ENZYMES
IMPORTANCE OF ENZYMES
All biochemical reactions are enzyme catalyzed in the living organism
Enzymes play an important role in metabolism, diagnosis, and therapeutics.
DEFINITION
Enzymes - are biological catalysts
They catalyze nearly all the chemical reactions taking place in the cells
of the body.
They are proteins that increase the rate of reaction by looking for an
alternative pathway that has a low energy of activation
CHARACTERISTICS
They are not altered or consumed during reaction.
Reusable
Enzymes show specificity to the reaction they control
Enzymes are sensitive to their environment so they can be controlled
by adjusting the temperature, the pH or the substrate concentration
STRUCTURE OF ENZYMES
TERMS TO UNDERSTAND BIOCHEMICAL NATURE OF ENZYMES
Active Site - The area on the enzyme where the substrate or
substrates are attached to.
Substrate – the reactant in the chemical reaction
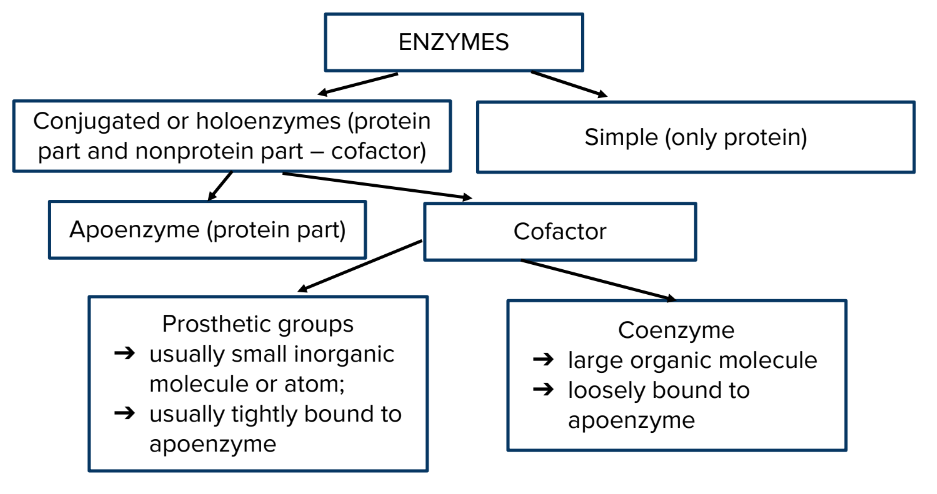
Apoenzyme – the enzyme without its non-protein moiety is termed as
apoenzyme and it is inactive.
Holoenzyme – the active enzyme with its non-protein component.
Cofactor - is a non-protein chemical compound that is bound (either
tightly or loosely) to an enzyme and is required for catalysis.\
Coenzymes - large organic molecule, loosely bound to apoenzyme
Prosthetic groups - Could be lipids metals or carbohydrates, usually tightly bound to apoenzyme
NAMING ENZYMES
The name of an enzyme in many cases end in –ase
For example, sucrase catalyzes the hydrolysis of sucrose
The name describes the function of the enzyme
For example, oxidases catalyze oxidation reactions
Sometimes common names are used, particularly for the digestion
enzymes
For example, pepsin and trypsin
Some names describe both the substrate and the function
For example, alcohol dehydrogenase oxides ethanol
CLASSIFICATIONS OF ENZYMES
Enzymes Are Classified into six functional Classes (EC number Classification) by the International Union of Biochemists (I.U.B.) on the Basis of the Types of Reactions that they catalyze
Oxidoreductases - Catalyze oxidation-reduction reactions
Transferases - Catalyze group transfer reactions
Hydrolases - Catalyze hydrolysis reactions where water is the acceptor of the transferred group
Lyases - Cleave various bonds by means other than hydrolysis and oxidation.
Isomerase - Catalyze isomerization reactions
Ligases - Join two molecules with covalent bonds , Catalyze reactions in which two chemical groups are joined (or ligated) with the use of energy from ATP.
Enzymatic Action
THE ACTIVATION ENERGY - Chemical reactions need an initial input of energy
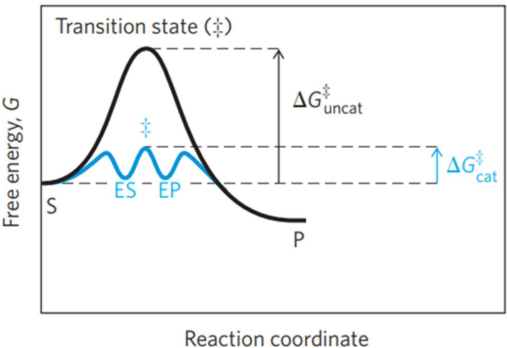 During this part of the reaction the molecules are said to be in a Transition state.
During this part of the reaction the molecules are said to be in a Transition state.
● Biological systems are very sensitive to temperature changes.
● Enzymes can increase the rate of reactions without increasing the temperature.
● They do this by lowering the activation energy. They look for a new reaction pathway “a short cut”
● Enzyme controlled reactions proceed 108 to 1011 times faster than corresponding non-enzymatic reactions.
Enzyme-Substrate Binding
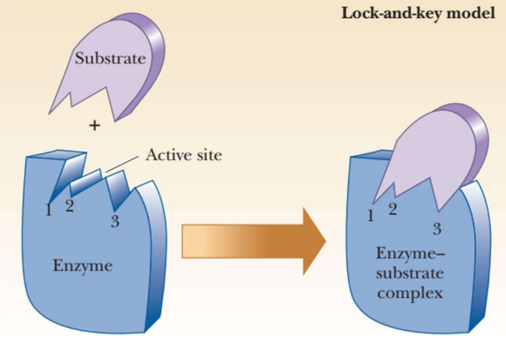
Lock and Key model
the active site has a rigid shape
only substrates with the matching shape can fit
the substrate is a key that fits the lock of the active site
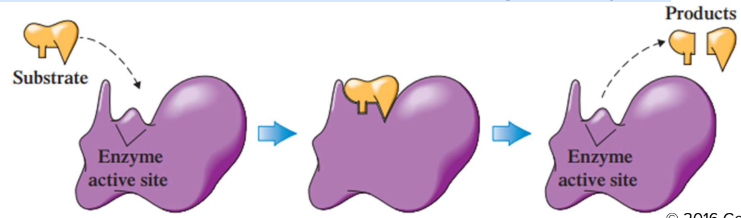
Induced Fit Model
the active site is flexible, not rigid. It can change its conformation.
the shapes of the enzyme, active site, and substrate adjust to maximize the fit, which improves catalysis
there is a greater range of substrate specificity
this model is more consistent with a wider range of enzymes
Enzyme Specificity
Absolute -Catalyze only one reaction for single substrates
Group - Catalyze only one reaction for substrates with similar functional groups
Linkage - One type of reaction for a specific type of bond
Stereochemical - act on a particular stereoisomer. Chirality is inherent in an enzyme active site
Factors Affecting Enzyme Activity
Environmental Conditions
Temperature
pH
Substrate concentration
Enzyme Concentration
Cofactors and Coenzymes
Allosteric regulation
Positive regulation
Negative regulation
Enzyme Inhibitors
Effect of Temperature
Temperature increases enzyme activity up to its optimum temperature
Optimum temperature is the temperature at which enzymatic reaction
occur fastest
For most enzymes the optimum temperature is about 40°C
Many are a lot lower, cold water fish will die at 30°C because their
enzymes denature
A few bacteria have enzymes that can withstand very high temperatures
up to 100°C
Most enzymes however are fully denatured at 70°C
Effect of pH
If pH slightly different from the enzyme’s optimum value, there will be small changes in the charges of the enzyme and it’s substrate’s molecule.
This change in ionization will affect the binding of the substrate with the active site.
different optimum pH where enzyme activity is at its highest
Extreme changes in pH can cause denaturation