skin
1. Definition of the Integumentary System
Skin (cutis, integument) and its derivatives together form the integumentary system.
The skin:
Forms the external covering of the body
Is the largest organ
Accounts for 15–20% of total body mass
2. Layers of the Skin
The skin has two main layers:
1⃣ Epidermis
Histology
Keratinized stratified squamous epithelium
Growth
Grows continuously
Maintains normal thickness by desquamation
Embryologic origin
Derived from ectoderm
2⃣ Dermis
Histology
Dense connective tissue
Functions
Provides:
Mechanical support
Strength
Thickness to the skin
Embryologic origin
Derived from mesoderm
3. Hypodermis (Subcutaneous Layer)
Contains variable amounts of adipose tissue
Adipose tissue is arranged in lobules
Lobules are separated by connective tissue septa
Location:
Deep to the dermis
Equivalent to subcutaneous fascia in gross anatomy
Clinical variation:
Well-nourished individuals and people in cold climates → adipose tissue can be quite thick
4. Epidermal Derivatives (Skin Appendages)
Also called epithelial skin appendages or integumentary products
Includes:
Hair follicles
Hair
Sweat (sudoriferous) glands
Sebaceous glands
Nails
Mammary glands
5. Functions of the Integumentary System
The skin performs essential functions related to its external surface location.
Skin and its derivatives contain many different cell types that work together to help the body cope with the external environment.
Major Functions
1⃣ Barrier Function
Protects against:
Physical agents
Chemical agents
Biologic agents
Types of barriers:
Mechanical barrier
Permeability barrier
Ultraviolet barrier
2⃣ Immunologic Function
Provides immunologic information
Information obtained during antigen processing
Delivered to appropriate effector cells in lymphatic tissue
3⃣ Homeostasis
Participates in homeostasis by regulating:
Body temperature
Water loss
4⃣ Sensory Function
Conveys sensory information about the external environment
Information transmitted to the nervous system
5⃣ Endocrine Function
Skin performs endocrine functions by:
Secreting hormones
Secreting cytokines
Secreting growth factors
Converts precursor molecules into hormonally active molecules:
Example: Vitamin D₃
6⃣ Excretory Function
Functions in excretion via exocrine secretion of:
Sweat glands
Sebaceous glands
Apocrine glands
6. Classification of Skin
Skin is categorized as:
Thick skin
Thin skin
This classification reflects thickness and location.
8. Thick Skin
Found at:
Palms of the hands
Soles of the feet
Characteristics:
Areas subject to most abrasion
Hairless
Have a much thicker epidermal layer than other skin regions
This hairless skin is called thick skin.
9. Thin Skin
Found everywhere else
Has a much thinner epidermis
Contains hair follicles in almost all locations
10. Histologic Terminology Note
In histology:
The terms thick skin and thin skin actually refer only to the thickness of the epidermal layer, not the whole skin.
Epidermis
1. Epidermis: General Histology
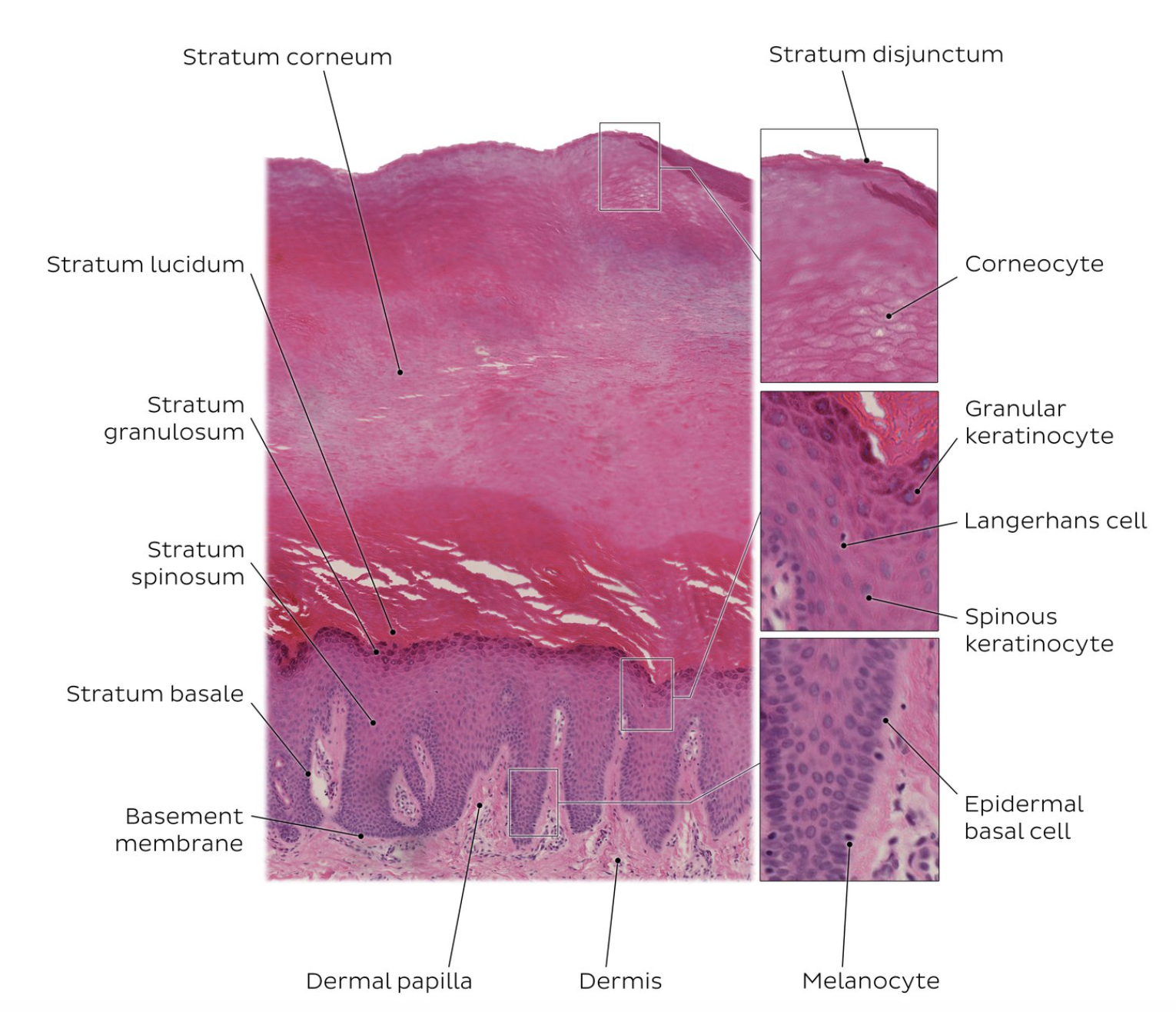
The epidermis is composed of stratified squamous epithelium.
Four layers are normally identified.
In thick skin, a fifth layer is present.
Epidermal layers (deep → superficial)
Stratum basale (stratum germinativum)
Stratum spinosum
Stratum granulosum
Stratum lucidum (only in thick skin)
Stratum corneum
2. Epidermal Cell Differentiation
Differentiation of epithelial cells is a specialized form of apoptosis.
Process
Begins with cell division in the stratum basale.
As cells migrate upward:
Cells in stratum granulosum show typical apoptotic nuclear morphology.
Includes DNA fragmentation.
However:
Typical apoptotic cellular fragmentation does not occur.
Instead:
Cells become filled with keratin filaments.
Eventually sloughed from the skin surface.
3. Stratum Basale (Stratum Germinativum)
Structure
Single layer of cells
Rest on the basal lamina
Cell Characteristics
Contain stem cells
Produce keratinocytes via mitotic division
Cell morphology
Cells are:
Small
Cuboidal to low columnar
Have less cytoplasm than cells of upper layers
Nuclei closely spaced
Staining property
Basophilic cytoplasm
Closely spaced nuclei + basophilic cytoplasm → noticeable basophilia
Melanin
Basal cells contain melanin in cytoplasm
Melanin is transferred from melanocytes
Melanocytes are interspersed in this layer
Cell Junctions
Cells connected to each other and keratinocytes by desmosomes
Cells connected to basal lamina by hemidesmosomes
Cell Migration
New keratinocytes formed by mitosis
Cells move upward into the next layer
Migration continues until cells become mature keratinized cells
Finally sloughed off at skin surface
4. Stratum Spinosum
Thickness
Several cells thick
Cell Characteristics
Keratinocytes larger than in stratum basale
Spiny appearance
Cells exhibit numerous cytoplasmic processes (spines)
Cell junctions
Processes attach to adjacent cells by desmosomes
Light microscopy feature
Desmosome site appears as slight thickening
Called the node of Bizzozero
Cause of visible spines
During tissue preparation:
Cells shrink
Intercellular spaces expand
Makes spines more visible
Alternate name
Cells often called prickle cells
Maturation changes
As cells move toward the surface:
Increase in size
Become flattened parallel to surface
In superficial cells:
Nuclei elongate
Change from ovoid → elongated
Reflect acquired squamous shape
5. Stratum Granulosum
Position
Most superficial layer of the nonkeratinized portion of epidermis
Thickness
1–3 cells thick
Major feature
Keratinocytes contain numerous keratohyalin granules
Keratohyalin granules
Contain:
Cysteine-rich proteins
Histidine-rich proteins
These proteins are precursors of filaggrin.
Filaggrin function
Aggregates keratin filaments
Present in cornified cells of stratum corneum
Granule characteristics
Irregular shape
Variable size
Staining
Intensely basophilic
Easily seen in routine histologic sections
6. Stratum Corneum
Cell type
Anucleate squamous cells
Cell contents
Cells filled largely with keratin filaments
Differentiation
Cells are the most differentiated cells in the skin
Transition from granulosum
Abrupt transition between:
Nucleated cells of stratum granulosum
Flattened anucleate cells of stratum corneum
Cellular changes
Cells:
Lose nucleus
Lose cytoplasmic organelles
Become filled with keratin filaments
Plasma membrane
Thick plasma membrane
Lipid layer
External surface coated with extracellular lipids
Lipids form major component of the epidermal water barrier
Thickness
Layer varies most in thickness
Thickest in thick skin
Role in thick vs thin skin
Thickness of this layer is the principal difference between thick skin and thin skin
Adaptation to friction
Becomes thicker at sites of unusual friction
Example:
Calluses
Palms
Fingertips
7. Stratum Lucidum
Occurrence
Normally visible only in thick skin
Histologic appearance
Appears refractile under light microscopy
May stain poorly
Cell characteristics
Contains eosinophilic cells
Keratinization stage
Keratinization well advanced
Cellular degeneration
During keratinization:
Nucleus disappears
Cytoplasmic organelles disappear
Cell fills with keratin
Cells of the Epidermis :
Cell Type | Function | Approximate Percentage |
|---|---|---|
Keratinocytes | Specialized epithelial cells that separate the organism from the external environment | ~85% |
Melanocytes | Pigment-producing cells | ~5% |
Langerhans’ cells | Antigen-presenting cells involved in immune signaling | ~2–5% |
Merkel’s cells | Sensitive mechanoreceptor cells associated with sensory nerve endings | ~6–10% |
2. Keratinocytes
Predominant epidermal cell type
Origin: Stratum basale
Two main activities
Production of keratins (cytokeratins)
Participation in formation of the epidermal water barrier
3. Keratin Proteins
Keratins = major heteropolymeric structural proteins of the epidermis
Keratins form intermediate filaments
They constitute almost 85% of fully differentiated keratinocytes
4. Ultrastructure of Basal Keratinocytes
Basal keratinocytes contain:
Numerous free ribosomes
7–9 nm intermediate (keratin) filaments
Small Golgi apparatus
Mitochondria
Rough endoplasmic reticulum (rER)
Cytoplasmic staining
Cytoplasm appears basophilic due to large number of free ribosomes
5. Keratin Filament Formation
Ribosomes synthesize keratin proteins.
Keratin proteins assemble into keratin filaments.
Terminology
Intermediate filaments → tonofilaments
In the stratum spinosum
Tonofilaments aggregate into bundles.
These bundles are called:
Tonofibrils
Cytoplasmic change
Cytoplasm becomes eosinophilic
Due to accumulation of tonofibrils.
6. Keratohyalin Granules
Keratohyalin granules contain intermediate filament–associated proteins that help aggregate keratin filaments.
Major proteins
Filaggrin
Trichohyalin
Synthesis
Keratinocytes in the upper stratum spinosum begin synthesizing keratohyalin granules.
Clinical relevance
Expression of filaggrin can be used as a clinical marker for initiation of the final stage of apoptosis.
7. Keratinization
When keratohyalin granules increase:
Granule contents released into cytoplasm
Filaggrin + trichohyalin promote aggregation of keratin filaments into tonofibrils
This converts:
Granular cells → Cornified cells
Process name
Keratinization
Timing
Occurs in 2–6 hours
Keratin produced
Called soft keratin
(Contrast: hard keratin of hair and nails)
8. Cellular Changes During Keratinization
Transformation of a granular cell into a keratinized cell involves:
Breakdown of nucleus
Breakdown of cytoplasmic organelles
Thickening of plasma membrane
pH change
pH decreases:
~7.17 in stratum granulosum
pH 4.5–6.0 at surface of stratum corneum
9. Desquamation of Surface Keratinocytes
Surface keratinocytes are continuously exfoliated.
Regulation
Desquamation is regulated by:
Proteolytic degradation of desmosomes
10. Lamellar Bodies
Keratinocytes in stratum spinosum producing keratohyalin granules also produce:
Lamellar bodies (membrane-coating granules)
Characteristics
Membrane-bound organelles
Tubular or ovoid shaped
Unique to mammalian epidermis
Lipid components synthesized
Glycosphingolipids
Phospholipids
Ceramides
Enzymes present
Acid sphingomyelinase
Secretory phospholipase A₂
Also contain
Proteases including:
SC chymotryptic enzyme
Cathepsin D
Acid phosphatase
Glycosidases
Protease inhibitors
11. Secretion of Lamellar Bodies
Lamellar body contents are:
Released by exocytosis
Into intercellular spaces
Between stratum granulosum and stratum corneum
Function
Forms intercellular lipid lamellae
These lamellae are responsible for formation of the:
Epidermal water barrier
12. Epidermal Water Barrier
Essential for mammalian “dry” epithelia.
Main role
Maintains body homeostasis
Formed by two factors
Insoluble proteins deposited on inner plasma membrane
Lipid layer attached to outer membrane surface
13. Structure of the Epidermal Barrier
A. Cell Envelope (CE)
Thickness
~15 nm
Location
Inner surface of plasma membrane
Function
Provides mechanical strength
Thickness increases in areas with high mechanical stress
Examples:
Lip
Palm of hand
Sole of foot
Formation
CE formed by cross-linking:
Small proline-rich (SPR) proteins
Larger structural proteins
Major proteins
Cystatin
Desmoplakin
Elafin
Envoplakin
Filaggrin
Involucrin
Keratin chains
Loricrin
Important fact
Loricrin
Major structural CE protein
Accounts for ~80% of total CE protein mass
Additional feature
26 kDa insoluble protein
Has highest glycine content of any known protein
B. Lipid Envelope
Thickness
~5 nm
Attachment
Attached to cell surface via ester bonds
Major components
Ceramides
Cholesterol
Free fatty acids
Key molecule
Acylglucosylceramide
Function:
Forms monomolecular “Teflon-like” coating on cell surface.
Additional roles of ceramides
Participate in cell signaling
Induce cell differentiation
Trigger apoptosis
Reduce cell proliferation
14. Maintenance of the Epidermal Barrier
Barrier continuously maintained by keratinocytes undergoing terminal differentiation.
Lamellar lipid arrangement
Lamellae may appear as:
Recognizable discs
Broad sheets or layers in intercellular space
Dermis –
1. Epidermal–Dermal Junction
Appears:
Uneven boundary in LM (except thinnest skin)
Structure:
Dermal papillae
Finger-like connective tissue projections
Project into epidermis
Epidermal (rete) ridges
Downward projections of epidermis into dermis
2. Sectional Appearance (Exam Trap)
Perpendicular section:
Shows dermal papillae clearly
Parallel section:
Epidermis appears continuous
Contains circular islands of connective tissue
These = cross-sections of dermal papillae
3. Functional Significance
Increased interface:
Enhances attachment between epidermis and dermis
4. Dermal Ridges
Arrangement:
Parallel
Dermal papillae lie between ridges
Function:
Form surface patterns → epidermal grooves & ridges
Clinical relevance:
Basis of:
Dermatoglyphics (fingerprints, footprints)
Genetically unique
5. Distribution of Ridges & Papillae
Most prominent in:
Thick skin
Palmar & plantar surfaces
6. Thick Skin Characteristics (Related Concept)
Basal surface:
Much larger than free surface
Result:
More cells enter stratum corneum per unit time
Leads to:
Thicker cornified layer
7. Attachment Mechanisms
Hemidesmosomes
Function:
Attach epidermis to basal lamina
Mechanism:
Link:
Intermediate filaments → basal lamina
Focal Adhesions
Function:
Anchor:
Actin filaments → basal lamina
Additional Feature (TEM)
Basal epidermal cells:
Show irregular cytoplasmic protrusions
Increase attachment surface area
8. Layers of Dermis
A. Papillary Layer
Location:
Superficial dermis
Tissue:
Loose connective tissue
Fibers:
Thin collagen fibers
Types:
Type I and Type III collagen
Elastic fibers:
Thread-like, irregular network
Contains:
Dermal papillae
Blood vessels
Do NOT enter epidermis
Nerve processes
Either terminate in dermis
Or penetrate basal lamina → epidermis
Note:
Sensory endings concentrated → prominent in papillae
B. Reticular Layer
Location:
Deep to papillary layer
Characteristics:
Thicker
Less cellular
Thickness:
Varies by body region
9. Special Features of Reticular Layer
Smooth Muscle
Found in:
Areola, penis, scrotum, perineum
Arrangement:
Loose plexus in deep reticular layer
Function:
Causes skin puckering (especially erectile organs)
Arrector Pili Muscle
Origin:
From dermal smooth muscle
Attachment:
Connects:
Hair follicle → superficial dermis
Function:
Contraction causes:
Hair erection
Goose flesh
10. Hypodermis (Subcutaneous Layer)
Structure
Located:
Deep to reticular layer
Components:
Adipose tissue (panniculus adiposus)
Loose connective tissue
Functions
Energy storage
Insulation
Thickness:
Variable
Increased in cold climates
Terminology
Also called:
Hypodermis
Subcutaneous fascia
11. Muscle in Hypodermis
Striated Muscle
Present in some areas
Special layer:
Panniculus carnosus
Prominent in animals
Vestigial in humans
Human Remnants
Present in:
Neck, face, scalp
Forms:
Platysma
Muscles of facial expression
Melanocytes
1. Origin
Neural crest–derived cells
2. Migration (Embryology)
Melanocyte precursors:
Migrate from neural crest
Enter developing epidermis
3. Location in Skin
Found in:
Stratum basale
Distribution:
Scattered among basal keratinocytes
4. Epidermal–Melanin Unit
Definition:
Functional association between:
1 melanocyte + keratinocytes
In humans:
≈ 1 melanocyte : 36 keratinocytes
Ratio range:
1:4 → 1:40 (or higher)
Notes:
Constant in all races
Influenced by:
Age
Environmental factors (sun exposure)
5. Adult Stem Cell Pool
Location:
Hair follicle – follicular bulge
Cell type:
Undifferentiated melanocyte stem cells
6. Molecular Regulation
Key gene:
Pax3 gene (PAX transcription factor family)
Pax3 function:
Activates:
MITF (microphthalmia transcription factor)
MITF role:
Essential for:
Melanocyte development
Melanocyte differentiation (melanogenesis)
7. Proliferation
Melanocytes:
Replicate throughout life
Slower rate than keratinocytes
8. Cell Type & Morphology
Type:
Dendritic cells
Cell body:
Located in stratum basale
Processes:
Extend between keratinocytes into:
Stratum spinosum
9. Cell Junctions
Do NOT form desmosomes with keratinocytes
Near basal lamina:
Have structures resembling:
Hemidesmosomes
12. Main Function
Produce and secrete:
Melanin
Role:
Protection against:
Nonionizing ultraviolet radiation
13. Melanin Synthesis (Melanogenesis)
Substrate:
Tyrosine
Intermediate:
3,4-dihydroxyphenylalanine (DOPA)
Final product:
Melanin
Enzyme:
Tyrosinase
14. Site of Melanin Production
Occurs in:
Premelanosomes
Characteristics:
Membrane-bound
Lysosome-related organelles
Derived from:
Golgi apparatus
15. Regulation of Melanin Production
Hormone:
Melanocyte-stimulating hormone (MSH)
Source:
Anterior pituitary
Receptor:
Melanocortin 1 receptor (MC1R)
Mechanism:
G-protein signaling cascade
↑ Tyrosinase activity
↑ Melanin synthesis
16. Melanosome Development Stages
Premelanosomes / Early melanosomes
Low melanin content
Organized internal structure (visible on TEM)
Progressive melanin deposition:
Structure becomes obscured
Mature melanosomes
Electron-opaque granules
17. Intracellular Distribution
Premelanosomes:
Near Golgi apparatus
Nearly mature melanosomes:
At base of cell processes
Mature melanosomes:
At ends of processes
18. Transfer to Keratinocytes
Mechanism:
Pigment donation
Process:
Keratinocytes phagocytose:
Tips of melanocyte processes
Type:
Cytocrine secretion
Includes:
Transfer of some cytoplasm
19. Fate of Melanosomes
Degradation:
Via macroautophagy
Variation:
Darker skin:
Slow degradation
Melanosomes remain discrete
Lighter skin:
Faster degradation
Langerhans’ Cells
1. Definition
Antigen-presenting cells (APCs) of the epidermis
Dendritic-appearing cells
2. Origin
Derived from:
Common lymphoid progenitor (CLP) cells in bone marrow
Pathway:
Bone marrow → bloodstream → epidermis
Differentiate into:
Immunocompetent cells
3. Location
Found in:
Epidermis
Readily seen in:
Stratum spinosum (with special staining)
4. Function
Immunosurveillance of epidermis
Process:
Encounter antigens entering through skin
Phagocytose and process antigen
Display antigen on cell surface
Migrate to regional lymph node
Interact with T lymphocytes
Part of:
Mononuclear phagocyte system (MPS)
5. Cell Ratio
Ratio in normal epidermis:
1 Langerhans cell : 53 other epidermal cells
7. Special Staining / Identification
Identified using:
Gold chloride impregnation
Immunostaining for CD1a molecules
8. Morphology
Dendritic processes
Similar to melanocytes
Cell junctions:
No desmosomes with keratinocytes
10. Surface Markers / Receptors
Express:
MHC I molecules
MHC II molecules
Fc receptors for IgG
Complement C3b receptors
CD1a molecules (variable amounts)
Langerhans’ Cells –
1. Definition
Antigen-presenting cells (APCs) of the epidermis
Dendritic-appearing cells
2. Origin
Derived from:
Common lymphoid progenitor (CLP) cells in bone marrow
Pathway:
Bone marrow → bloodstream → epidermis
Differentiate into:
Immunocompetent cells
3. Location
Found in:
Epidermis
Readily seen in:
Stratum spinosum (with special staining)
4. Function
Immunosurveillance of epidermis
Process:
Encounter antigens entering through skin
Phagocytose and process antigen
Display antigen on cell surface
Migrate to regional lymph node
Interact with T lymphocytes
Part of:
Mononuclear phagocyte system (MPS)
5. Cell Ratio
Ratio in normal epidermis:
1 Langerhans cell : 53 other epidermal cells
6. Light Microscopy (H&E)
Cannot be reliably identified in routine H&E sections
Features (when noted):
Nucleus:
Darkly stained (hematoxylin)
Cytoplasm:
Clear
7. Special Staining / Identification
Identified using:
Gold chloride impregnation
Immunostaining for CD1a molecules
8. Morphology
Dendritic processes
Similar to melanocytes
Cell junctions:
No desmosomes with keratinocytes
9. Electron Microscopy (TEM) Features
Nucleus:
Irregular, indented (uneven nuclear profile)
Cytoplasmic organelles:
Birbeck granules
Shape:
Tennis racquet–shaped
Appearance:
Rod with bulbous expansion at one end
10. Surface Markers / Receptors
Express:
MHC I molecules
MHC II molecules
Fc receptors for IgG
Complement C3b receptors
CD1a molecules (variable amounts)
Merkel’s Cells –
1. Definition
Epidermal cells involved in cutaneous sensation
2. Location
Found in:
Stratum basale
4. General Characteristics
Dendritic cells
Possess:
Antigenic markers of both epidermal and neural type
5. Distribution
Most abundant in:
Areas of acute sensory perception
Example:
Fingertips
6. Cell Junctions
Connected to keratinocytes by:
Desmosomes
7. Cytoskeleton
Contain:
Intermediate (keratin) filaments in cytoplasm
8. Light Microscopy Features
Nucleus:
Lobed
Cytoplasm:
Denser than melanocytes and Langerhans’ cells
9. Cytoplasmic Contents
May contain:
Melanosomes
Characteristic feature:
80-nm dense-cored neurosecretory granules
Resemble granules of:
Adrenal medulla
Carotid body
10. Association with Nerve Fibers
Closely associated with:
Expanded terminal bulb of afferent myelinated nerve fibers
Nerve fiber features:
Loses Schwann cell covering
Penetrates basal lamina
Expands into:
Plate-like ending (disc receptor)
11. Merkel’s Corpuscle
Formed by:
Merkel cell + nerve terminal
Function:
Sensitive mechanoreceptor
12. Key Exam Points
Located in stratum basale
Desmosomes present (contrast with melanocytes & Langerhans cells)
Neurosecretory granules (80 nm) = hallmark
Associated with afferent nerve ending
Forms Merkel’s corpuscle → mechanoreception
Skin – Nerve Supply
1. General Organization
Skin contains:
Sensory receptors
Peripheral terminals of sensory nerves
Motor nerve endings to:
Blood vessels
Arrector pili muscles
Sweat glands
2. Free Nerve Endings
Definition
Most numerous receptors in epidermis
Location
Terminate in:
Stratum granulosum
Structure
“Free” because:
No connective tissue capsule
No Schwann cell covering
Function
Detect:
Fine touch
Heat
Cold
Pain
Note:
No distinct morphology for each modality
Association with Hair
Networks surround:
Hair follicles
Attach to:
External root sheath
Function:
Act as mechanoreceptors (hair movement)
Specialization
Around tactile hairs (vibrissae):
Highly specialized
Each hair has specific cortical representation
3. Encapsulated Nerve Endings
General Feature
Surrounded by:
Connective tissue capsule
Types
Pacinian corpuscles
Meissner’s corpuscles
Ruffini’s corpuscles
4. Pacinian Corpuscles
Function
Detect:
Pressure
Vibration
Type:
Deep pressure mechanoreceptors
Location
Deep dermis
Hypodermis
Also found in:
Connective tissue
Joints
Periosteum
Internal organs
Especially:
Fingertips
Structure
Central:
Myelinated nerve fiber
Entry:
Enters capsule with myelin intact
Inside:
Myelin lost after 1–2 nodes
Axon becomes unmyelinated
Core (Inner Core)
Formed by:
Flattened Schwann cell lamellae
Outer Core (Capsule)
Concentric lamellae
Between lamellae:
Fluid-filled spaces
Sparse collagen fibrils
Occasional capillaries
Mechanism
Pressure → displacement of lamellae
→ Axon depolarization
5. Meissner’s Corpuscles
Function
Touch receptors
Sensitive to:
Low-frequency stimuli
Location
Dermal papillae
Just beneath:
Epidermal basal lamina
Distribution
Hairless skin
Lips
Palmar surfaces
Volar surfaces of fingers and toes
Structure
Contains:
1–2 unmyelinated nerve endings
Derived from myelinated fibers
Arrangement:
Spiral course within corpuscle
Cellular Component
Flattened Schwann cells
Form:
Irregular lamellae
6. Ruffini’s Corpuscles
Function
Detect:
Stretch
Torque
Stimulus:
Mechanical displacement of collagen fibers
Type
Simplest encapsulated mechanoreceptors
Structure
Capsule
Thin:
Connective tissue capsule
Contains:
Fluid-filled space
Collagen Fibers
Pass through capsule from surrounding CT
Neural Component
Single myelinated fiber enters
Loses myelin inside capsule
Branches into:
Dense arborization of axonal endings
Each ending:
Knob-like bulb
Arrangement
Axonal endings:
Dispersed and intertwined
Mechanism
Respond to:
Displacement of collagen fibers
Due to:
Sustained mechanical stress
Physiological Type
Rapidly adapting (phasic receptors)
Generate:
Action potentials at:
Beginning and end of stimulus
Hair Follicles and Hair –
1. Definition
Hair follicle = invagination of epidermis
Site of:
Hair formation
2. Distribution
Present:
Almost entire body
Absent from:
Palmar surfaces (hands)
Plantar surfaces (feet)
Sides of hands & feet
Lips
Region around urogenital orifices
3. Hormonal Influence
Hair distribution influenced by:
Sex hormones
At puberty:
Male:
Thick pigmented facial hair
Both sexes:
Pubic & axillary hair
Aging:
Male → hairline recedes
Both sexes → scalp hair thins
Due to ↓ estrogen & estrogen-like hormones
4. Hair Follicle Function
Responsible for:
Production and growth of hair
5. Hair Color
Determined by:
Type and amount of melanin
6. Follicle Variation
Depends on:
Growing vs resting phase
Growing follicle:
Most elaborate (exam focus)
7. Regions of Hair Follicle
1. Infundibulum
Extends:
Surface opening → sebaceous gland opening
Part of:
Pilosebaceous canal
Function:
Route for:
Sebum discharge
2. Isthmus
Extends:
Infundibulum → arrector pili insertion
3. Follicular Bulge
Location:
Near arrector pili insertion
Contains:
Epidermal stem cells
4. Inferior Segment
Present in:
Growing follicle
Shape:
Uniform diameter except base → expands into:
Hair bulb
8. Hair Bulb & Dermal Papilla
Hair Bulb
Expanded base of follicle
Contains:
Hair matrix
Dermal Papilla
Composition:
Vascularized loose connective tissue
Invaginates into:
Hair bulb
Hair Matrix
Composed of:
Matrix cells
Location:
Adjacent to dermal papilla
Function:
Rapid division → hair growth
Origin:
Cells migrated from:
Follicular bulge (stem cells)
Melanocytes (Matrix)
Present in:
Germinative layer
Function:
Transfer melanosomes → developing hair cells
Differentiation
Matrix cells → form:
Hair shaft cells
Internal root sheath
9. Root Sheaths
A. Internal Root Sheath (IRS)
Function:
Surrounds deep part of hair
Structure:
Multilayered
Layers (must remember)
Henle’s layer
Outer layer
Single layer of cuboidal cells
Huxley’s layer
Middle layer
Single or double layer of flattened cells
Forms:
Middle plate
Cuticle of IRS
Squamous cells
Faces:
Hair shaft
B. External Root Sheath (ERS)
Represents:
Down-growth of epidermis
Continuous with:
Epidermal layers
10. Key Relationships
Henle’s layer:
In contact with:
External root sheath
IRS cuticle:
Faces:
Hair shaft
Hair Follicle & Hair –
1. Follicular Bulge (Stem Cell Niche)
Located in external root sheath at level of arrector pili muscle insertion
Contains relatively undifferentiated epithelial cells
Called follicular bulge
Function
Acts as a niche of epidermal stem (ES) cells
ES cells:
Self-renew
Differentiate into specific lineages
Role under normal conditions
Provide stem cells for:
Hair follicle growth
Hair matrix
Internal root sheath
Cortex
Medulla
Sebaceous glands
Important exam point
Do NOT contribute to basal stem cells of epidermis normally
Activated in injury → help in skin regeneration
2. External Root Sheath – Key Relations
Contains:
Insertion of arrector pili muscle
Origin of sebaceous duct and gland
Nerve endings surround it at this level
3. Keratinization of Hair
Occurs in keratogenous zone
Located in lower third of follicle
Process
Cells:
Differentiate
Lose organelles
Become packed with keratin intermediate filaments
Final product:
Fully keratinized hard keratin (hair shaft)
Internal Root Sheath
Contains soft keratin
Does NOT emerge with hair
Breaks down at isthmus level
4. Supporting Structures
Glassy membrane
Thick basal lamina
Separates follicle from dermis
Surrounding tissue:
Dense irregular connective tissue
Arrector pili muscle
Attached near follicular bulge
Landmark for stem cell niche
5. Hair Structure (Hair Shaft Layers)
A. Medulla
Central core
Large, loosely connected keratinized cells
Contains soft keratin
Present only in thick hairs
B. Cortex
Largest layer (~80% of hair mass)
Located outside medulla
Contains:
Hard keratin intermediate filaments
Keratin-associated proteins (KAPs)
Function
Provides:
Strength (disulfide cross-linking)
Texture
Elasticity
Color
Pigment
Melanin produced by melanocytes in hair bulb germinative layer
C. Cuticle of Hair Shaft
Outermost layer
Made of overlapping keratinized squamous cells
Cells resemble:
Fish scales / roof tiles
Free edges point away from follicle
Function
Protects against:
Physical damage
Chemical damage
Determines hair porosity