Electrolysis
Electrolysis is the passage of electric current through an aqueous solution or a liquid that causes it to decompose.
In a liquid state the ions are mobile.
Just heating, to melt, so there is no water present.
In electrolysis there are 2 types of electrodes:
Inert and Active
The elctrodes are points at which current enter and leave the electrolyte
Inert
Do not take part in electroylisis (reaction process) EG: Graphite & plantinum.
Active
Takes part electrolysis in the chemical reaction. (reaction process) EG: cupper
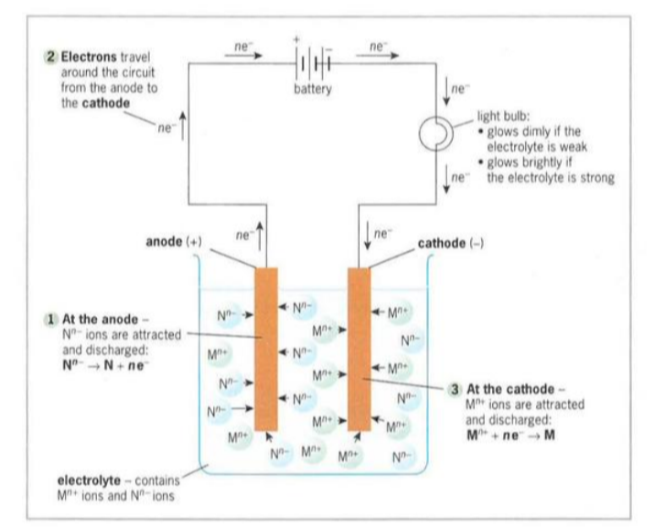
Electrodes connected to the negative terminal of the power supply is known as the cathode.
Electrodes connected to the positive terminal of the power supply is known as the anode.
Electrolites is a molten ionic compound or solution which contains mobile ions that can decompose when an electric current or electricity is passed through.
There are 2 types of electrolytes
Strong electrolytes completely ionize or dissociate in aqueous solution. Eg MgSO4, NaCl - ionic compound
Weak electrolytes partially ionize or dissociate in aqueous solution. EG: Ionic compounds- CH3COOH, NH4OH, CaOH
Non-electrolyte, these do not allow electricity to pass through (conduct electricity).EG- Molecular compounds: sugar
Discharge - the tendency of an ion to gain electrons and in turn to get reduced.
Preferential discharge - if more than one type of ion is attracted towards a particular electrode, then the one discharged is the ion that requires the least energy (reactivity series - the more reactive the ion is harder it is to discharge. Ease of discharge increases down the reactivity series
Electrochemical series - The electrochemical series of metals places metals in order of how easily they lose electrons (ionise) and can be used to predict certain chemical reactions. The ability of metal atoms give away (donate) electrons to another reactant increases going up the series. Therefore, the strength as a reducing agent increases up the series.
How a given property changes with time - rate of reaction
A catalyst alters the rate of a reaction but remain unchained at the end of the reaction.
Gasses have the largest amount of energy, solid have the least amount and liquids have a higher amount of energy than solid.
A salt is formed when some or all of the H+ ions in an acid are replaced by a metal or ammonium ion.
APPLICATION OF ELECTROLYSIS
Purification
The impure copper that is at the anode will go into solution and the pure copper will be deposited on the cathode. The impurities will fall to the bottom of the solution. (anode is made of impure copper). The cathode is a thin strip of copper.
Extraction of metal
Aluminum - because it is one of the more reactive metals
Blast furnace - extraction of Fe
Electroplating
Coating with a metal
is the process by which a thin layer of one metal is deposited on another metal by electrolysis.
Anodizing
Anodising is a process used to increase the thickness of an unreactive oxide layer on the surface of a metal, usually the aluminium oxide (AI,O,l layer on the surface of aluminium objects.
coated with oxygen
Factors affecting the amount of product formed
the charge
Mass
Electrochemical Series
Concentration
Type of electrode
Calculating the quantity
Mass of substances of substances deposited or liberated during electrolysis.
The quantity of products formed during electrolysis is proportional to the charge that had passed through the cell.
Q = It
Q - charge
unit coulomb C
I - current
Unit Ampere A
t - time
Unit seconds S
Faraday’s constant
96,500C mol^-1
The charge required to convert 1 mol of singly charged ions to atoms.
Step 1: write half equations
o
Look at number of electrons
FACTORS
Electrochemical Series
Concentration
Type of electrode
Monday: graded class work on Saponification and general organic chemistry
-