Gen Chemistry 3 exam
Starting with the orbital diagram of boron atom, describe the steps needed to construct hybrid orbitals associated with the bonding in BF3:
promotion (excitation): one electron from the full 2s orbital is promoted to an empty 2p orbital. this creates three unpaired electrons: one in the 2s orbital and two in the 2p orbitals (2s^1 2p^x^1 2p^y^1)
hybridization: these three orbitals (one s and two p) mix or hybridize to form three equivalent hybrid orbitals of equal energy
What is the name of these orbitals?
these are called sp²
[How to use VE’s to draw lewis dot structures]:
Identify the total number of valence electrons in the molecule. Count the valence electrons for each atom and sum them up. If the molecule is an ion, add or subtract electrons accordingly based on the charge.
Next, determine the central atom, typically the one with the lowest electronegativity, and arrange the surrounding atoms around it, connecting them with single bonds to account for the initial valence electrons used.
Distribute the valence electrons to satisfy the octet rule, forming bonds and lone pairs as necessary.
[Geometry plays a rule in]:
structural integrity of materials
polarity and attraction between molecules
phase of matter (s,l,g)
absorbance of light
biochemical reactions and drug behavior —> THALIDOMIDE
[The valence shell electron pair repulsion (VESPR) model]:
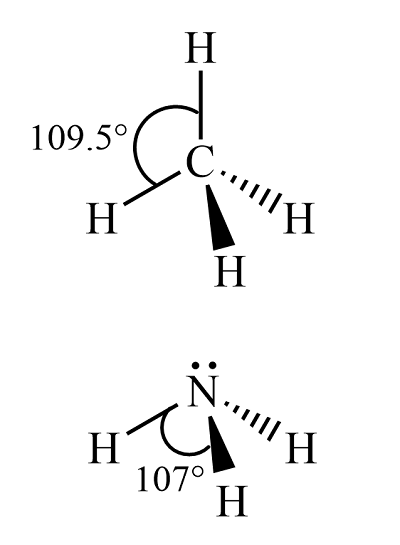
CH4, Use the valence shell electron pair repulsion theory to determine the shape of CH4.

4 covalent bonds (4 short VE pairs)
central C atom is surrounded by 4 areas of (-) charge (4 areas of e^- density)
since like charges repel, they want to get fair apart
the resulting geometry is tetrahedral (Added: with bond angles of approximately 109.5 degrees, which minimizes the repulsion between the electron pairs around the central carbon atom.
[Expanded octets]:
Central atom is large (3rd row of PT or beyond)
peripheral atoms are small, highly electronegative
Ex: PCL5, SF6 —> (5 areas and 6 areas of e^- density)
[Electron domain geometry]:
arrangement of all atoms of density (bonds and lone pairs) around the central atom
Linear, trigonal planar, tetrahedral, trigonal bipyramidal, octahedral, and their corresponding angles which determine the molecular geometry.
[molecular geometry];
arrangement of just the bonds around the central atom
Describes only the positions of the atoms, excluding lone pairs.
Linear, trigonal planar, bent, tetrahedral, trigonal pyramidal, bent



[location and effect of lone pairs]:
for tetrahedral and trigonal planar edg, a lone pair can go in any position (results in the same MG)
octahedral and trigonal bipyramidal lone pairs will go where there is most room since lone pairs are considered as larger of (-) charge than single bonds are
What does this mean?
[This means]:
octahedral: single lone pairs can occupy any position, two lone pairs will occupy opposite ends
trigonal bipyramidal: lone pairs occupy equilateral positions
[lone pairs affect bond angles]:
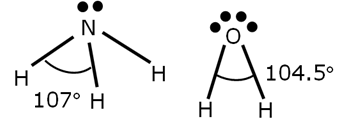
example: CH4, NH3, H20, all tetrahedral in EDG

[polarity]:
to determine molecular polarity you must:
draw the lewis structure and count the area if of e^- density
find EDG and then find MG
draw dipole moments vectors for each polar bond, each vector has: magnitude (EN of the bonded atom), and direction (which way the arrow points)
determine the overal (net) effect of all vectors combined,:
if all dipoles cancel (due to equal magnitude and opposite direction) the molecule is non-polar
if the dipole do not cancel, the molecule is polar
[examples]:
Carbon dioxide (CO2): Non-polar molecule due to symmetrical shape causing dipole moments to cancel out.
Water (H2O): Polar molecule as the bent shape does not allow dipole moments to cancel, leading to a net dipole moment. (The bent molecular geometry results in a permanent dipole moment, making molecules like sulfur dioxide SO2 polar as well.) WATER IS NOT LINEAR, DRAW IT GEOMETRICALLY ACCURATE, IT IS BENT, if you draw it linear it will make it look non-polar and water is polar
Ammonia (NH3): Polar molecule because the trigonal pyramidal shape results in a net dipole moment.
COCL4 is non polar
Phosphorus trichloride (PCl3): Polar molecule due to its trigonal pyramidal shape, which creates a net dipole moment.
SO3 is a nonpolar molecule because its trigonal planar geometry leads to a symmetrical distribution of charge, effectively canceling out any dipole moments—> forms a resonance structure
cclf is polar? check photo
Chlorine trifluoride, or ClF3, expanded octet, is a polar molecule due to its T-shaped molecular geometry, the bond dipoles do not cancel out.
SF6
[lone pairs can influence polarity]
molecules with a lone pair on the central atom or with different peripheral atoms may be polar or non-polar, you need to draw them to find out.
NH3 has greater EN but smaller dipole moment than NF3, this means NH3 is more polar than NF3.
UNIT 9 -oxidation, single replace, double, etc, TYPES OF REACTIONS
[Single replacement reaction]
single replacement reaction is when one element (typically a metal) replaces another in a compound
AB+C—> CB+A OR A + BC → AC + B
𝑍𝑛(𝑠)+2𝐻𝐶𝑙(𝑎𝑞)→𝑍𝑛𝐶𝑙2(𝑎𝑞)+𝐻2(𝑔) WITH METAL REPLACING METAL (OR HYDROGEN)
𝐶𝑙2(𝑔)+2𝑁𝑎𝐵𝑟(𝑎𝑞)→2𝑁𝑎𝐶𝑙(𝑎𝑞)+𝐵𝑟2(𝑙) WITH NONMETAL REPLACING NONMETAL
jim usually does experiment but usually gets fire alarm going so showed video, check slides—i think reaction had to do with aluminum
THERMITE VS AUTOMOBILE
cardinals moving field—→ thermite, aluminum oxide
[Double replacement reaction]
A double displacement reaction is also known as metathesis
Is a chemical process where two ionic compounds in an aqueous solution exchange ions to form two new compounds.
These reactions typically produce a precipitate, gas, or molecular compound like water, driving the reaction forward.
AB+CD—> AD +BC
Precipitation Reaction Example: Lead(II) nitrate and potassium sulfide forming lead sulfide (solid) and potassium nitrate.
Neutralization Reaction Example: An acid and a base reacting to form water and a salt.
Gas-Forming Example: Sodium sulfide and hydrochloric acid reacting to produce hydrogen sulfide gas.
[Oxidation number]
Valence electrons = e^- farthest away from the center of an atom (and closest to the VE’s of a neighbouring atom) shared or donated VE’s result in a bond
ON is # that indicates the change in the # of VE’s between an element by itself and when it is apart of a compound, used to determine ion changes, name compounds, and follow e^- transfer in reactions
[oxidation number rules]
C, each O in O2, each H in H2, Fe, Ar, all have ON =0
Cl^- is -1 , N^-3 is -3, O^-2 is -2
Mg^+2 is +2. Cr^+6 is +6
Monatomic ions: The oxidation number is equal to the charge of the ion (e.g., Na^+ has ON = +1, Cl^- has ON = -1).
Oxygen generally has ON = -2, except in peroxides where it is -1.
Hydrogen usually has ON = +1 when bonded to nonmetals and -1 when bonded to metals.
The sum of ON’s has to add up to the overall charge of the compound or ion, for neutral compounds, this sum is zero
NaCl: Na is +1 and Cl is -1 so sum is zero
CrO4^-2: for O -2(4)=-8 and Cr is +6 which you get -2 which adds up to the -2 seen in the formula
other metals have positive ON number equal to their group number on PT
Li in LiH? The ON for Li is +1
Ca in CaCl2? The ON for Ca is +2
Zn in ZnS? The ON for Zn is +2
Al in Al2O3? The ON for Al is +3
F has ON of -1, H is +1 but can be -1 if bonded to a metal per rule 4, O has ON of -2, halogens (group 7 nonmetals) have an ON of -2, S and Se (group 6 nonmetals) have ON of -2, N and P (group 5 nonmetals) have ON of -3
MgF2: Mg is +2 and F is -1(2)=-2
NaH: Na is +1 and H is -1
K2O2: K is +1(2)=+2 and O is -1(2)=-2
Rules must go IN ORDER start w the first rules then to rules 5 bc of exceptions and how transition metals go last due to it being ever changing
ASSIGN THESE THINGS IN ORDER
[oxidation numbers] DOUBLE CHECK
H2O2 oxidation numbers : H is +1(2)=+2 and O is -1(2)=-2, resulting in a total oxidation state of 0 for the molecule
CO2 oxidation numbers:C is +4 and O is -2 (2) = -4, resulting in a total oxidation state of 0 for the molecule.
NH3 oxidation numbers: N is -3 and H is +1(3)=+3, giving a total oxidation state of 0 for the molecule.
SiCl4 oxidation numbers: Si is +4 and Cl is -1(4)=-4, resulting in a total oxidation state of 0 for the molecule.
SO4^-2 oxidation numbers: S is +6 and O is -2(4) = -8, resulting in a total oxidation state of -2 for the ion.
Cr2O3 oxidation numbers: Cr is +3 and O is -2(3) = -6, resulting in a total oxidation state of 0 for the compound. You do Cr last due to it being transition metal
Cr2O7^-2 oxidation numbers: Cr is +6 and O is -2(7) = -14, resulting in a total oxidation state of -2 for the ion.
HNO3 oxidation numbers: H is +1, N is +5, and O is -2(3) = -6, resulting in a total oxidation state of 0 for the compound.
[How to predict single replacement reactions]
Zn(NO3)2+Mg—> Mg(NO3)2 + Zn but if we did Zn(NO3)2+Cu—> No reaction for that, Zn gives up NO3^-2 to Mg but not Cu, why?
well in first reaction, zn is +2, mg 0 —> mg +2 and zn 0
This is a RE-DOX reaction
Zn and Mg are changing their ON in a reduction/oxidation reaction where zinc is being oxidized from an oxidation number of +2 to 0, while magnesium is being reduced from 0 to +2. In contrast, copper does not have a strong enough oxidizing power to displace zinc from its compound, so no reaction occurs.
Zn gains 2e^- and Mg loses 2e^-
Mg is more “reactive metal than Zn and mg is more likely than zn to lose e^- and become a + ion in a solution
Reduction is a process in which a substance gains electrons, leading to a decrease in its oxidation number
Oxidation is a process in which a substance loses electrons, causing an increase in its oxidation number
[activity series]
metals of higher activity will be oxidized by those of lower activity , hence why they can replace metals of lower activity in a compound
H can also act like a metal in some reactions
Zn(NO3)2 was mixed in class—NO REACTION
AgNO3 was mixed in class—-YES REACTION IT TURNED A BIT BLUE/SILVER AND FUZZY so it became cu+2zn(no3)2—> cu(no3)2+2ag
Mg placed in HCL, was mixed in class—-YES REACTION became BUBBLY AND CLOUDY, it became mg+2hcl—>mgcl2+h2
[DOUBLE REPLACEMENT]
a chemical process where two ionic compounds in aqueous solution exchange ions to form two new compounds.
Generally expressed as AB+CD—>AD+CB
these reactions often produce a solid precipitate, a gas, or a weak electrolyte like water.
They are also known as metathesis or double decomposition
IT IS NOT A REDOX REACTION, YOU CAN’T PREDICT THAT WAY OF ACTIVITY SERIES, YOU MUST LOOK AT THE NATURE
[decomposition reactions]:
composition breaks down into two or more simpler substances
Often requiring energy input like heat, light, or electricity as it really can not be done alone or spontaneously by itself
AB—> A+B
Most decomposition reactions are endothermic, meaning they require an input of energy (heat, electricity, or light) to break the chemical bonds.
Thermal Decomposition (Thermolysis): Decomposition induced by heat. For example, calcium carbonate decomposes when heated: CACO3—>CAO+CO2
Electrolytic Decomposition (Electrolysis): Decomposition induced by electricity. a classic example is breaking water into hydrogen and oxygen: 2H2O—> 2H2+O2
Photolytic Decomposition (Photolysis): Decomposition induced by light.
Hydrogen Peroxide: (often catalyzed by manganese dioxide or enzyme catalase): H2O2—→ 2H2O + O2 THIS IS SPONTANEOUS but faster w light
Reversibility: can be either reveserable or irraversable
[combination/synthesis]
two or more reaction combine to form a compound (opposite of decomposition)
2Na+Cl2—>2NaCl
[Combustion reaction]:
combination and or/ re-dox reaction where a fuel burns in the presence of an oxidizer (typically O2), producing heat and a flame or glow
GUMMY BEAR KClO3 being burned
{Jim and the reactions of combustion in class}
Alch and money in the alch bottle
cornstarch over the fire and BOOM burns bigger flame
spore moss on hand thats used as a lubricant like the ones of condoms as it is smooth powder
flash powder over the fire big big FLAME OVER FIRE
steel wool over fire
Mg—> magnesium over the flame it CREATED LIGHT
model t car could run on alch, ethanol with the cork thingy SOMETHING SHOT RIGHT OUT…cork?
balloon, hydrogen, put the fire at the balloon and big explode and pop with fire in the air
[precipitation reaction]
double replacement reaction in which a SOLID (precipitate) is formed in a solution
NaI and Pb(NO3)2 are mixed together in a solution: identify iions NaI—> Na^+ + I^- and Pb(NO3)2—> Pb^+2 + NO3^- … Na+NO3—>NaNO3 AND PB + I—> PbI2
2NaI+Pb(NO3)2—>2NaNO3+PbI2
then add phases using solubility rules
NaNO3: soluble so it remains in aqueous form (aq).
PbI2: insoluble according to solubility rules, therefore it precipitates as a solid (s).
write ionic equations for the reaction
2Na^+ (aq) 2I^- (aq) + Pb^+2 (aq) +2NO3^- (aq)—> 2Na^+ (aq) + 2NO3^- (aq?) + PbI2 (S)
spectator ions are ions that don’t take part in the reaction and appear on both sides (Na and NO3 in this case)
2Na^+ (aq)2I^- (aq) + Pb^+2 (aq) +2NO3^- (aq)—>2Na^+ (aq) + 2NO3^- (aq?)+ PbI2 (S)net ionic equation is the complete ionic equation w no spectator ions
Pb^+2 (aq) + 2I^- → PBI2 (s)
[acid base]
Firstly, what is the definition of acid and bases? The definition differs based on which you go by, there are three:
Arrhenius: Acid is produces H+ (used interchangeable with H3O^+) when added to water. A base produces OH^- when added to water
Bronsted-lowry: acid donates H^+ to another species and becomes a conjugate base. A base accepts H^+ from another species and becomes a conjugate acid.
based on bronsted-lowry: NH3 (aq) + H2O (l) —> ←— NH4^+ (aq) + OH^- (aq)
Base + acid —> ←- conjugate acid + conjugate base
lewis: an acid is electron-pair acceptor (electron-deficient), it is any molecule or ion that can accept a pair of nonbonding electrons. A base is an electron-pair donor (electron-rich), it is any molecule or ion that can donate a pair of nonbonding electrons. They are generally electron-rich, possessing lone pair electrons. (maybe: This then forms a covalent bond between them.)
WILL SHOW UP ON TEST, EXAMPLES LIKE HANDOUT 9
There is also things called amphoteric, where they can act as both acid and base.
Examples of amphoteric substances include water (H₂O), bicarbonate (HCO₃⁻), and amino acids, which can participate in acid-base reactions depending on the conditions.
[Their strengths]
a strong acid would be one that dissociates completely in solution, releasing all of its hydrogen ions (H⁺) or H2O^+, while a weak acid only partially dissociates, establishing an equilibrium between the undissociated acid and its ions.
a strong base would be one that dissociates completely into OH^- in solution.
an acidic solution is one where the solution [H3O^+] > [OH^-]
a basic solution is one where the solution [H3O^+] < [OH^-]
a neutral solution is one where the solution [H3O^+] = [OH^-]
an auto-ionization dissociation(???) H3O + H2O —> ←- H3O^+ + OH^-, auto something in water .. i think it is autoionization
autoionization: water at 25 °C has [H⁺] = 1.0×10⁻⁷ M (pH = 7).
[H3O+][OH-]=10×10^-14 (at 25 degrees C)
REVIEW AND GO OVER HOW TO DO CONC PROBLEMS
[The PH scale]
the PH scale is a logarithmic scale to represent acidity/bascitiy
PH=-log[H3O+]
[H3O+]= 10^-ph
log means what power/exponent of 10 produces a given number. 10 is your base, log base 10.
log(100)=2 bc log base 10(100)=2 due to 10² = 100
[when you have an acidic solution]
PH<7.0
so stomach acid which primarily HCl, lemon juice, soda
[neutral solution]
PH=7.0
pure water, human blood
[basic solution]
PH>7.0
lye (NaOH or KOH), bleach, antacid
[strong acid/base neutralization reactions]
HCl(aq) + NaOH(aq)
strong acid + strong base
HCl (aq) —→ H+ (aq) + Cl- (aq)
NaOH (aq)—→ Na+ (aq) + OH- (aq)
Complete ionic: H+ (aq) + Cl- (aq) + Na+ (aq) + OH- (aq)—→ Na+ (aq) + Cl- (aq) + H2O (l)
Get rid of spectator ions for ur net ionic, spectator ions are ones that show up on both side of the equation:
H+ (aq) +
Cl- (aq) + Na+ (aq)+ OH- (aq)—→Na+ (aq) + Cl- (aq)+ H2O (l)net ionic: H+ (aq) + OH- (aq) —→ H2O (l)
[Neutralization reaction]
double replacement reaction where an acid and base react to form a salt and often water
[weak acid and bases]
page 7 on handout so check that example
ch3cooh or ch3coo is weak acid
[jim experiment]
flask has hydrochloric acid (stomach acid similarity) and deflated balloon over the flask containing HCl, adds antiacid to flask and puts deflated balloon over the flask and the balloon fills up and expands as the reaction between the antacid and hydrochloric acid produces carbon dioxide gas, demonstrating the neutralization reaction. This experiment effectively illustrates the principles of acid-base reactions and gas evolution, showcasing how antacids neutralize stomach acid and relieve symptoms of heartburn.
[GAMESHOW, SOME ON PHONE]
most