Urea Cycle
The urea cycle, also known as ornithine cycle, is a series of biochemical reactions that convert ammonia to urea, which is then excreted from the body through urine.
The urea cycle mainly occurs in the liver because arginase is present in high concentrations there, facilitating the final step of urea production. This pathway is partially mitochondrial and cytosolic because the first 2 reactions of the urea cycle takes place in the mitochondria, whereas the next three reactions takes place in the cytosol.
This cycle is crucial for:
Formation of Ammonia: Ammonia is generated from the breakdown of amino acids and nucleotides and must be processed to prevent toxicity.
Disposal of Ammonia: The urea cycle efficiently disposes of excess ammonia, which is harmful in high concentrations, by converting it into urea.
Transport and Storage of Ammonia: Ammonia is transported as part of blood plasma; regulating its levels is essential for maintaining homeostasis.
Function of Ammonia: While ammonia has various roles in metabolic processes, its levels must be carefully controlled to avoid neurotoxicity.
Key Enzymes Involved:
Six important enzymes participate in the urea cycle, each catalyzing a specific reaction that transforms ammonia into urea:
Carbamoyl Phosphate Synthetase 1 (CPS1): This enzyme catalyzes the first step and plays a vital role in the synthesis of carbamoyl phosphate from ammonia and bicarbonate. This enzyme is mitochondrial.
Carbamoyl Phosphate Synthetase 2 (CPS2): An enzyme located in the cytosol, it facilitates the conversion of aspartate and carbamoyl phosphate into pyrimidine nucleotides, showcasing a distinct physiological role compared to CPS1.
Ornithine Transcarbamylase (OTC): Converts carbamoyl phosphate into citrulline, which is a crucial intermediate.
Argininosuccinate Synthetase (ASS): Catalyzes the formation of argininosuccinate from citrulline and aspartate; this step introduces a second nitrogen into the cycle.
Argininosuccinate Lyase (ASL): Converts argininosuccinate into arginine and fumarate, releasing nitrogen in the process.
Arginase: This enzyme catalyzes the final step, converting arginine into urea and ornithine, allowing the cycle to restart.
N-acetylglutamate synthase: A regulatory enzyme of the urea cycle because it catalyzes the formation of N-acetylglutamate from acetyl-CoA and glutamate, which activates carbamoyl phosphate synthetase I, the first enzyme of the cycle. This regulatory mechanism ensures proper functioning of the urea cycle, especially during conditions of elevated ammonia levels, thereby promoting nitrogen disposal and maintaining metabolic homeostasis.
Urea Cycle
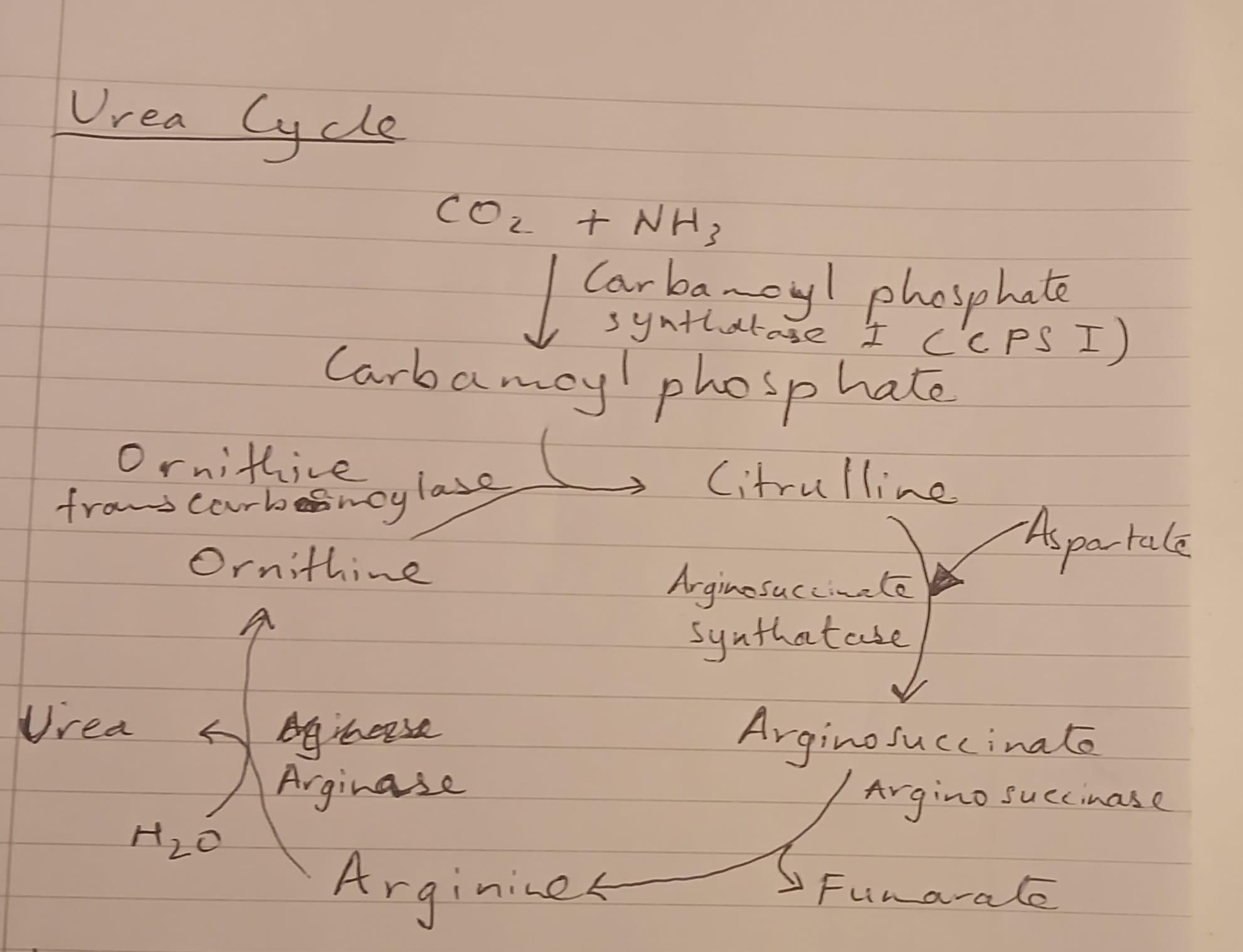
Urea cycle begins when CO2 and ammonia combine to form carbamoyl phosphate, a reaction catalyzed by carbamoyl phosphate synthetase I (CPS I). This reaction does not require ATP.
Carbamoyl phosphate reacts with ornithine to form citrulline, catalyzed by ornithine transcarbamylase, which is a crucial step in moving forward in the cycle. This step occurs in the mitochondria, where both CPS I and ornithine transcarbamylase are located, thus facilitating the conversion of ammonia into a less toxic form before it enters systemic circulation.
Subsequently, citrulline is converted into argininosuccinate through the action of argininosuccinate synthetase, requiring aspartate as a key substrate, and this step marks the incorporation of nitrogen into the cycle. 2 ATP molecules are used up in this step because the conversion of citrulline to argininosuccinate involves the phosphorylation of citrulline.
Following this conversion, argininosuccinate is then cleaved by argininosuccinate lyase to produce arginine and fumarate, completing a significant part of the cycle. Fumarate enters the Krebs cycle, where fumarate is converted into malate, facilitated by fumarase, and then malate is converted into oxaloacetate by malate dehydrogenase, supporting the energy production in aerobic respiration and enabling the regeneration of aspartate, which can re-enter the urea cycle, thus ensuring continuous nitrogen removal from the body. In this way, the urea cycle efficiently sustains the detoxification of ammonia, maintaining nitrogen balance within the organism.
ATP in this step is a crucial energy donor, highlighting the energetic cost associated with synthesizing argininosuccinate, reflecting the metabolic investment required for effective nitrogen excretion.
Ornithine is produced from arginine through the action of arginase, which catalyzes the hydrolysis of arginine, releasing urea as a byproduct which is excreted by the kidneys, thus completing the cycle of nitrogen removal and allowing ornithine to re-enter the mitochondria, where it can participate in the urea cycle once more.
The cycle is interconnected with the Krebs cycle, using CO2 and is regulated by cellular energy levels, requiring an investment of ATP for effective nitrogen excretion
Associated Disorders
Deficiencies in these five enzymes lead to various metabolic disorders. Important conditions include:
Hyperammonemia Type 1: Occurs due to deficiency of carbamoyl phosphate synthetase 1, resulting in elevated levels of ammonia in the blood, which can cause severe neurological damage if left untreated.
Hyperammonemia Type 2: Resulting from deficiency of ornithine transcarbamylase.
Citrullinemia: Caused by a deficiency of argininosuccinate synthetase.
Argininosuccinic Aciduria: Due to a deficiency of argininosuccinate lyase which leads to the accumulation of argininosuccinic acid in the urine and can result in neurological deficits and developmental delays if not properly managed.
Hyperargininemia: Related to defects in arginase. Increased levels of ammonia leads to the diversion of glutamate, affecting neurotransmitter synthesis and resulting in neurotoxicity.
Regulation of the Urea Cycle
Allosteric Regulation: The urea cycle is allosterically regulated by energy levels within the cell, reflecting the status of cellular metabolism.
Compartmentalization: The urea cycle's steps are compartmentalized; two steps occur in the mitochondria while three steps occur in the cytosol, maximizing the efficiency of substrate utilization.
ATP Utilization: The process involves the utilization of 2 ATP molecules for ATP-AMP conversion; a net consumption of 4 ATP is noted overall in the urea cycle, highlighting its energy demands.