Chem and Water Quiz
What to know...
Basic atomic structure
Be familiar with common elements on the periodic table.
ions
compounds
valence electrons
isotopes
pH scale - know the pH of some typical household items.
what are buffers and how do buffers work
structure and polarity of water molecules
properties of water
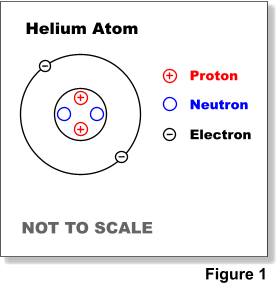
Atomic structure
extremely small, positively charged nucleus surrounded by a cloud of negatively charged electrons
protons, neutrons and electrons
Common elements on the table
Hydrogen (H)
Helium (He)
Carbon (C)
Oxygen (O)
Nitrogen (N)
Sodium (Na)
Aluminum (Al)
Potassium (K)
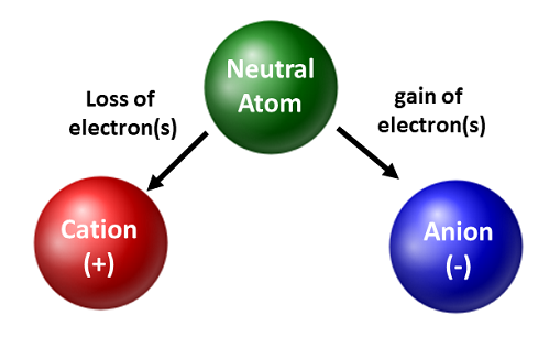
Ions
What is a ion? - an atom or group of atoms that carries a positive or negative electric charge as a result of having lost or gained one or more electrons
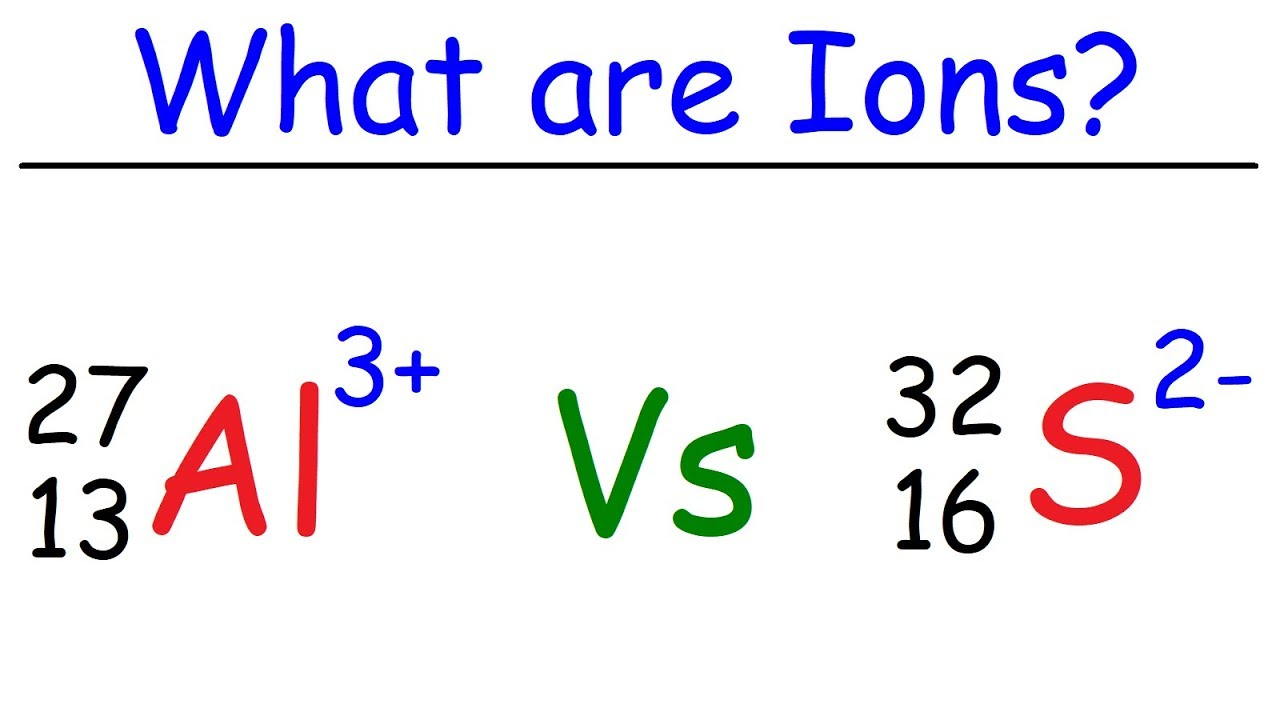
Compounds
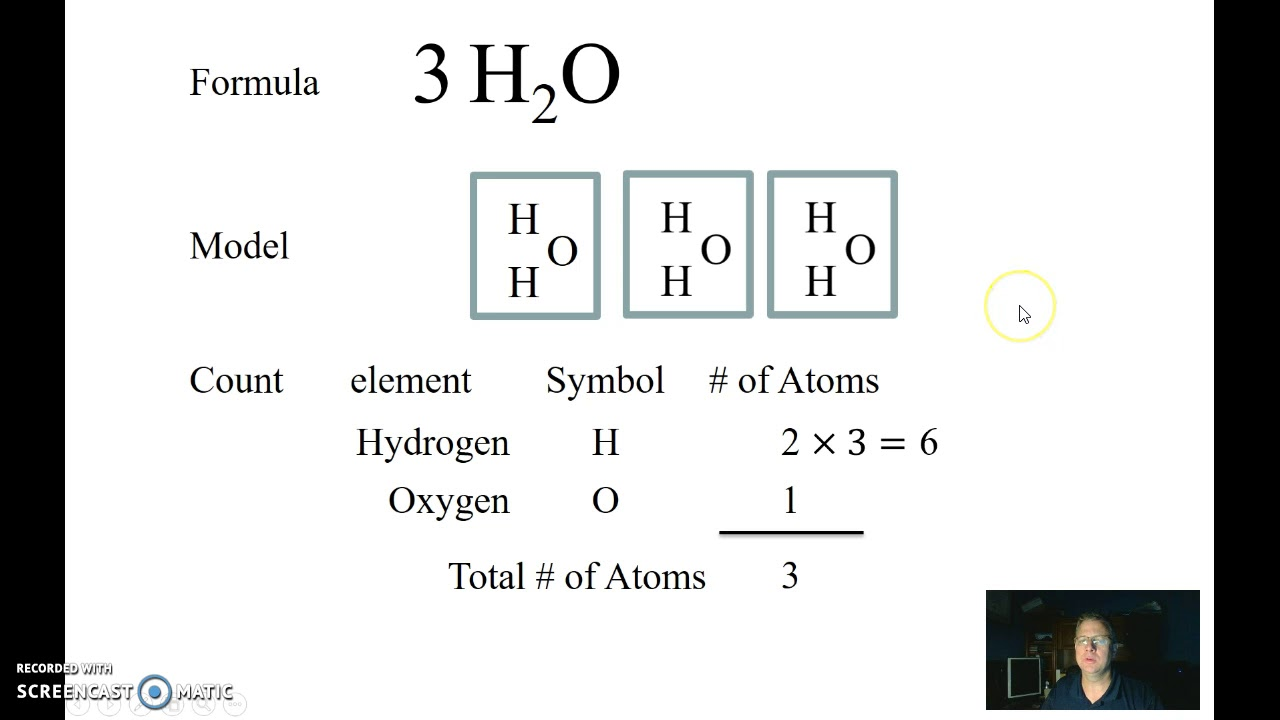
Definition: A substance made from two or more different elements that have been chemically joined. Examples of compounds include water (H2O), which is made from the elements hydrogen and oxygen, and table salt (NaCl), which is made from the elements sodium and chloride.
Valence Electron
What is it? - Valence electrons are electrons in the outermost shell of an atom, and that can participate in the formation of a chemical bond if the outermost shell is not closed
Examples - Examples include hydrogen (H), lithium (Li), and sodium (Na). Any element in group 18 has eight valence electrons (except for helium, which has a total of just two electrons). Examples include neon (Ne), argon (Ar), and krypton (Kr). Oxygen, like all the other elements in group 16, has six valence electrons.

Isotopes
What are Isotopes? - Isotopes are when a form of chemical element in which the atoms have the same number of protons but different number of neutrons. They share almost the same chemical properties, but differ in mass and therefore in physical properties. There are stable isotopes, which do not emit radiation, and there are unstable isotopes, which do emit radiation. The latter are called radioisotopes.

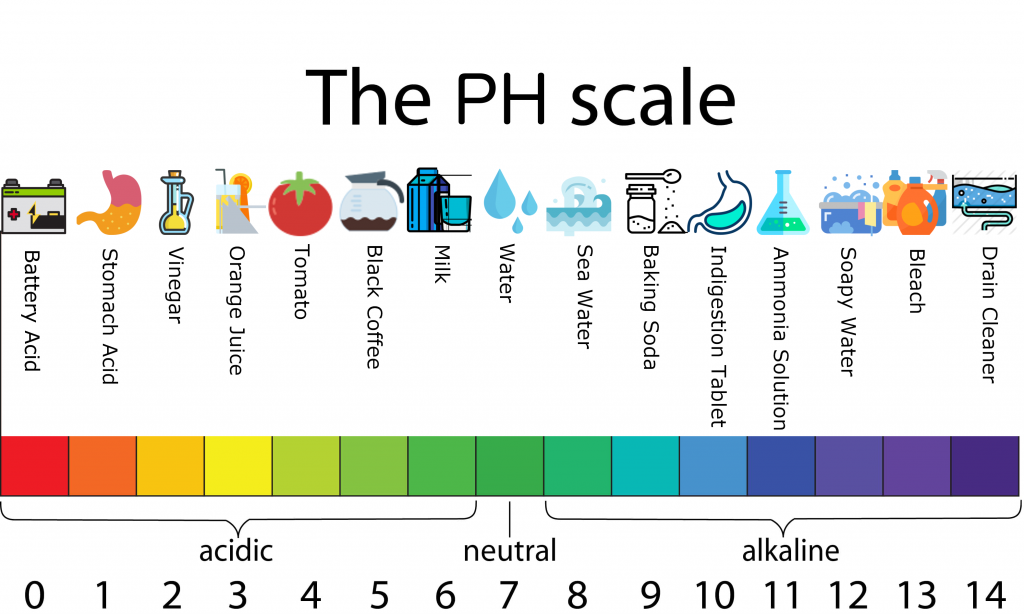
pH Scale
What is it? - The pH scale measures how acidic an object is. Objects that are not very acidic are called basic. The scale has values ranging from zero (the most acidic) to 14 (the most basic).
pH stands for “Potential of Hydrogen.” This term represents the hydrogen ions (H+) concentration in a solution
BUFFER - A buffer is a solution that can resist pH change upon the addition of an acidic or basic components. It is able to neutralize small amounts of added acid or base, thus maintaining the pH of the solution relatively stable
Everyday home objects and their pH scale.
Apples, 3, acidic
Bananas, 4, acidic
Borax, 9, basic
Water, 7, Neutral
Structure and Polarity of Water Molecules
Oxygen has a high electronegativity compared to hydrogen resulting in water molecule having polarity
Polarity allows molecules to form hydrogen bonds when oppositely charged region of two molecules interact
The term cohesion refers to molecules of the same type forming hydrogen bonds with another and adhesion refers to different types of molecules forming hydrogen bonds with one another
Living systems depending upon water properties, like surface tension
Polar = A structure having an uneven distribution of electron density'
Non-polar = Molecules that do not have any electrical charges or partial charge
Solubility = degree to which a substance dissolves in a solvent to make a solution
Properties of Water (similar to above)
COHESION = Cohesion is the act, state, or process of sticking together of like molecules or entities
SURFACE TENSION = The property of the surface of a liquid that allows it to resist an external force, due to the cohesive nature of its molecules
ADHESION = The binding of two or more cells together or the binding of a cell to a surfaces
CAPILLARY ACTION = It is the movement of a liquid through or along another material against an opposing force, such as gravity
DENSITY OF WATER = It is the movement of a liquid through or along another material against an opposing force, such as gravity
HIGH HEAT CAPACITY = One of water's most significant properties is that it takes a lot of energy to heat it