Midterm Revision
Module 1: high school revision
Adding or subtracting and multiplying or dividing measurements
Significant figures convey precision of measurements; the rules govern rounding during operations:
Addition/Subtraction: use the fewest decimal places among operands.
Multiplication/Division: use the fewest significant figures among operands.
Logarithms: the number of significant figures in the mantissa becomes the number of decimal places in the result.
1km = 1000 m
1s = 1 cs

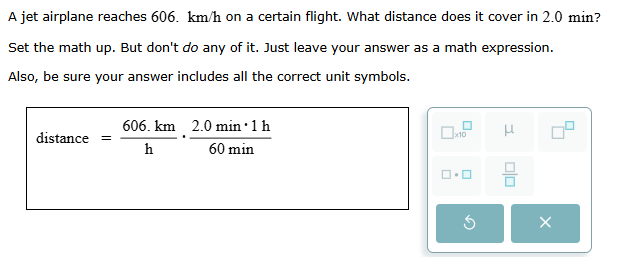
Setting up the math for one-step problem with unit conversion
"P = mv"
a fundamental physics equation representing linear momentum (p) as the product of an object's mass (m) and its velocity (v)
signifies that momentum is a measure of an object's "quantity of motion" and is a vector quantity, meaning it has both magnitude and direction

Distinguishing mixtures from pure substances through physical properties
Distinguishing chemical and physical change
If there is a reaction or a chemical released, it is a chemical change.
If there is a change in temperature or any physical properties mentioned, it is a physical change.
Understanding periods and groups of the Periodic Table
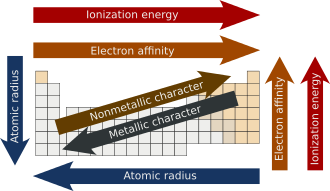
Positive ions (cations) are smaller than the neutral atoms from which they form because they lose electrons, reducing electron-electron repulsion and increasing the effective nuclear charge on the remaining electrons, which pulls them closer to the nucleus.

Counting protons and electrons in atoms and atomic lens
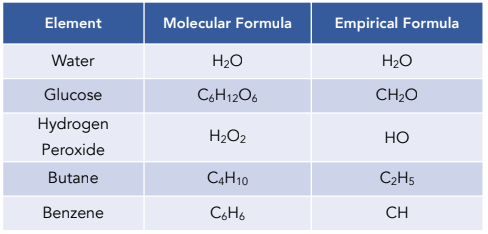
Understanding the difference between a molecular and empirical formula
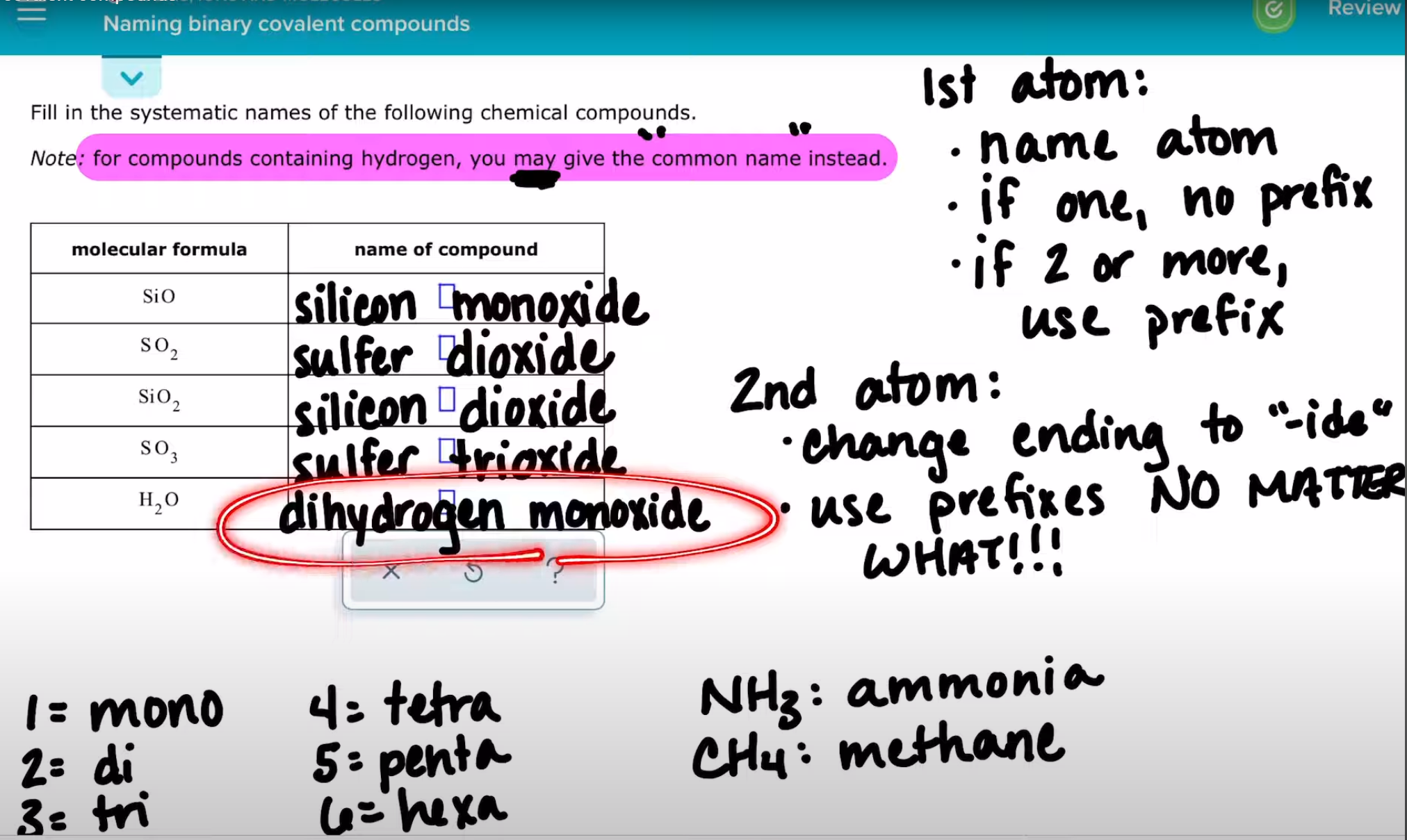
Naming binary covalent compounds
Predicting whether a compound is ionic or molecular
GGEZ
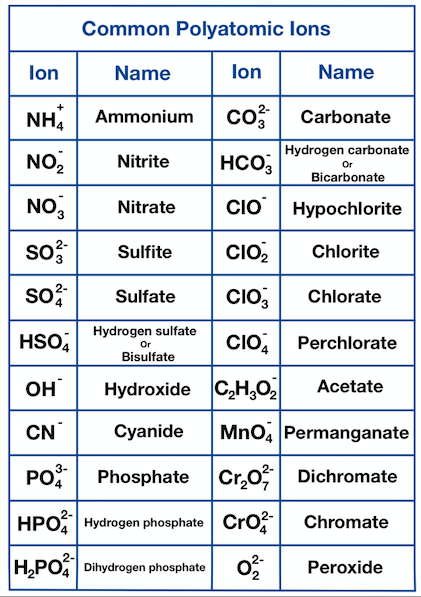
naming ionic compounds with common polyatomic ions
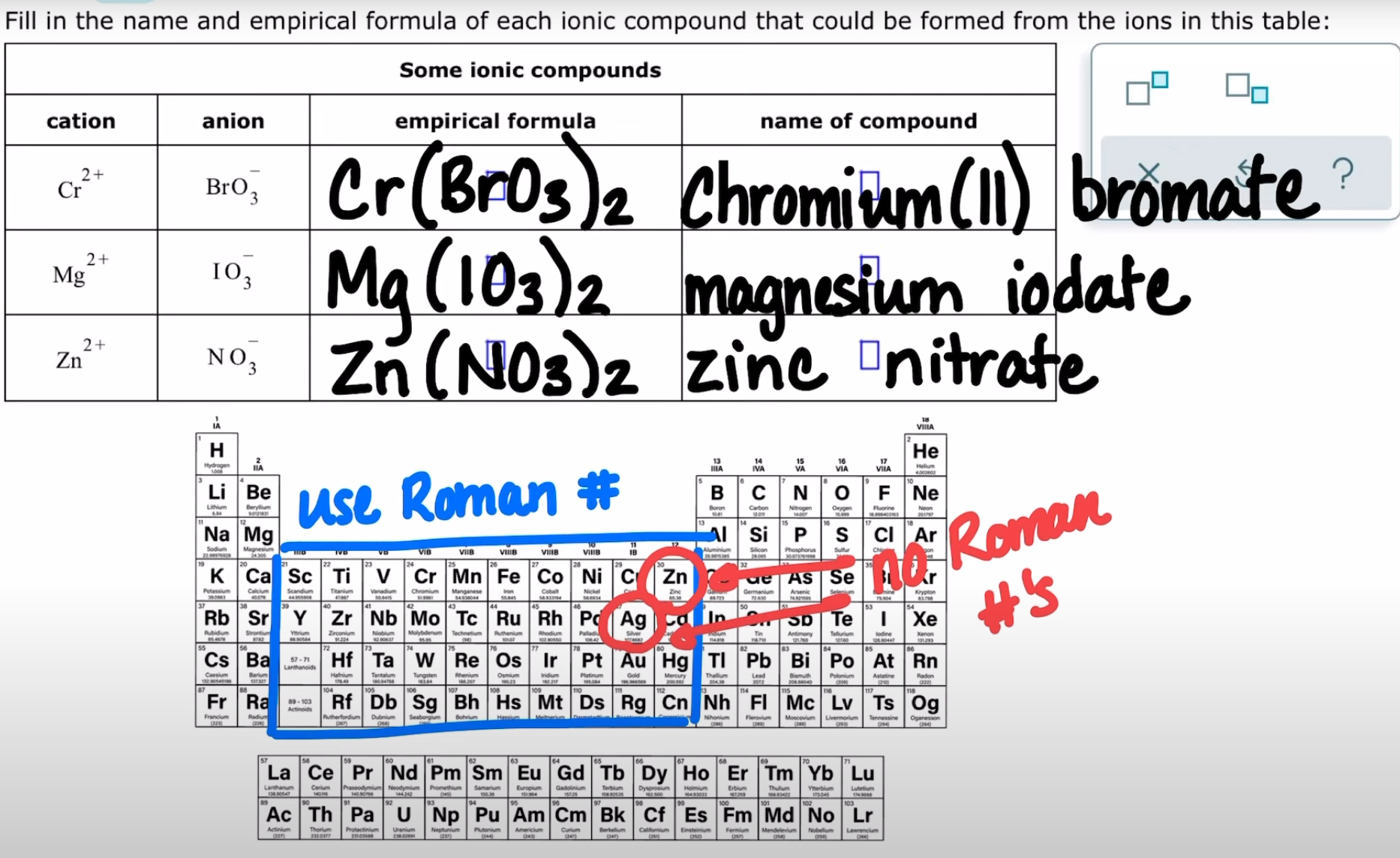
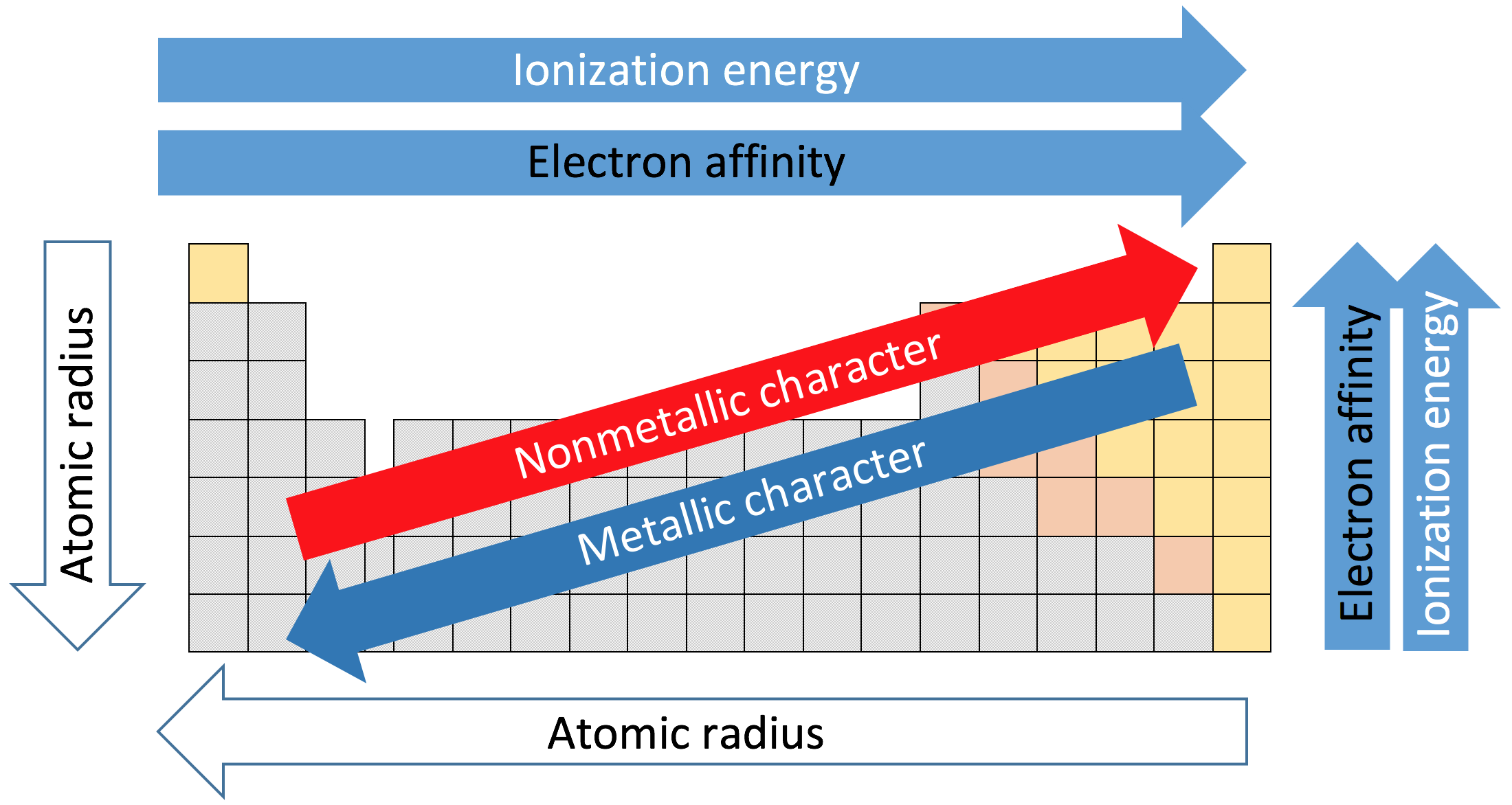
Understanding main-group periodic trends in metallicity
Module 2-3 : Quantum Atom and Bonding Models
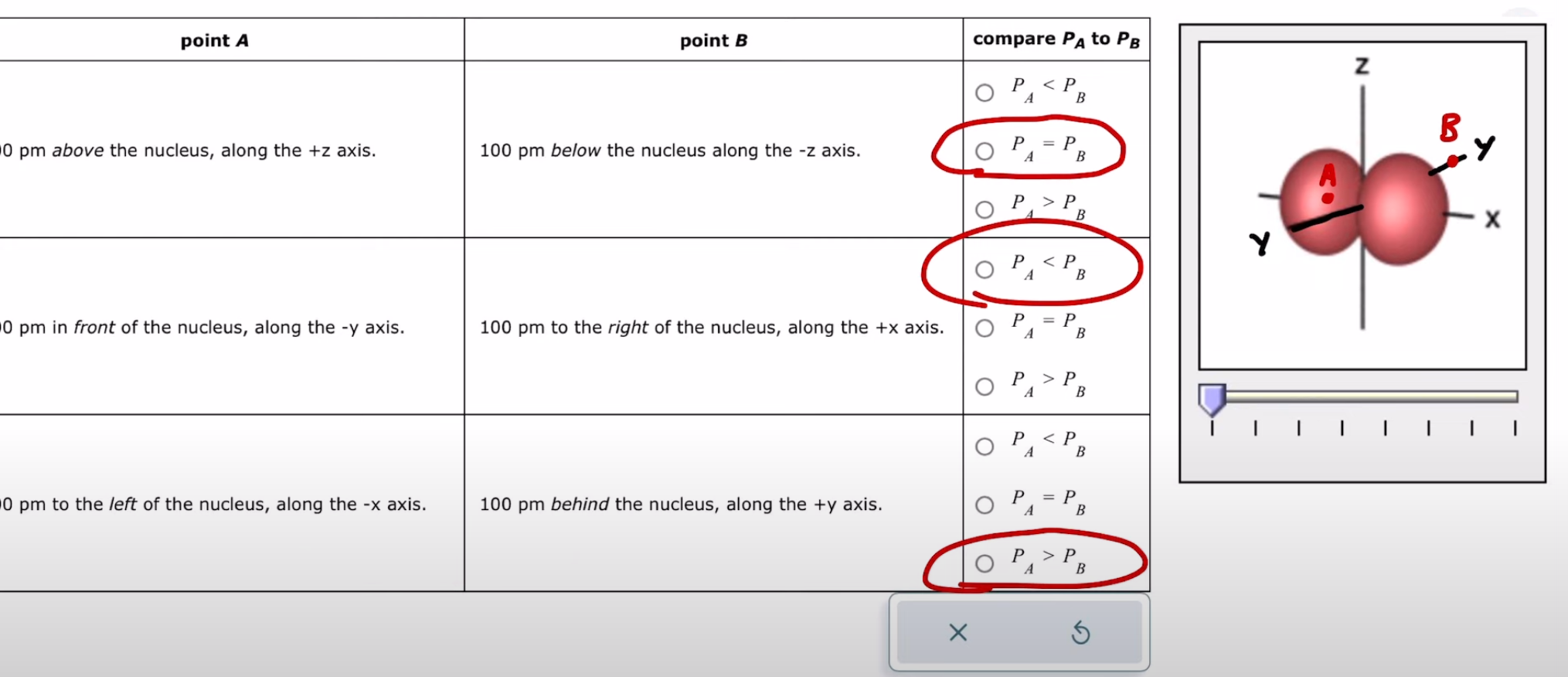
Interpreting radial and angular probability of distribution of an orbital
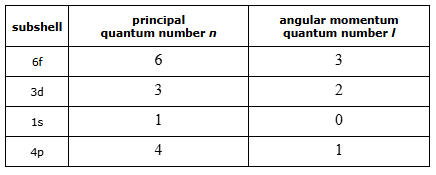
Deducing the allowed quantum numbers of an atomic electron
principle quantum number n has to be > 0 ad it is infinite.
angular momentum quantum number l range from 0 → n-1.
The highest possible value of l is less than n.
magnetic quantum number ml can be from -l to +l including zero.
magnetic quantum number ml determines the orientation of the orbital in space.
highest possible value of ml is 2l + 1.
spin quantum number ms can be +1/2 or -1/2. highest possible value is 2.
ml can only be 0 when l is 0 or more.
l can only be 0 when n is 1 or more.
interpreting the electron configuration of an atom or atomic ion in noble-gas notation
Use noble gas to shortcut writing all the electron configuration.
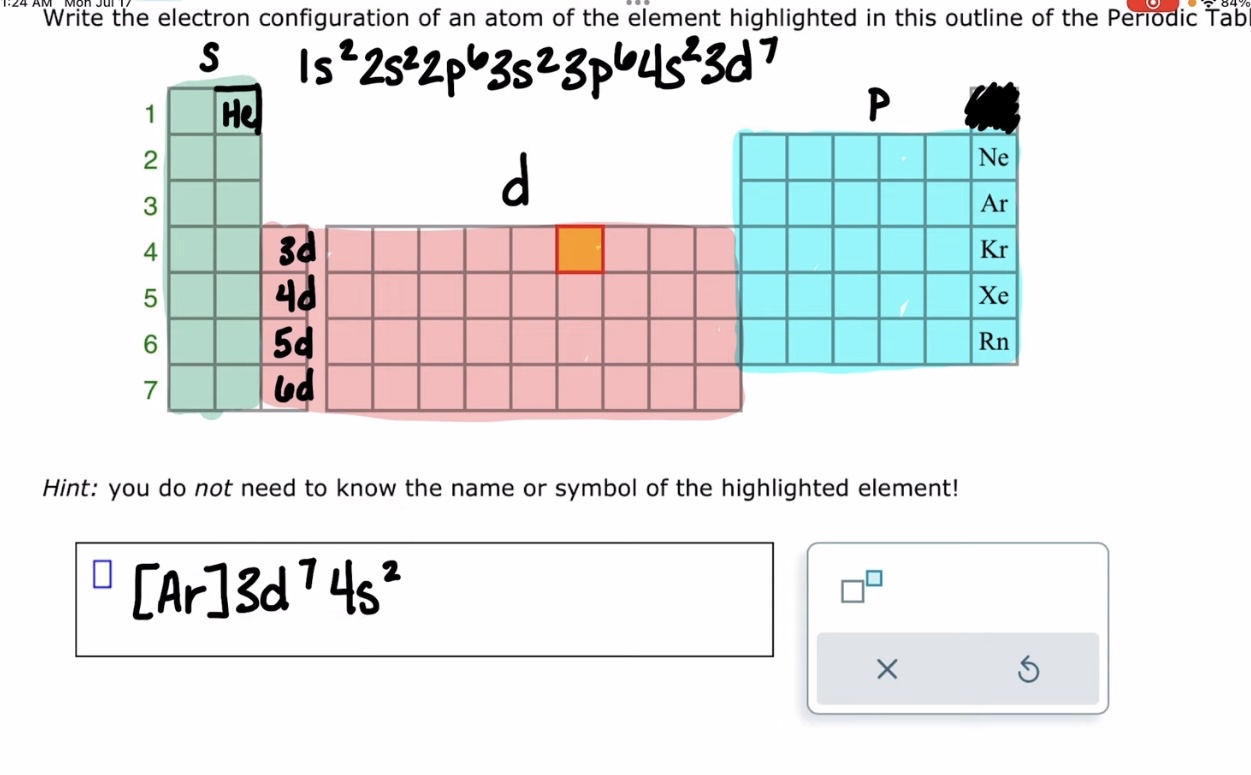
Madelung 's rule can be applied to predict the order in which these orbitals are filled, helping to determine the electron configuration of transition metals more efficiently.
The first nine subshells in order of increasing energy = 1s 2s 2p 3s 3p 4s 3d 4p 5s
Electrons in the same subshell have the same energy.
Subshells with higher n+l have higher energy.
If 2 subshells have the same n+l , the electrons with the higher n will have the higher energy.
Exceptions to the list of increasing energy are all transition metals
Cr[Ar] 4s13d5
Cu[Ar] 4s13d10

Nb[Kr] 5s14d4
Mo[Kr] 5s14d5
Ru [Kr] 5s14d7
Rh[Kr] 5s14d8
Pd[Kr] 4d10
Ag[Kr] 5s14d10
La [Xe] 6s25d1
Pt [Xe] 6s14f145d9
Au [Xe] 6s14f145d10
Ac [Rn] 7s26d1
Calculating the wavelength of a line in the spectrum of hydrogen
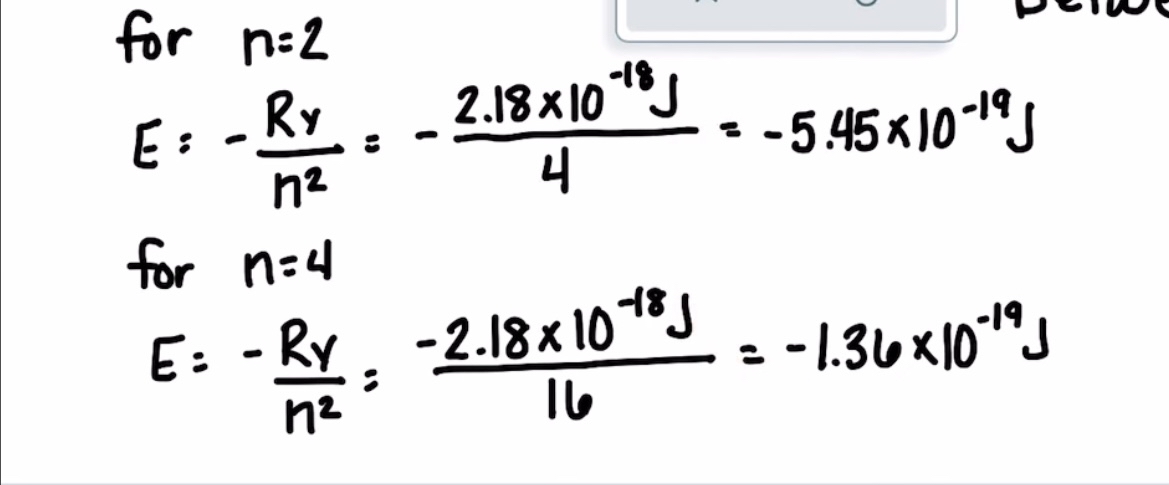

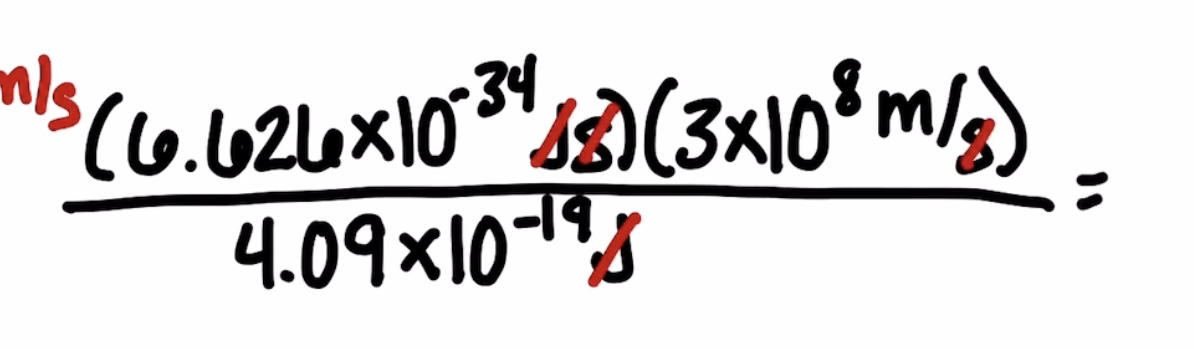
Writing the electron configuration of a first transition series atom

Module 4-5 : Molecular vs Crystal Structure
Counting electron pairs in a Lewis structure with double or triple bonds
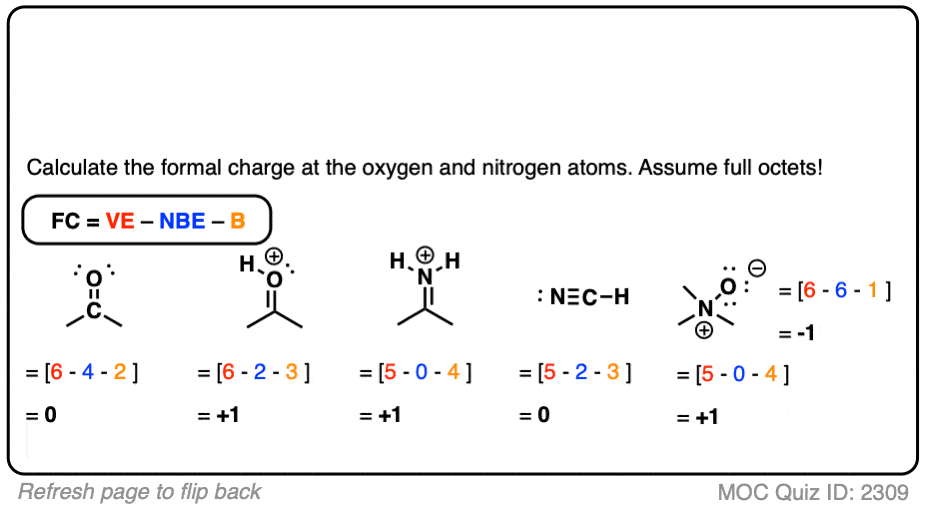
Calculating formal charge
Formal Charge = Normal Valence electrons - Non-bonding electrons - Number of bonds
Writing Lewis structures for an expanded valence shell electron atom

An element from Period 3 and below will be able to expand octet by making use of its energetically accessible, or low lying d-subshell for bonding.
This means only Period 2 elements such as C, N, O and F cannot expand octet and have to obey octet rule.

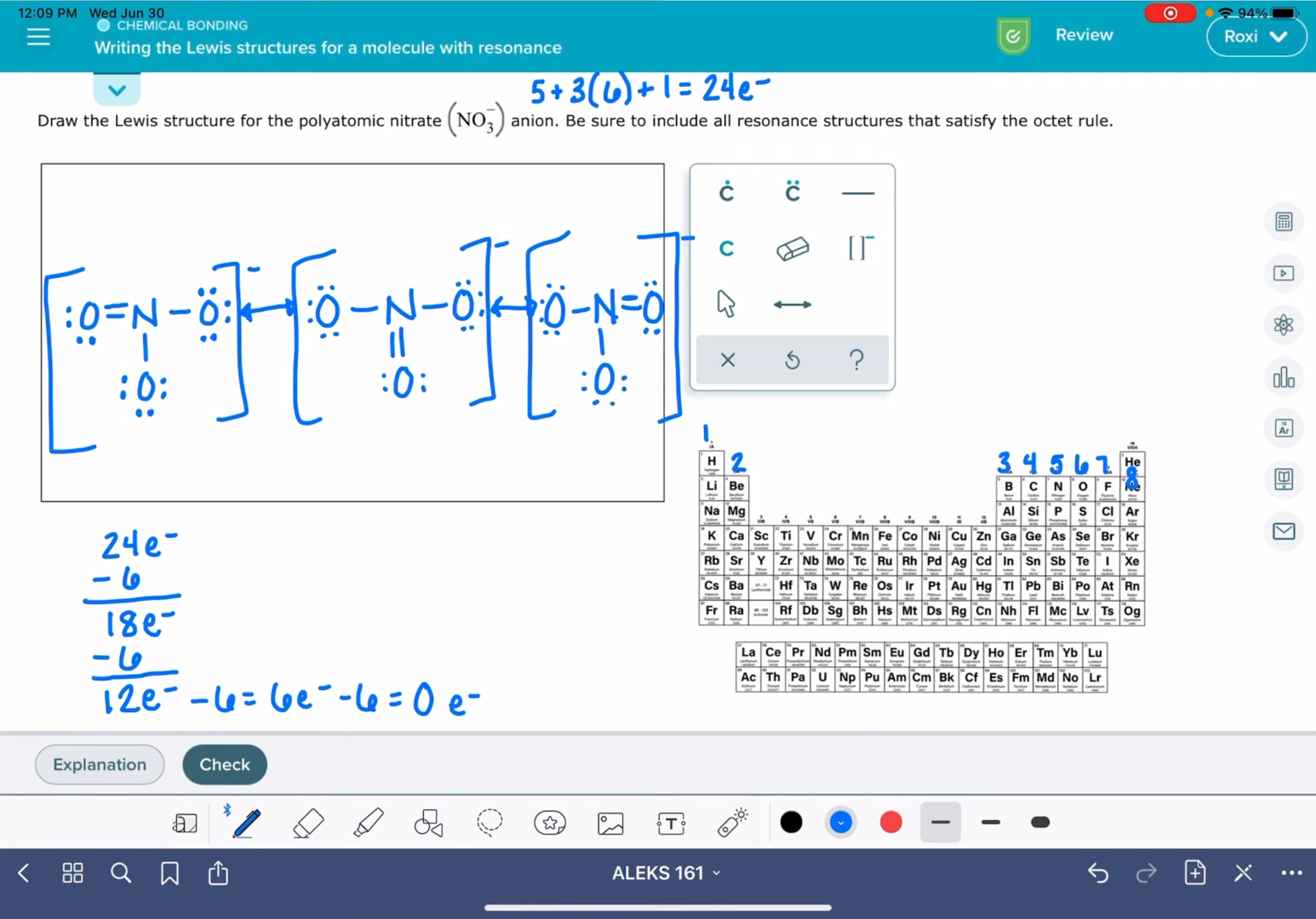
Writing Lewis structures for a molecule with resonance
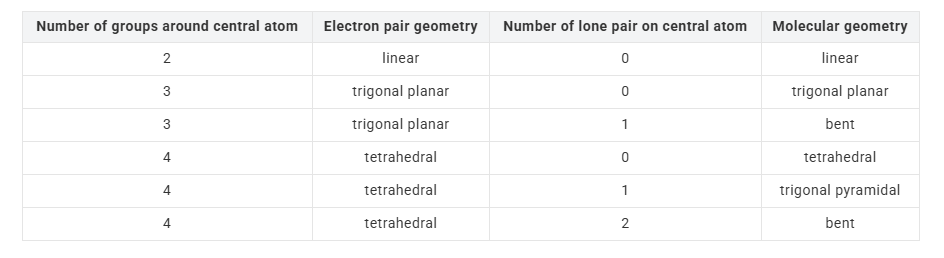
predicting the arrangement of electron groups around the central atom
of a molecule

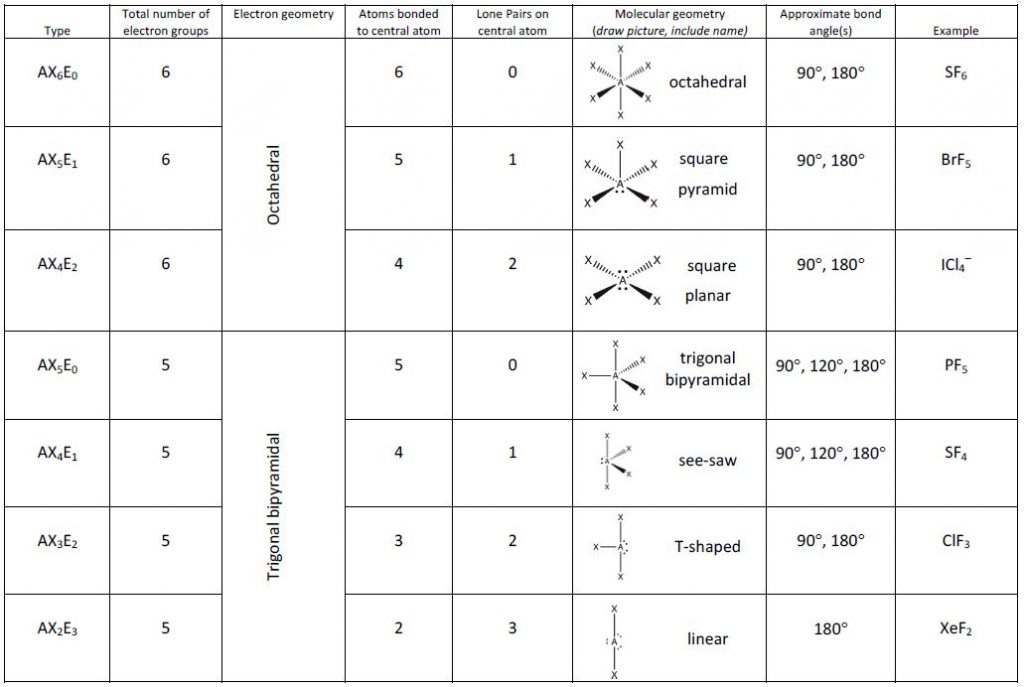
predicting and naming the shape of molecules with a central atom
Recognising and naming close-packed crystal lattices
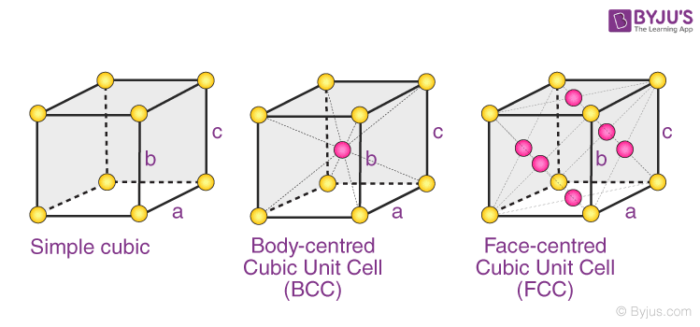
Finding an atomic radius from an fcc or bcc lattice constant
simple cubic lattice is a=2r
BCC is a=4r/root 3
FCC is a=4r/root 2
Module 6-7 : Quantum Bonding, Interactions & States of Matter
Predicting whether molecules are polar or non-polar
polar
En diff more than 0.5
asymmetrical
lone pairs → bent
non-polar
diatomic
symmetrical equal pulling
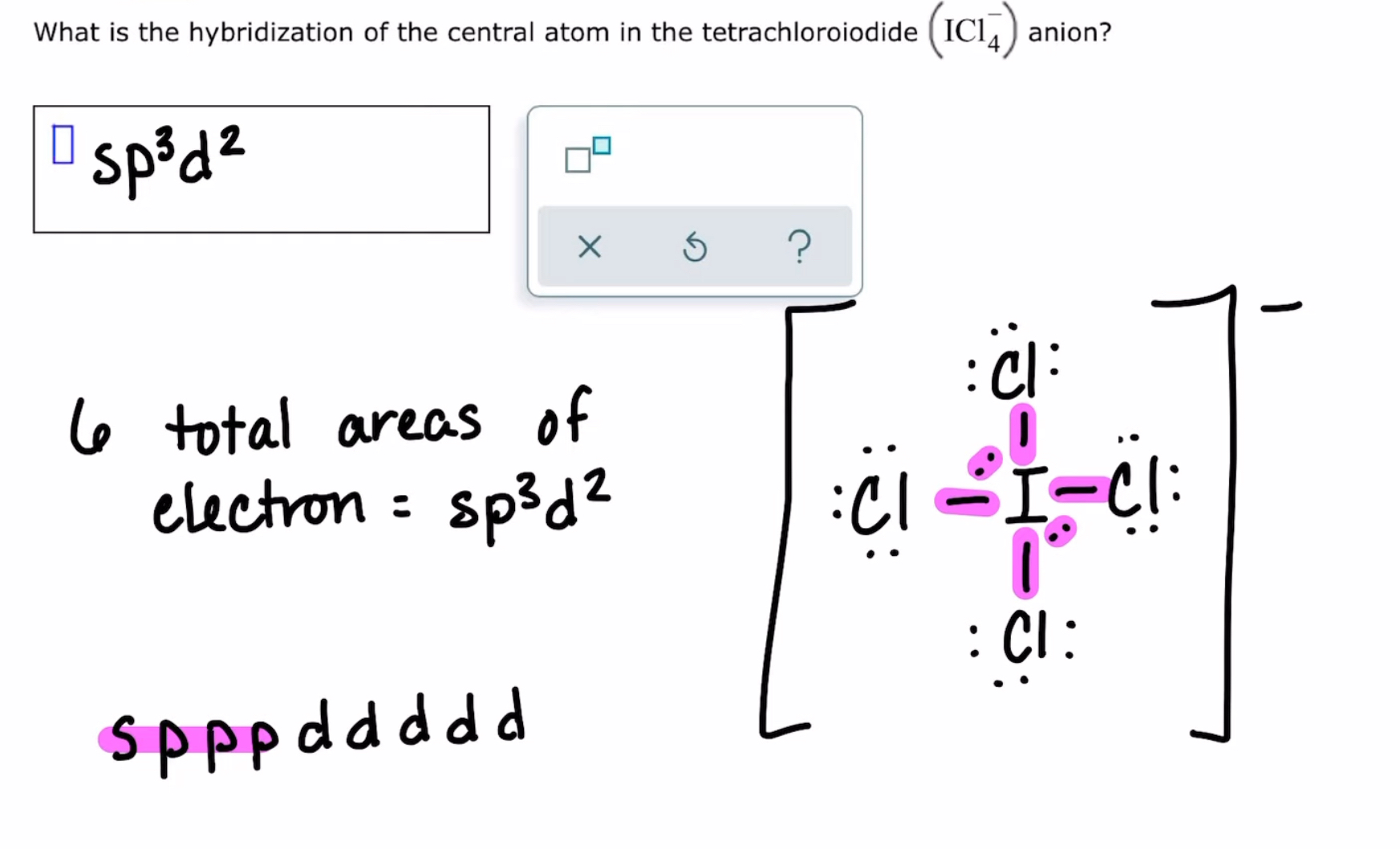
Identifying hybridization in a small molecule
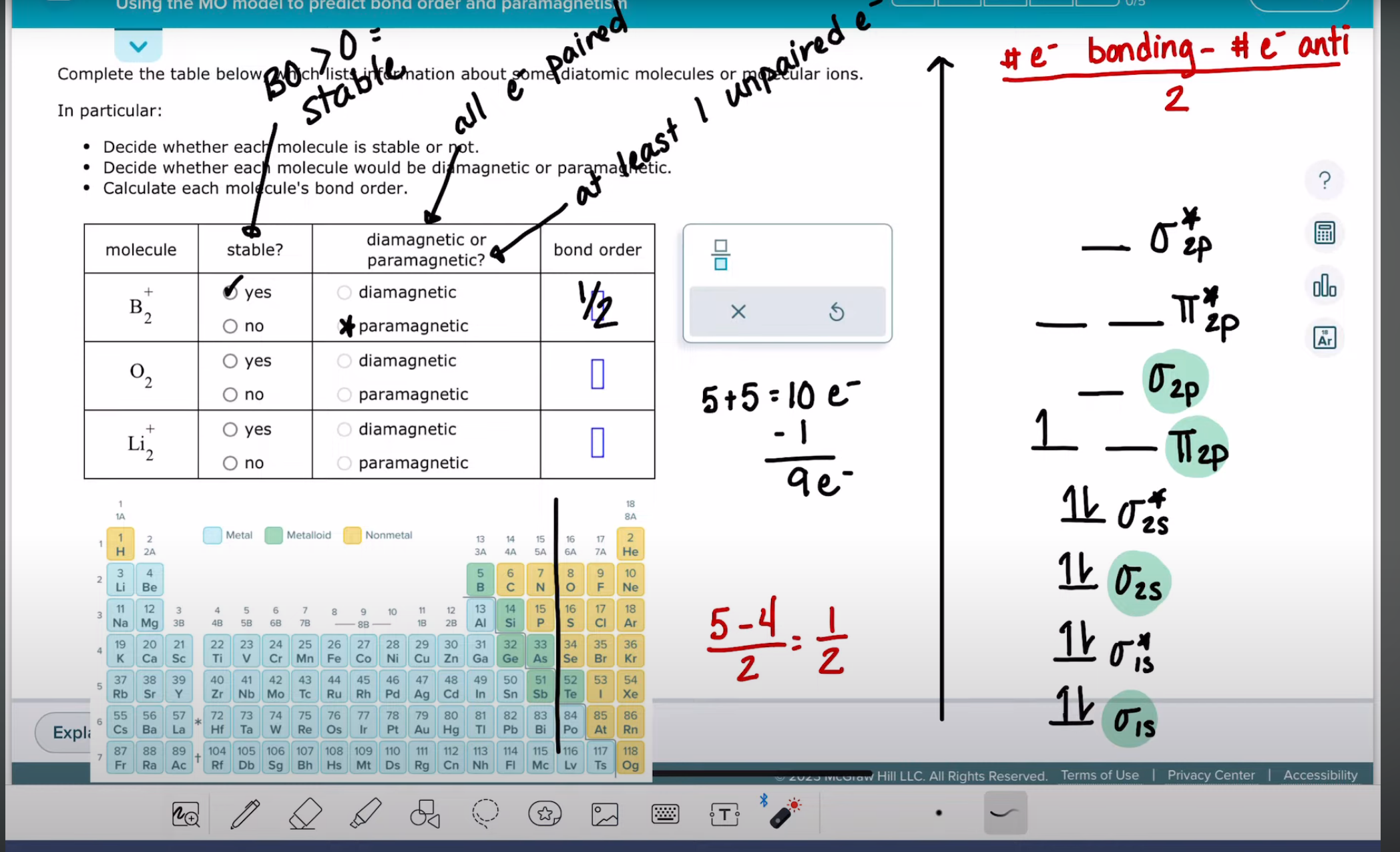
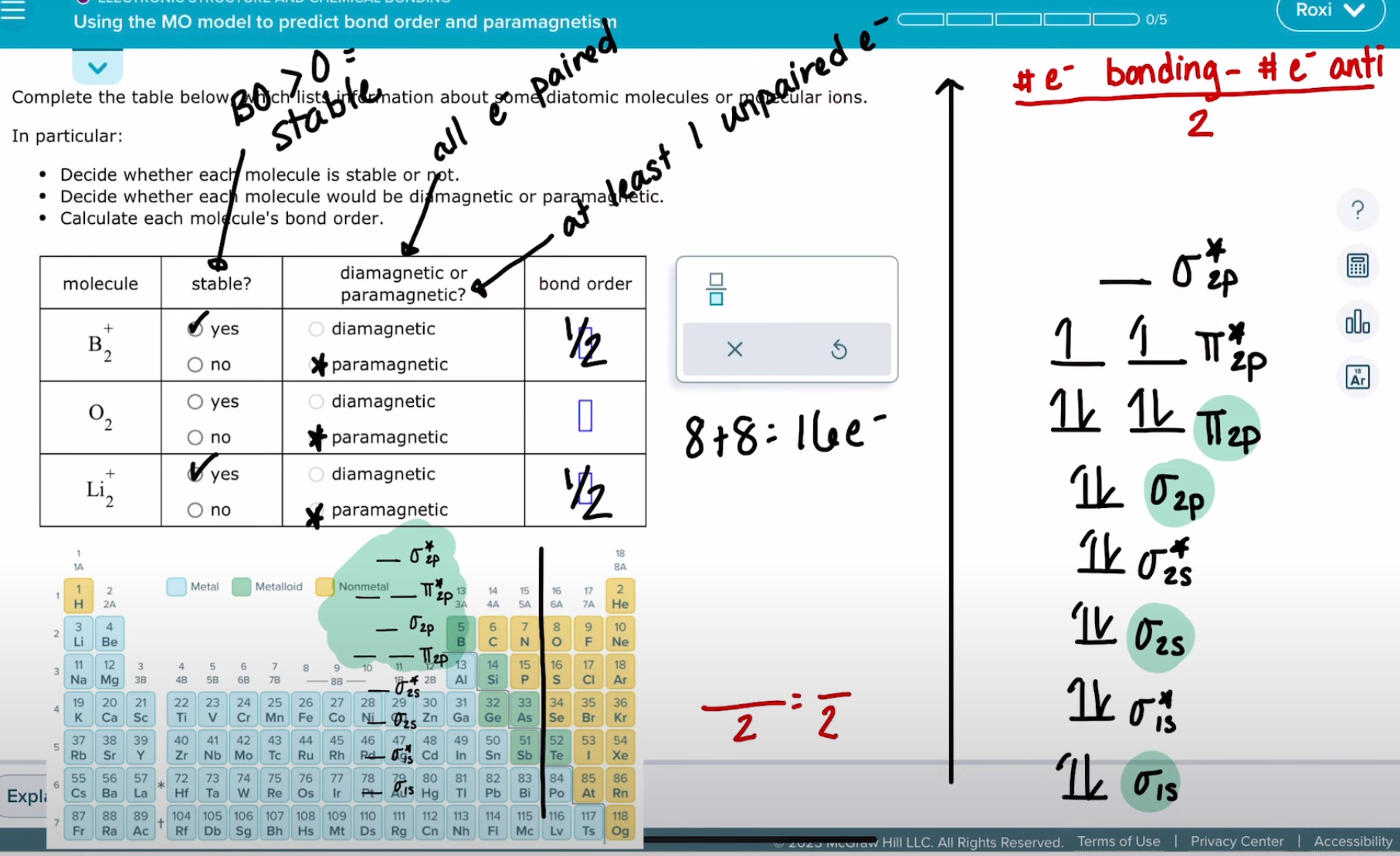
using the MO molecule to predict bond order and paramagnetism
Identifying the intermolecular forces between atoms, ions and molecules

Identifying important physical properties of liquids
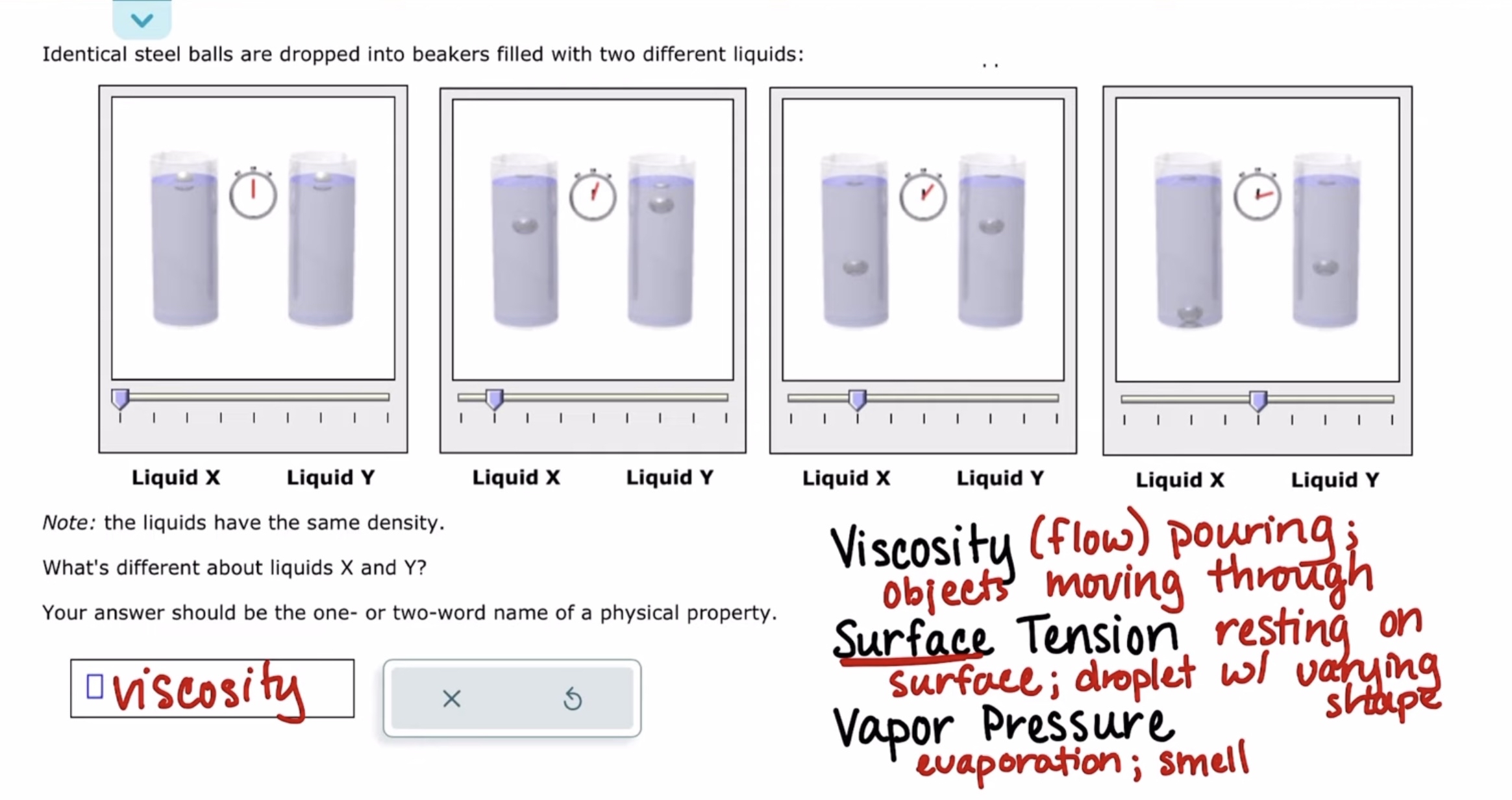
predicting the type of solid formed by a compound

using a phase diagram to predict phase at a given temperature and pressure
identifying phase transitions on a heating curve