Quantum Numbers and Electron Configuration
Quantum Numbers
- Quantum numbers are the distribution of electrons in an electron; orientation of orbitals
Four Quantum Numbers
- Principal Quantum Numbers - main energy level
- princical quantum numbers are denoted by n
- a principal quantum number’s values are positive integers (1,2,3,4…)
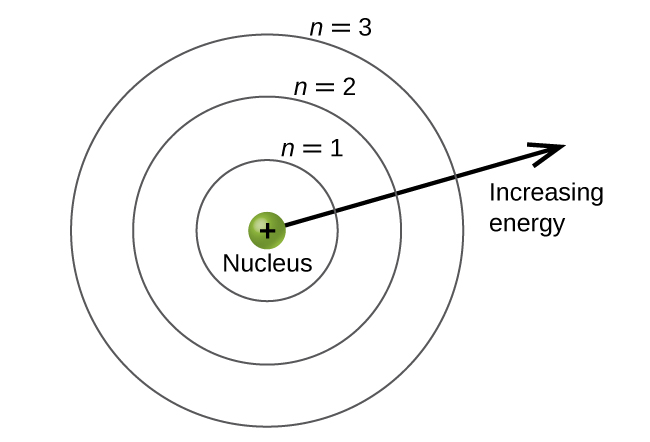
- Azimuthal Quantum Number
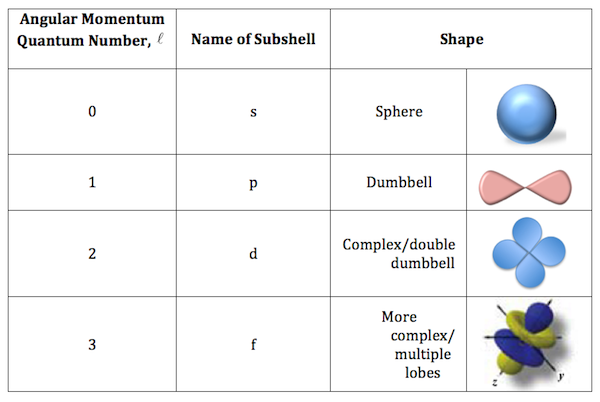
- azimuthal quantum numbers - describe the shape of an orbital
- azimuthal quantum numbers are denoted by l.
- azimuthal quantum numbers are also known as “angular momentum”
- The shapes of an azimuthal quantum number consists of the: sphere, dumbell, double dumbell, multiple
- s - 0
- p - 0,1
- d - 0,1,2
- f - 0,1,2,3
- Magnetic Quantum Numbers - are orientations of orbitals
- orbitals - place or location wherein electrons are created
- the values of magnetic quantum #s are: -3,-2,-1,0,1,2,3
- magnetic quantum #s are denoted by - Ml
- Spin Quantum Numbers - this quantum no. focuses on the direction of the electron
- the values of spin quantum numbers are: +1/2,-1/2
- spin quantum numbers are denoted by Ms
Electron Distribution
- Electron distribution is the arrangement of electrons in the space around the nucleus of an atom; distribution of electrons in the orbitals of an atom
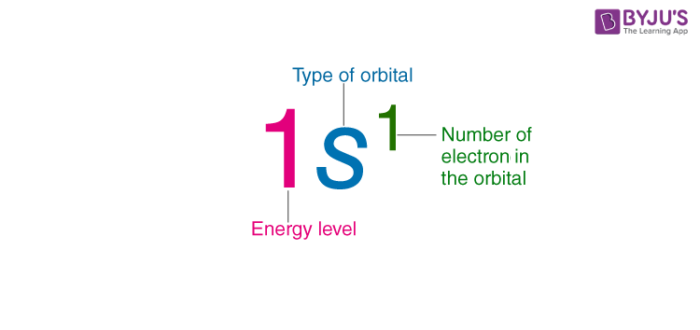
- electron distribution uses symbols of orbitals and the number of electrons (written as a superscript) that occupy each orbital
- the number of electrons is written as a superscript
Two Types of Configuration
- Electronic Configuration
- Orbital Diagram
Different Principles
- Aufbau’s Principle - states that electrons should occupy the lowest orbital before those w/ highest energy level
- Pauli’s Exclusion Principle - no two elements in an atom can possess the same set of quantum numbers
- Hud’s Rule - most stable arrangement of electrons in subshell is the one w/ greatest no. of parallel spins
Parts of an Electron Configuration
Maximum no. of Electrons
- s - 2 (subshell), -1 (orbit)
- p - 6 (subshell), -3 (orbit)
- d - 10 (subshell), -5 (orbit)
- f - 14 (subshell), -7 (orbit)
1s, 2s, 3p, 3s, 3p, 4s, 3d, 4p, 4d, 4d, 5p, 6s, 4f
Max no. of Energy Distribution
- 1st - 2
- 2nd - 8
- 3rd -18
- 4th - 32