BIOC C NOTE
1. General Metabolism Concepts
Metabolism - Concepts
- Metabolism is the SUM of chemical transformations
- Metabolic pathways involve enzymes
- Catabolism = degradation of biomolecules
- Anabolism = synthesis of biomolecules
Types of Pathways
- Converging catabolism
- Cyclic pathway
- Diverging anabolism
Concepts Review
- Metabolism is the sum of reactions
- Catabolism is the breakdown of molecules
- Anabolism is the synthesis (biosynthesis) of molecules
- Rate-limiting steps determine the overall speed of a pathway
- Rate- limiting steps represents points of regulation, and are exergonic
- Being strongly exergonic = reactions essentially irreversible
2. Rate limiting Enzymes
Rate - Limiting Steps
- Rates of biochemical pathways depend on the activities of enzymes that catalyze each step
- Reactions that catalyze one or more enzymes in any pathway will be limiting
- Rate-limiting steps are often exergonic and irreversible under cellular conditions
- ENZYMES catalyzing exergonic rate-limiting steps are targets of METABOLIC REGULATION
3. Glycolysis and regulation of:
1. Hexokinase (step 1)
- Glucose → G-6-Phosphate (using enzyme Hexokinase, and ATP → ADP)
### Hexokinase is allosterically inhibited by its product
- Hexokinase I is expressed in muscle (max activity)
- if G6P increases, enzyme is inhibited (-ve feedback)
- Hexokinase IV (glucokinase) is expressed in the liver
- high blood glucose relieves inhibition by glucokinase regulatory protein
- lower affinity for glucose
- inhibited by fructose 6-p (glucokinase regulatory protein)
- increase F6-P = binds to regulatory protein
- IV = HIGH BLOOD GLUCOSE = IN CYTOSOL
- I = LOW BLOOD GLUCOSE = SEQUESTERED INTO THE NUCLEUS
### Hexokinase IV has a relatively poor K0.5
- enzyme I do not increase rate when glucose is higher than optimal
- IV has higher K0.5 (10 mM) compared to 0.2 mM in the I enzyme
### Regulation
- High fructose-6-p = glucokinase regulatory protein sequesters hexokinase IV in the nucleus
- F6P binds to Regulatory protein causing a conformational change
- Regulatory protein binds to Hexokinase IV causing another conformational change (sprouts NLS)
- High glucose weakens the enzyme/regulator interaction (encourages cytosolic localization)
2. Phosphofuktokinase-1 (step 3)
- Fructose 6 phosphate + ATP → Fructose 1,6, bisphosphate + ADP (using citrate, and Fructose 2,6, bisphosphate)
- phosphorylation by PFK-1 commits fructose-6-phosphate to glycolysis (in equilibrium with G6P)
### PFK-1 Allostery
- ATP = negative heterotropic modulator (lowers affinity for fructose 6-P)
- ADP and AMP relieve inhibition by ATP
- Citrate increases inhibition by ATP
- Fructose 2,6, bisphosphate = strong activator
### Regulation of PFK-1 by ATP
- High ATP = reduce affinity of PFK-1 for f6p
- low ATP = high F6P affinity allows PFK-1 to be more active
3. Pyruvate Kinase (step 10)
- Phosphoenolpyruvate → (2) Pyruvate (2ADP → 2ATP)
### Pyruvate Kinase inhibition
- High ATP allosterically inhibits PK, decreasing its affinity for PEP
- Acetyl-CoA and long-chain fatty acids also inhibit PK
- when plentiful, so is ATP
- ALANINE (- allosteric modulator)
- F 1,6-BP accumulation (+ allosteric modulator)
4. Gluconeogenesis and regulation of:
1. Step 1
- Bypass for step 10 of glycolysis (pyruvate → phosphoenolpyruvate/PEP)
- Gluconeogenesis consumes NADH
- Liver produces NADH in the mitochondria during gluconeogenesis (degrading fatty acids)
- NADH = not transported to the cytosol
- mitochondrial malate dehydrogenase consumes NADH, cytosolic malate dehydrogenase produces NADH
- Lactate dehydrogenase produces cytosolic NADH (feedback for gluconeogenesis)
- Liver PEP carboxylase produces PEP directly, and extra NADH is used (not needed in cytosol)
2. Step 10
- G-6-Phosphatase catalyzes the dephosphorylation of G-6-P
- Expressed in a few tissues (liver, kidney, small intestine) = gluconeogenic tissues
3. Step 8
- Fructose 1,6 bisphosphatase converts F 1,6, Bis phosphate to F 6-P (phosphatase)
- AMP(-)
- F 2,6-BP (-)
STEP 3
- F 2,6 - BP (+)
- AMP (+)
- ADP (+)
- ATP (-)
Citrate (-)
F26PB = positive modulator for Glycolysis, negative modulator for gluconeogenesis
Positive regulator for PFK-1 and negative modulator for FBPase-1
PFK-1 low affinity for fructose-6-P in the absence of F2, 6BP
F2, 6BP has high affinity for fructose-6-P
FBP-1 = high affinity for f-1,6-BP in absence of F26PB
Affinity for F16PB decreases in the presence of F26PB
F 2,6 PB = controlled by two opposing enzyme activities: PRK-1 and Fructose 2,6 - bisphosphatase
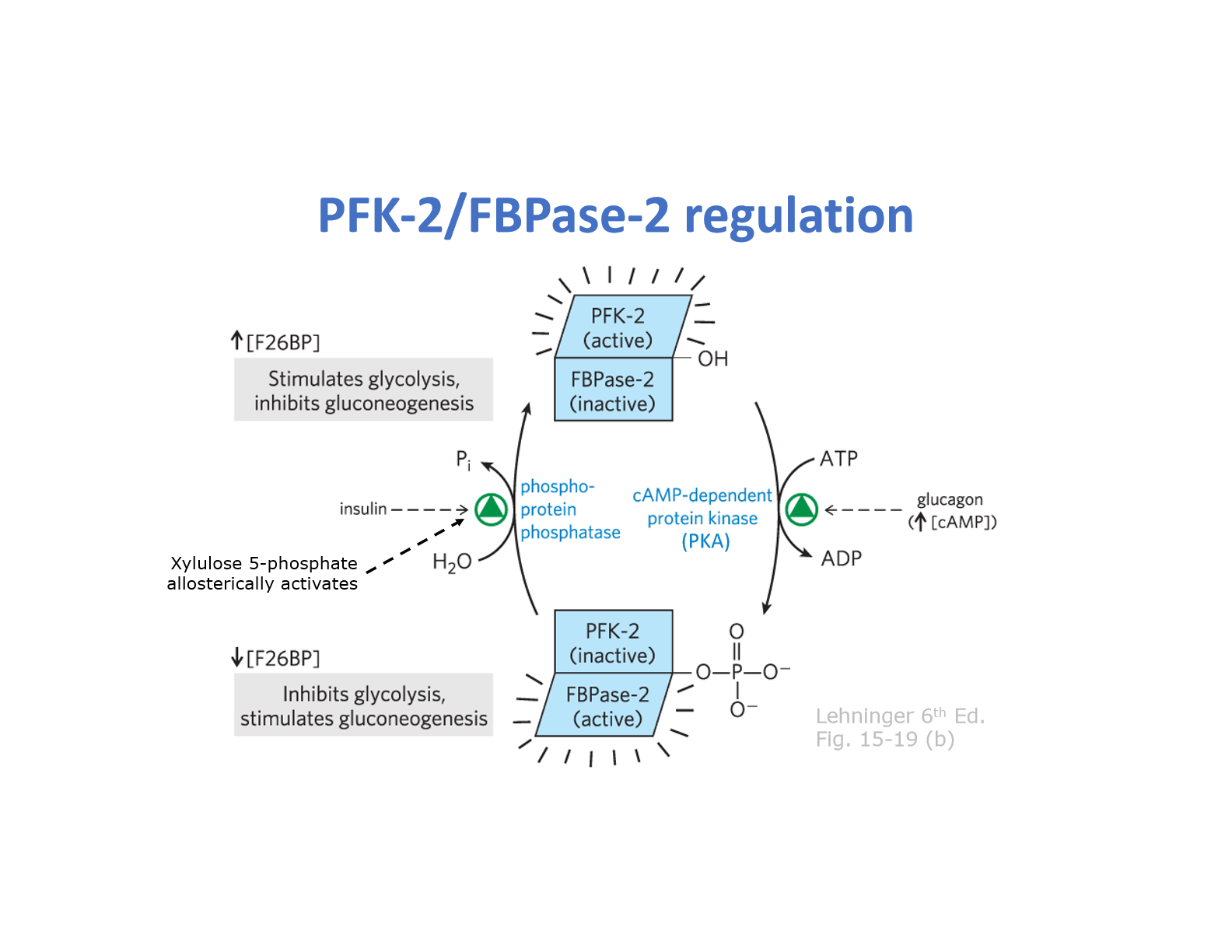
- PFK-2/FBPase-2 = BIFUNCTIONAL enzyme
- phosphorylation by PKA = activates FBPase 2 and INACTIVATES PFK-2
- dephosphorylation by phosphoprotein phosphatase (in response to insulin) activates PRK-2, and inactivates FBPase-2
- Xylulose 5-phosphate (pentose pathway) allosterically upregulates phosphoprotein phosphatase
- PFK-2/FBPase-2 isoenzyme response
- key enzymes are differentially regulated in these tissues
- Liver isozyme = phosphorylation on Ser 32 activates FBPase-2
- Cardiac muscle isozyme = phosphorylation on Ser 406 and Thr 475 activates PFK-2
Steps 1 and 10
- reciprocally regulated by acetyl-CoA
- PK allosterically activated by F1,6-BP = first molecules committed to glycolysis
- Pyruvate kinase = allosterically inhibited by:
- ATP
- Acetyl-CoA
- Long chain fatty acids
- Alanine
- Liver has different pyruvate kinase isoform
- phosphorylated by PKA in response to the hormone glucagon (which signals low blood sugar)
- Slows liver PK, reserving scarce sugar for organs that need it
- Pyruvate carboxylase (acetyl-CoA (+))
- PEP carboxykinase (ADP(-))
5. Pentose Phosphate Pathway
1. Oxidative
- Oxidation of glucose 6-P
- produces NADPH from reductive biosynthesis
- produces ribose 5-phosphate for the synthesis of nucleotides
2. Non-Oxidative
- Glycolic intermediates
- glucose 6-phosphate
- Glucose → Glucose 6-phosphate dehydrogenase
- stimulated by NADP+ (homotropic )
- inhibited by NADPH (heterotropic)
- Regulated by redox state of cytosol
- xylulose 5-phosphate (modulator of phosphatase that stimulates liver PFK-2)
- Transaldolase/Transketolase transfer 2 or 3 carbon atoms between sugar phosphates = rearrange 5C molecules into 6C moleules
- 3C sugars are glyceraldehyde=3P → turned into Glucose-6P by gluconeogenesis
- 6 X 5C → 5 X 6C
6. Glycogen Metabolism
- Glycogen: polymer of glucose (storage)
- Quick source of energy
- Liver: glycolysis + control of blood glucosse
- Muscle: glycolysis
1. Synthesis
- Making the precursor
- glucose 6-P → glucose 1-P (phosphoglucomutase)
- glucose 1-P + UTP → UDP-glucose + PPi (UDP-glucose pyrophophorylase)
- UDP-glucose then acts as an activated sugar donor
2. Glycogenolysis
- Glycogen Phosphorylase
- Glycogen -→ glucose + Glucose 1-phosphate (glycogen phosphorylase)
- Glucose binding of phosporylase = allosteric = dephosphorylation by phosphorylase a phosphatase
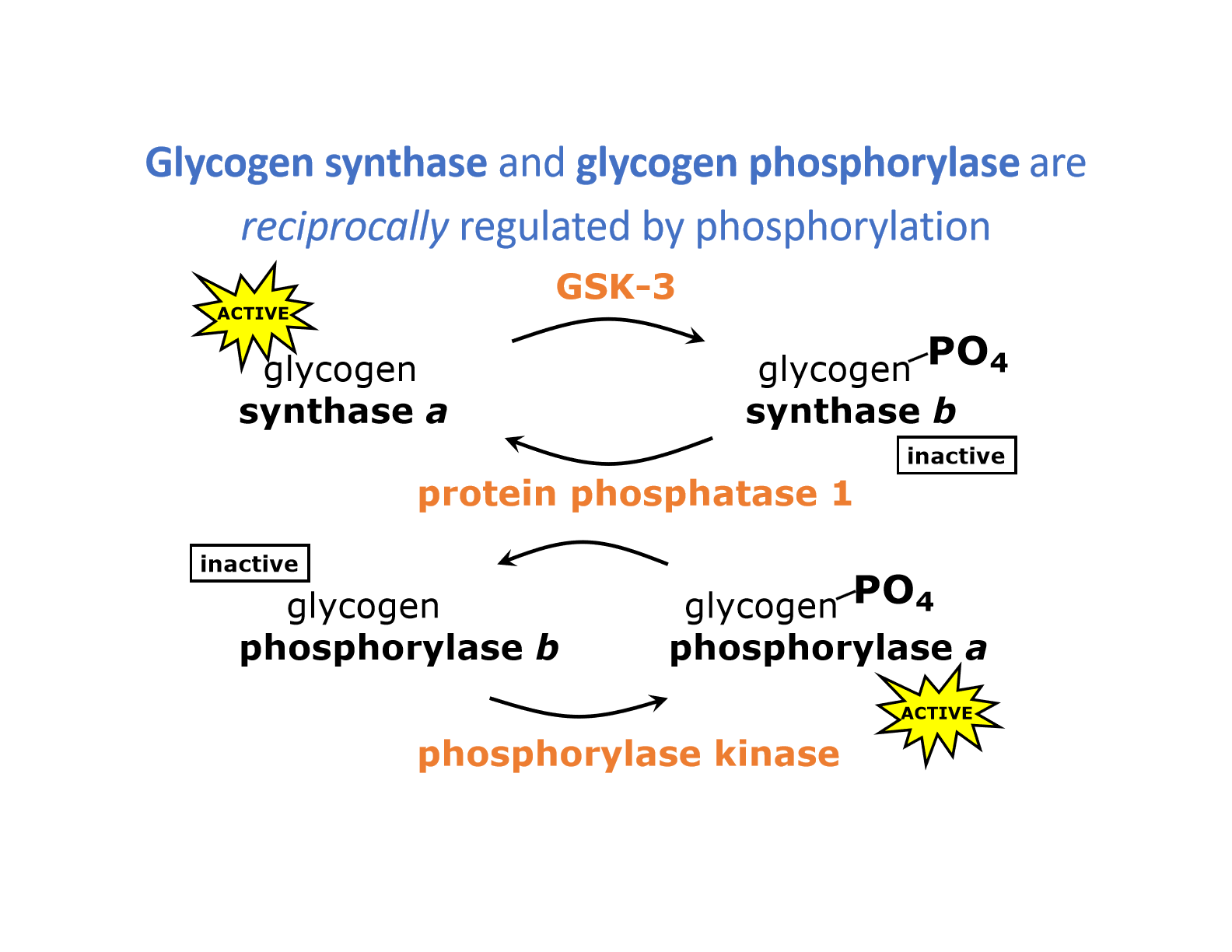
3. Regulation of glycogen synthase and phosphorylase
- Synthase = inhibited by phosphorylation (PKA and GSK3)
- Phosphorylase = activated by phsphorylation
- Phosphorylase B kinase = phosphorylates glycogen phosphorylase (activates it)
- Glycogen phosphorylase a phosphatase (dephosphorylates)
- REGULATED BY = PKA-mediated phosphorylation
7. Regulation by PKA and hormones
PKA regulation = low blood sugar leads to increased glycogen breakdown and decreased glycogen synthesis and glycolysis
Hormone-regulated enzyme activity coordinates tissue specific metabolism
- Why are muscles and liver metabolism different?
- Muscle lacks glucagon receptors
- muscle pyruvate kinase = not phosphorylated by PKA
- muscle lacks gluconeogenic enzymes
- muscle lacks a key enzyme for glucose export
- uses stored glycogen and glucose for itself