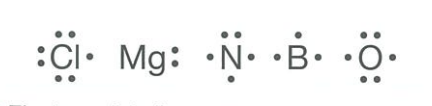Elements and the Periodic Table
An element is a pure substance that is made up of only one kind of atom. An element cannot be broken down into simpler substances due to a chemical reaction. There are over 100 individual elements seen throughout Science. Many of these react in different ways depending on the situation.
Some examples of elements are oxygen, carbon, nitrogen, hydrogen, neon, boron, and calcium.
Types of elements
Within chemistry, there are 3 main types of elements that are commonly seen. However, these groups can be broken down into even more groups based on their properties.
1) Metals: A solid material which is typically hard, shiny, malleable, fusible, and ductile, with good electrical and thermal conductivity. For eg: iron, gold, silver.
2) Metalloids: An elements whose properties are intermediate between those of metals and solid non-metals or semi-conductors. For eg: arsenic, antimony, tin.
3) Non-metals: An element or a substance that is not a metal. For eg: oxygen, chlorine, carbon.
Use of symbols on the periodic table
The periodic table is a record of all the elements. The periodic table gives us lots of information about each element. It tells us:
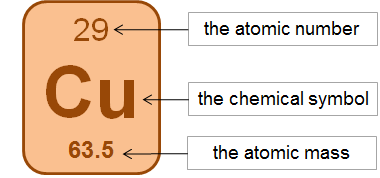
Each element’s symbol
The number of protons in the element’s atom. This is called its atomic number. The atomic numbers increase by 1 as you go through the periodic table.
The atomic mass of an element. This tells us the mass of an element compared to carbon. It is approximately the total number of protons and neutrons added together.
The number of electrons in an atom. The atomic number tells us this, too, because this number is always the same as the number of protons.
Some of the properties of different elements.
Relationships between elements
Elements are classified into families according to their different chemical properties, and are arranged in the periodic table so that:
Families of elements with similar properties are in vertical columns called groups.
Elements are arranged in order of increasing atomic number — small, light atoms are towards the top of the table, and larger, heavier atoms are towards the bottom.
Horizontal rows are called periods (numbered 1-7)
You can predict an element’s properties, the types of reactions it takes part in and even the formulae of its compounds, from its position on the periodic table.
Metals
Metals, found towards the left:
Conduct heat and electricity
Have high melting and boiling points
Are mostly solid at room temperature
Are malleable and ductile
Are shiny when freshly cut or polished
Nonmetals
Nonmetals, found clustered around the top right corner:
Are very poor conductors of heat and electricity
Have low melting and boiling points
Mostly liquids and gases at room temperature
If solid, are mostly brittle, crumbly and dull
Semi metals or metalloids
Semimetals, found along the boundary between metals and nonmetals, have some metallic and some non-metallic properties — many are semiconductors of electricity, useful in electronics. They include boron, silicon, germanium, arsenic, antimony, tellurium, polonium and astatine. Metals are found to the left of this zigzag, and nonmetals to the right.
The alkali metals
These are the most reactive metals (including sodium and potassium) and make up Group 1 (the first column of the table). They are soft enough to cut with a knife, extremely reactive and dangerous. They react instantly with oxygen, spontaneously catching fire in air and also reacting violently with water to form alkalis (soluble bases) and hydrogen gas, producing so much heat that the hydrogen often ignites. They need to be handled extremely carefully and usually stored in a bottle of kerosene, where they can’t come in contact with air or water.
The alkaline-earth metals
These include calcium and magnesium, and make up group 2 — they have similar behaviour to the alkali metals and form different types of compounds, but are harder and less reactive.
The transition metals
These include many of the most useful and most valuable metals, e.g. copper, iron, zinc, silver and gold. Found between groups 1 and 3. All are typical metals — good conductors with high melting and boiling points and most are strong, hard solids at room temperature. Many form coloured compounds, many are good catalysts and some are magnetic. They are less reactive that groups 1 and 2, and gold andd platinum are so unreactive, they can be found in nature as elements.
The halogens
These are the most reactive non-metals. They include fluorine, chlorine, bromine and iodine and make up group 7. They react with hydrogen to form acids and, are called halogens or ‘salt producers’ as they react with metals to form salts.
The noble gases
These include helium, neon, argon, krypton, xenon and radon, and make up group 0. They are sometimes called the ‘inert gases’ as they are extremely un-reactive, and are the only elements in nature to occur as single unattached atoms.
Compounds, mixtures and molecules
In science, there are many different types of substances that we can observe. These include:
Elements
Compounds
Mixtures
Molecules
Each of these have different characteristics and chemical makeups that make them different from each other.
Mixtures
A mixture is seen when 2 or more substances are combined, however not chemically. There are some general properties of mixtures that we need to understand:
The components of a mixture can be easily separated
The components each keep their original properties
The proportion of the components is variable.
Mixtures are formed when two or more substances are physically combined, but they don’t chemically react or form a new substance.
There are 2 main types of mixtures:
A heterogeneous mixture is a mixture in which the composition is not uniform throughout the mixture.
A homogeneous mixture is a mixture where the composition is uniform throughout the mixture.
Compounds
A chemical compound is a chemical substance that is made up of 2 or more molecules consisting of different types of atoms. The atoms of these different elements combined with each other in different ways to form a chemical compound. For example, a compound may have atoms of 1 element and 1 atom of another.
A compound is a substance formed when two or more different types of elements are chemically combined.
It has unique properties different from the elements that make it up.
Compounds are formed when two or more elements chemically combined in a specific ratio to create a new substance.
Compounds cannot be separated into their original elements by physical methods, only through chemical reactions.
Compounds have a specific formula that indicates the ratio of elements that they are composed of.
Note: All compounds are molecules but not all molecules are compounds.
Molecules
A molecule is the smallest unit of a substance that can exist independently and retain the chemical characteristics of that substance. It can be composed of the same or different types of atoms.
A molecule can be formed by the same or different elements, as long as the atoms are bonded together. A compound is a type of molecule, but it specifically refers to molecules made from two or more different elements. Oxygen is a molecule, and water is both molecule and compound.
A pure substance is a substance that has not been with anything else. Elements can be pure substances. For example, if you have a jar of oxygen that was not mixed with anything else.
Compounds can also be pure. Water is pure if it doesn’t contain anything other than water. Mixtures can contain two or more substances that are mixed up, but not chemically joined. A substance is a mixture if it can be separated into other substances. Mixtures can be made up of both elements and compounds. Air is a mixture that contains the elements nitrogen, oxygen and argon. It also contains the compounds water and carbon dioxide. Unlike compounds, you cannot write a formula for mixtures.
Electron Configuration of Atoms
Chemists are interested in the arrangement of electrons in atoms because electrons are involved in the formation and breaking of chemical bonds.
Electrons are arranged around the nucleus in each atom in layers called shells or energy levels. These are numbered, starting from the nucleus.
Each shell has a maximum possible number of electrons which is given by the formula, 2n2, where n is the number of the shell, and is called the principle quantum number.
The first or innermost shell can only contain 1 or 2 electrons.
The second shell has a maximum of 8 electrons.
The 3rd shell has a maximum of 18 electrons. However, for the first 20 elements, the third shell has only up to 8 electrons.
For the first 20 elements, the maximum number of electrons of the third shell of any atom is 8 (an octet). This number of electrons forms the most stable arrangement. The outermost electron shell is called the valence electron shell.
Electron configuration
The electron configuration is a series of numbers which shows the number and arrangement of the electrons orbiting the nucleus of an element’s atom. For example, the electron configuration of carbon is 2.4. This tells us that each carbon atom has two electrons in the first shell and 4 electrons in the outer (2nd) shell. If you add up the numbers of the configuration, you get the configuration, you get the atomic number of the element.
As you move across the period, an extra proton and electron is added to the atoms of each successive element. As one shell fills, the electrons go to the outer shells. At each step you move down the group, an extra shell is added.
The properties of atoms, including their ability to form chemical bonds, can be explained by the arrangement of electrons in the atom, particularly those in the outer valence shell. For most atoms, the most stable arrangement of electrons in the valence shell is 8 electrons arranged as 4 pairs. Atoms with an unstable outer valence shell will react readily, unlike those with a stable valence shell.
Each group on the periodic table contains elements with the same number of electrons in the outer shell of their atoms. This is why they have the same valency (combining power) and similar chemical properties.
Sometimes, we draw electron dot diagrams which only show the outermost shell of electrons. These are called Lewis electron dot diagrams after Gilbert Lewis, the scientist who first suggested the idea.