Esters
If the acid and alcohol have functional groups on both ends of the molecule, they can make many ester groups, resulting in a polyester (condensation polymer)
Reactions of esters
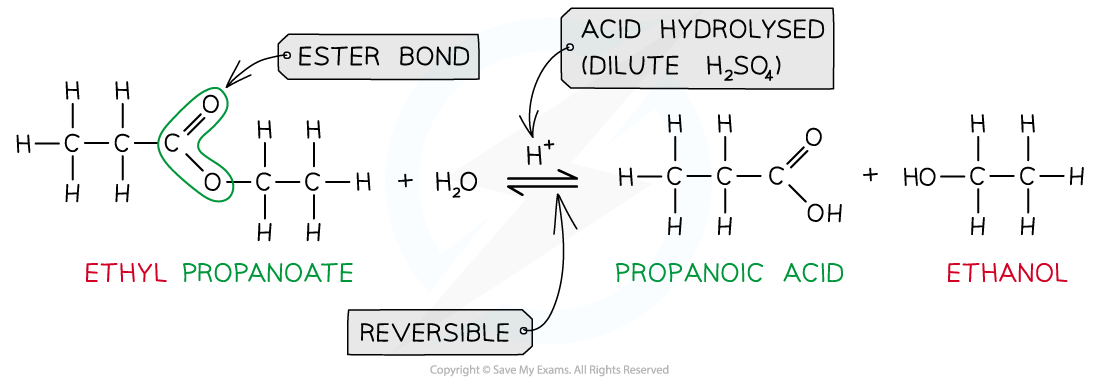
Acid hydrolysis
The reverse of the esterification
Makes carboxylic acid and alcohol
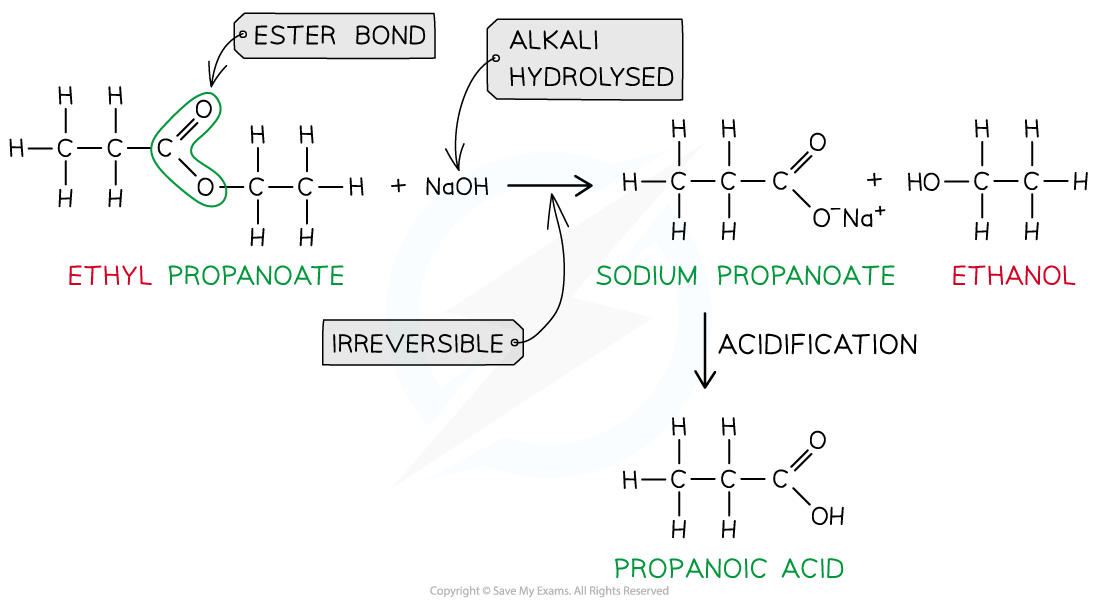
Alkaline hydrolysis
React with hot aqueous NaOH
Makes carboxylate salt and alcohol
Uses of esters
Small chained esters are used in perfumes and food flavourings
Smell nice and are volatile (vaporise easily)
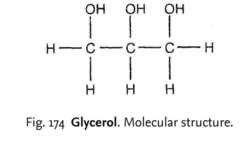
Glycerol, propane 1, 2, 3 triol
Can react glycerol with fatty acids (carboxylic acids) to form triglycerides (esters)
Animal and vegetable oils and fats are triglycerides
Hydrogen bonds as well as Van der Waals forces
Relatively high melting/boiling points
Can form hydrogen bonds to water so very soluble in water
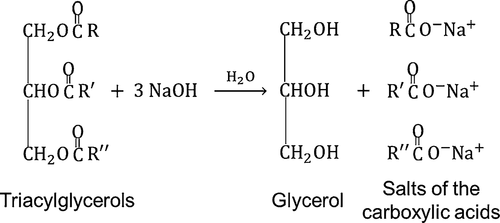
Triglycerides (esters)
Alkaline hydrolysis
Boil was NaOH
Forms glycerol + carboxylate salts (soaps)
Carboxylate salts have a polar (mixes with water) and non polar end (mixes with oils/grease). They allow grease and water to mix so are used for cleaning products
Methanol + alkali catalyst
Glycerol + biodiesel
Mixture of methyl esters
Can be used as a fuel