Quantitative Chemistry
moles
avogadro’s number is the number of atoms in 12 g of carbon-12 - 6.02 × 1023
1 mole is is that many particles of a substance (particles here could mean atoms, ions, electrons, molecules, formulae etc). this is a constant used for ease. the mole is a unit for the amount of a substance. it allows predictions to be made about the masses of different substances involved in a reaction. it’s measured in mol
number of particles = avogadro’s number × the amount of substance in mol
RAM/Ar - relative atomic mass. the average mass of one atom / 1/12 of the mass of one atom of carbon-12, aka the average mass of all isotopes of an element. it’s the number above the atomic symbol on the periodic table. it can be calculated with:
(mass x abundance) + (mass x abundance) etc.. / total abundance
RFM/Mr - relative molecular/formula mass. the average mass of a molecule/compound 1/12 of the mass of one atom of carbon-12. calculate by adding the RAMs of each atom in the molecule together. but also relative formula mass. e.g:
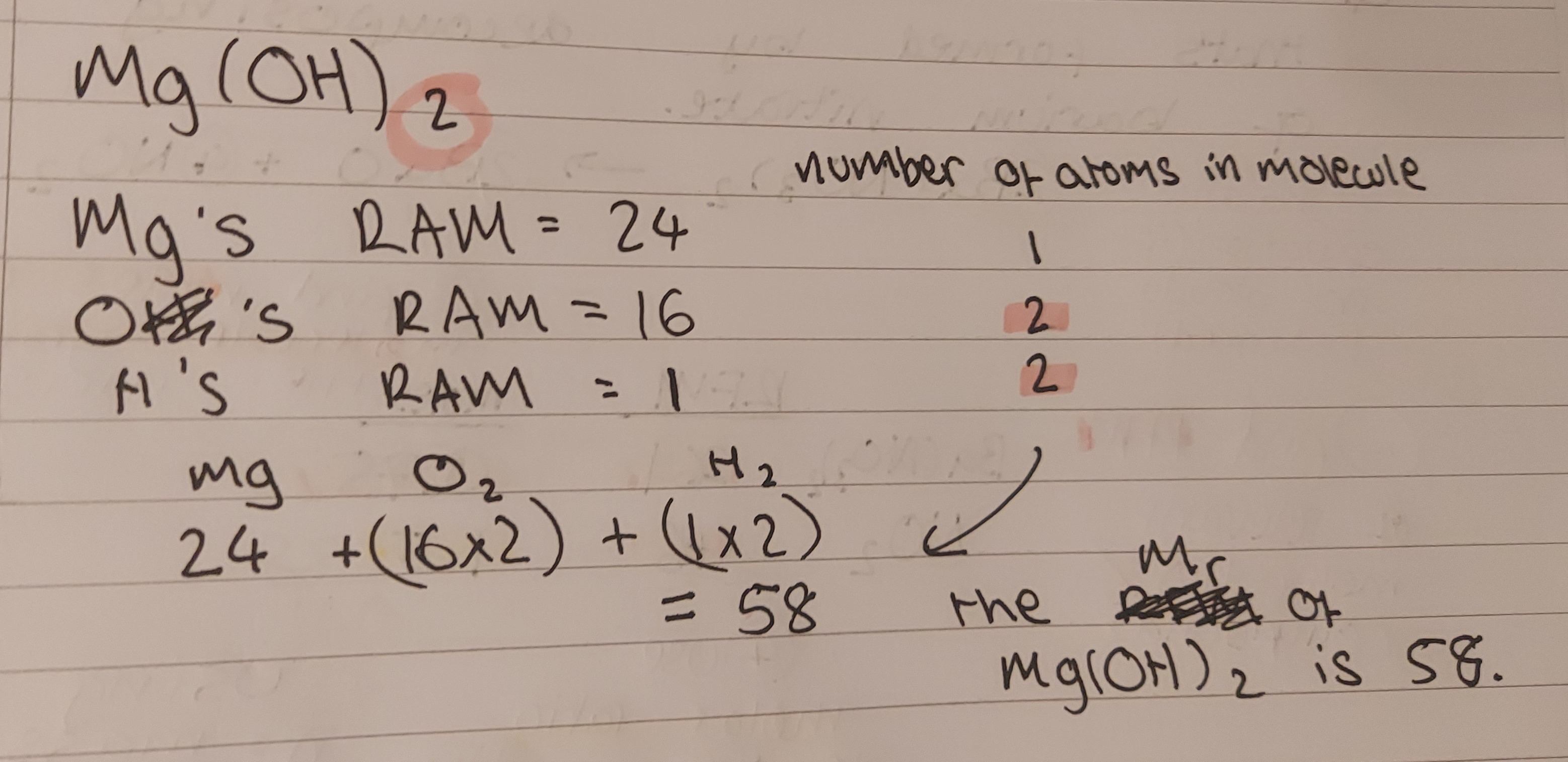
if a molecule has a large number before it, remember to multiply the entire RFM by that number. that number signifies the number of moles of the substance needed in a balanced reaction.
a molecule is a group of 2 or more atoms - of the same or of different types - chemically bonded. e.g. Cl2 is a molecule and an element. mostly refers to covalent structures - you get simple covalent molecules but ionic compounds.
molar mass of a substance is the mass of 1 mol of it. molar mass is equal to a substances RAM or RFM, but measured in g/mol. e.g oxygen has an RAM of 16. it’s molar mass is therefore 16 g/mol (there’s 16g of oxygen in every mole of oxygen)
number of moles in a substance = mass of substance (g - MUST BE IN G) / Ar OR Mr
the concept of moles can be applied to chemical equations. for example in the equation
CH4 + 2O2 → CO2 + 2H2O
1 mole of methane is reacting with 2 moles of oxygen to product 1 mole of carbon dioxide and 2 moles of water. the small numbers are representing bonds (e.g. you need 4 hydrogen atoms to complete the outer ring of electrons on a carbon atom, hence CH4)
masses can be used to form equations, based on molar ratios. e.g:

a very common type of question is to do with reacting masses - you’ll be asked to find the mass of a certain molecule based on the mass of another molecule in the reaction. e.g:

atom economy
atom economy measures how much of the reactants end up as a useful product.
high atom economy means a process is more sustainable + creates less waste in the form of byproducts (unwanted waste products).
transition metals often used as catalysts in reactions to increase atom economy by decreasing activation energy by providing an alternative reaction pathway. lower energy requirements = less energy used = cheaper, more sustainable, less waste.
% atom economy = (Mr of desired product / Mr of all products or all reactants*) × 100
* why? conservation of mass - you’ll create x amount of products from x amount of reactants, even if it doesn’t always seem that way - e.g. a product could be a gas which is hard to weigh unless you’re making a point to collect it.
e.g:

limiting reactants
in a reaction, you may have a reactant in excess (more than needed for reaction to occur) to ensure all of other reactants reacts → maximises product
the limiting reactant is the one with fewer moles:
once it’s used up, no more product can be made
if you had more of it, you’d be able to make more product. it’s limiting the amount that could be made
if the molar ratio of the balanced equation is 1:1:1 (:1) this is simple:
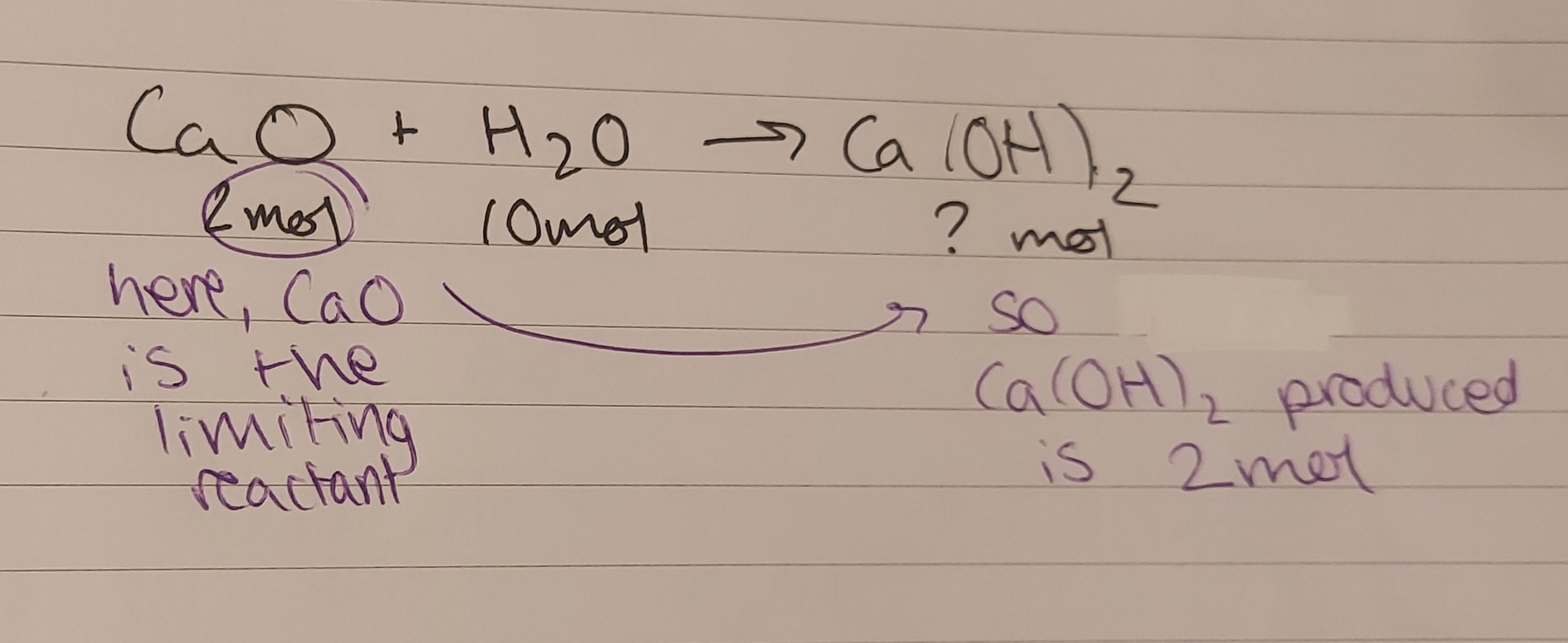
however this is often not the case:
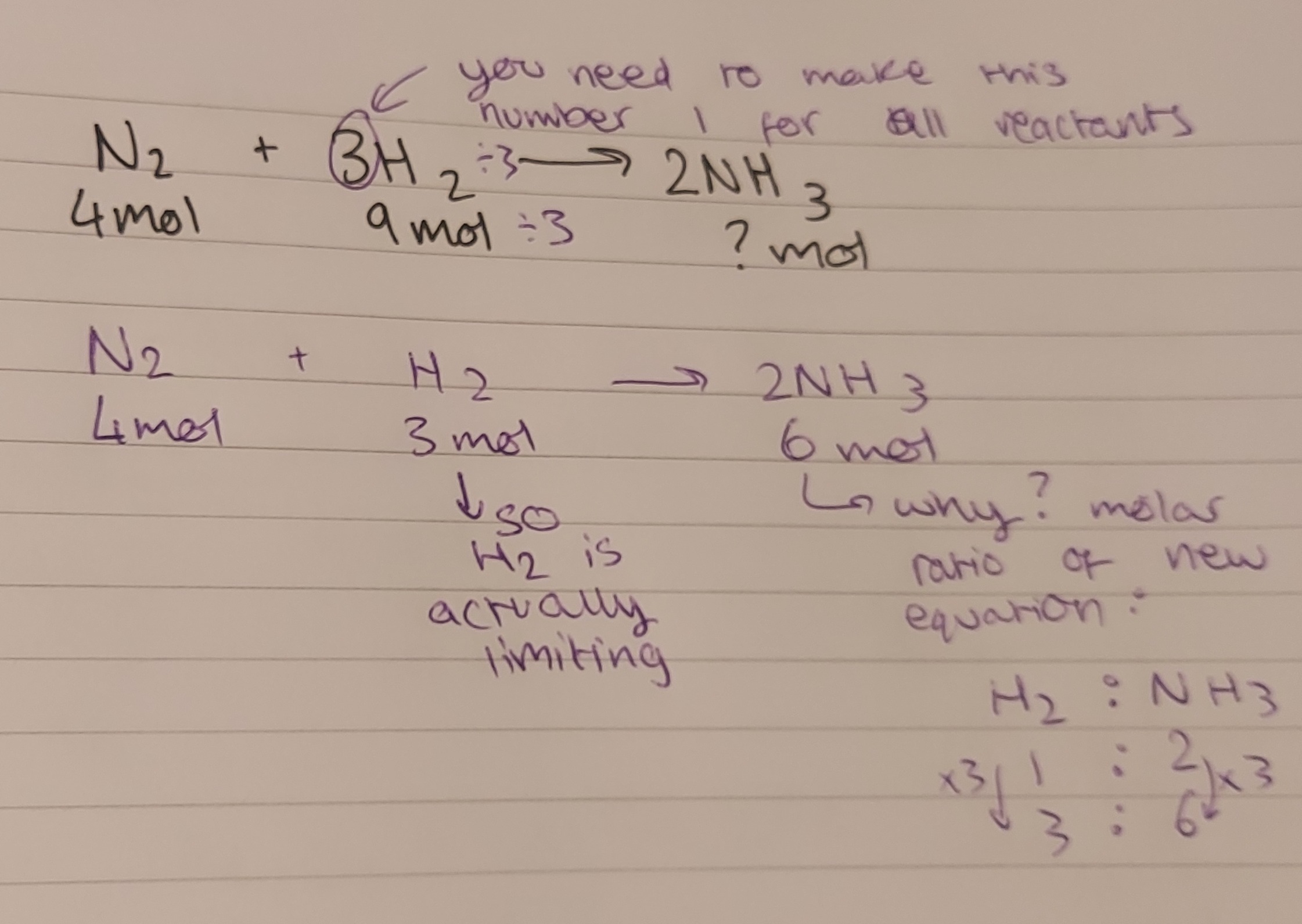
limiting reactants will come up in questions asking you to find the mass of product(s) given the masses of the reactants. e.g:
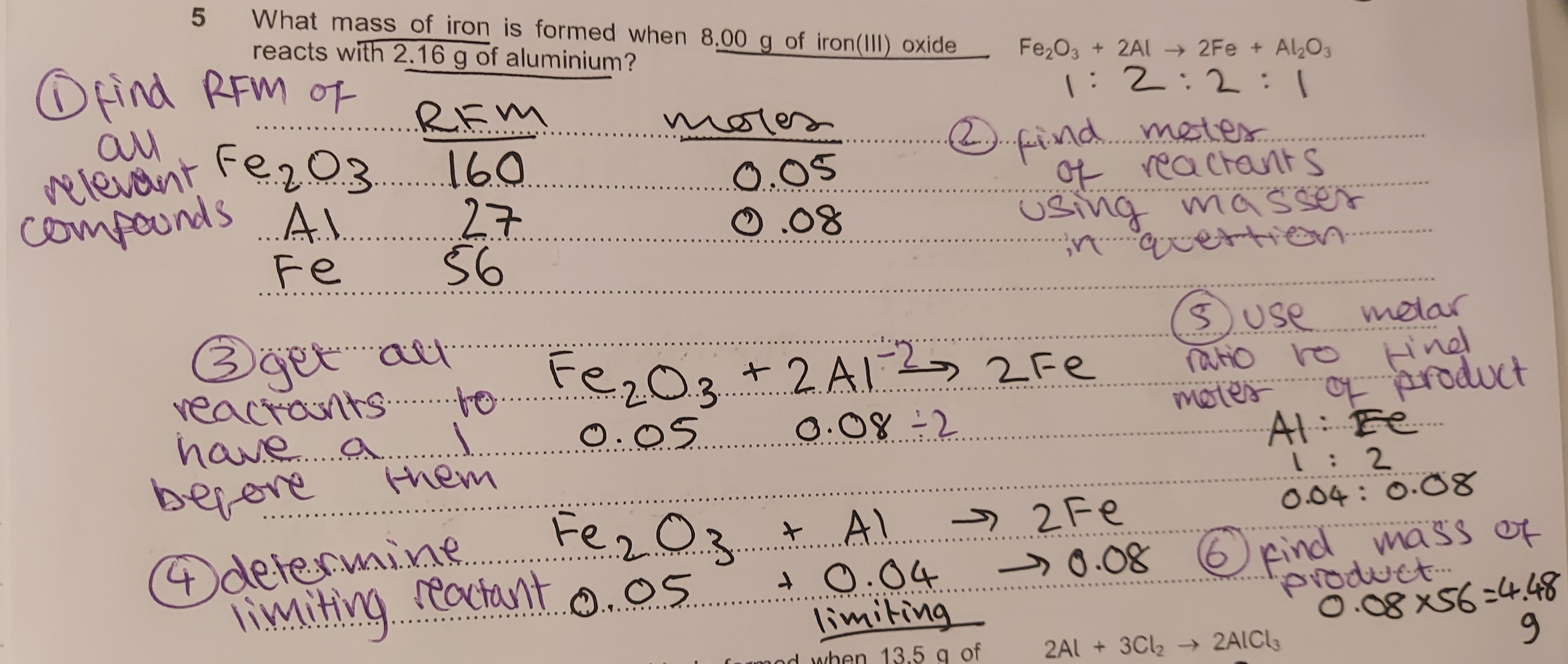
percentage yield
percentage yield = (actual yield (in g) / theoretical yield (in g)) × 100
yield is how much product is made in a reaction. you want this to be high to reduce amount of wasted reactants, reducing costs.
actual yield is how much product is actually made
theoretical yield is how much product should be made given the mass of the reactants
% yield is rarely 100%. in fact 70%+ is considered a good yield. why?
product may be lost transferring (filtering, pouring, etc)
reaction may be incomplete - some of reactants didn’t react to form product. may be because reaction is reversible
side reactions occurring and competing with desired reaction for its resources
a percentage yield question will often be preceded by a question asking you to find theoretical yield → see reacting masses.
Moles in a Solution
moles in a solution - concentration (in mol/dm3) × volume (in dm3)
1000cm3 = 1cm3 (basically if the question gives you a value in cm3 divide by 1000)
concentration in g/dm3 = concentration in mol/dm3 × Mr
these types of questions will often come up with titrations. it’s useful to use a table to organise your information:

Moles in a Gas
1 mole of any gas at room temperature and pressure occupies the same volume - 24 dm3 this is the molar volume.
volume of a gas at room temp + pressure (in dm3 = amount of gas (in mol) × 24 dm3
these questions will come in 3 slightly different forms:
given volume is in dm3 → substitute into equation to find moles
given volume is in cm3 → convert to dm3 then substitute in to find moles
given volume is in g → use n = mass / Mr to find moles
also useful to arrange in a table with volume and moles. remember to consider molar ratio of the compounds when deciding on moles.