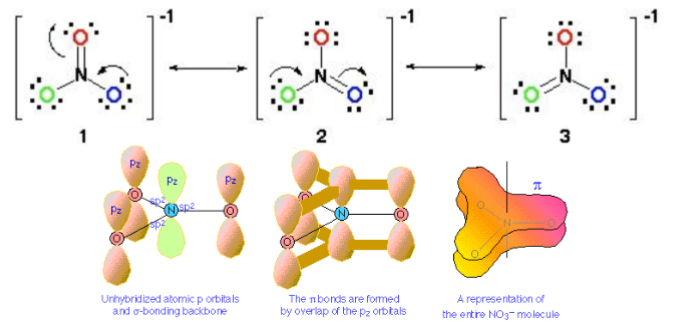
This happens because the \pi bonds are formed between all oxygens and the nitrogen, not just one
Delocalisation - Benzene
Laboratory analyses show all 6 C-C bonds in the benzene ring to be the same strength and the same length.
Each C is sp2 hybridised with a p orbital out of the molecular plane.
Delocalisation - Hexatriene
Each C is in 3 coordination and is trigonal planar and sp2 hybridised.
This leaves a p orbital out of the molecular plane for p bonding.
p orbitals overlap on both sides causing delocalisation of 6 p electrons over the molecule.
Orbitals can overlap in-phase or out of phase (analogous to positive and negative interference of waves).
The in-phase overlap gives the lowest energy orbital.
Molecular Orbital Theory
In a molecular orbital we are making a combination of wave function.
Waves can have constructive and destructive interference.
Molecular Orbital (MO) Theory – The Principles
The number of MOs produced is always equal to the number of atomic orbitals combined.
The bonding MO is lower in energy than the parent orbitals and the antibonding orbital is higher in energy.
Electrons in the molecule are assigned to orbitals of successively higher energy according to the Pauli exclusion principle and Hund’s rule.
Atomic orbitals combine to form MOs most effectively when the atomic orbitals are of similar energy.
In contrast to VB theory that assumes one bond is formed by the combination of 2 atomic or hybrid orbitals. MO theory assumes that 2 molecular orbitals result from 2 atomic orbitals.
\text{Bond Order} = \frac{\text{Number of Bonding Electrons - Number of Antibonding Electrons}}{2}
Bond order tells you the number of bonds that are made between molecules
Bond order > 0 - Stable molecule
Bond order = 0 - Unstable/nonexistent molecule
H2 Molecule – Energy Level Diagram
There are 2 electrons in the H2 molecule so electron configuration is \sigma_{1s²}.
This means there are 2 bonding electrons and no antibonding electrons, so the bond order is 1.
Bonding energy = 2E_b, H2 is energetically favourable.
He2 Molecule – Energy Level Diagram
There are 4 electrons in the He2 molecule so electron configuration is (\sigma_{1s^2} \sigma^*_{1s^2}) .
There are 2 bonding electrons and 2 antibonding electrons so the bond order is 0.
Bonding energy = 2E_b - 2E_{ab}, He2 is energetically unfavourable.
Li2 Molecule – Energy Level Diagram
Net bonding effect due to 2 electrons in s2s
The Overlap of p Orbitals
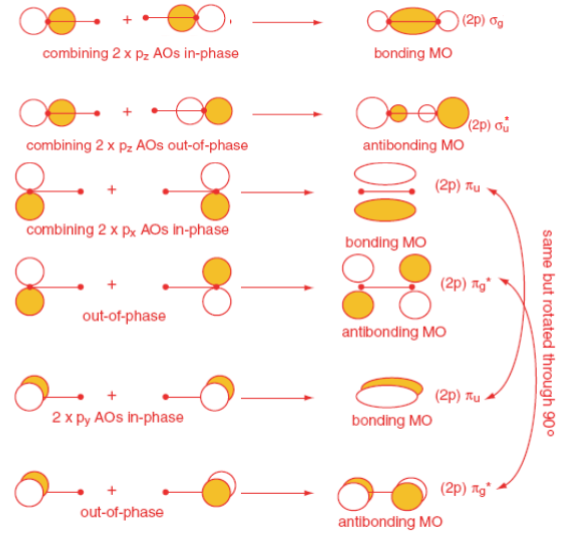
The O2 Molecule
Atomic orbitals for O: 1s^2 2s^2 2p^4
Lewis structure and VB theory predict no unpaired electrons, but O2 is paramagnetic.
The O2 Molecule
6 valence electrons on each O atom, so 12 electrons to accommodate in the MOs created by overlapping the 2s and 2p orbitals.
Still get the equivalent of a double bond.
Polar Bonds
AOs with the same energy.
AOs with different energy.
Ionic Bonding (LiF)
Orbital energy differences so great that MOs approximate unperturbed AOs. MO theory can describe ionic bonding
An Introduction to Crystal Field Theory (CFT)
Consider transition metal ions in octahedral sites in minerals like olivine, pyroxene, amphibole, garnet etc. or indeed complexed in aqueous solution (negative ions clustered around them).
Octahedral field is one possibility.