12. Fluorescence spectroscopy
Luminescence is the emission of light by a substance not resulting from heat (cold body radiation). There are 3 forms of biological luminescence: fluorescence, phosphorescence, and chemiluminescence. Fluorescence is where light is absorbed and rapidly emitted at a longer wavelength
Morse potential energy curve
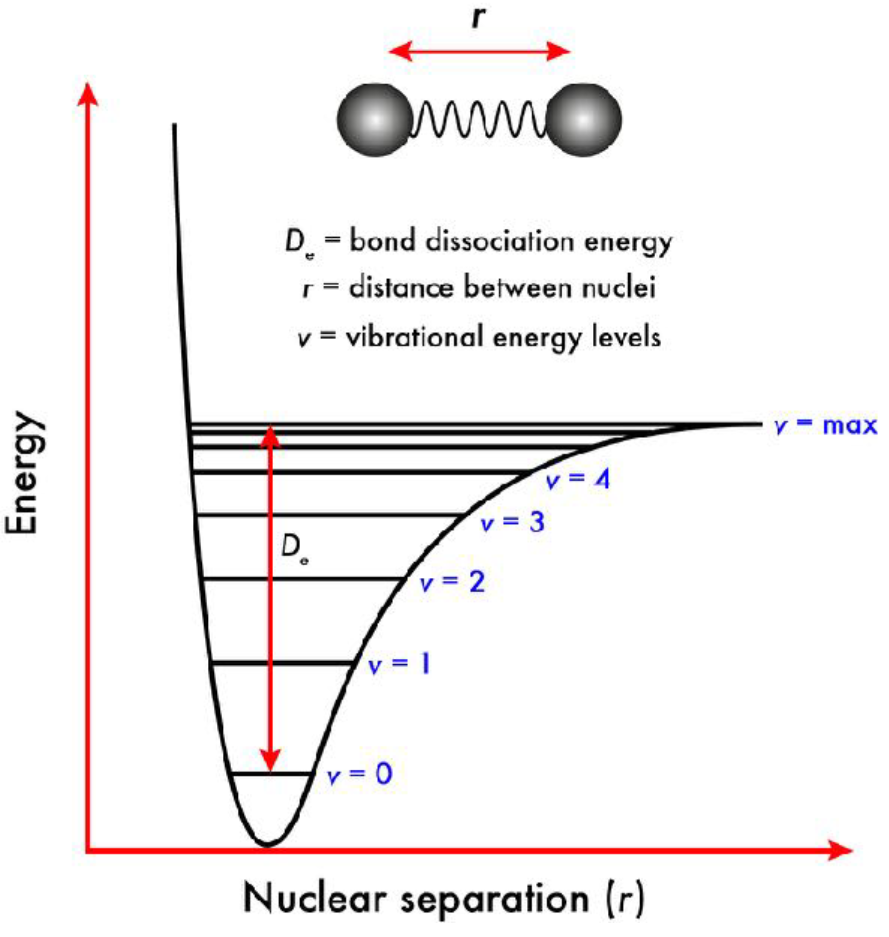
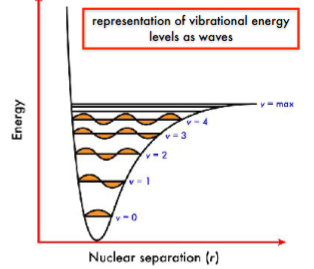
Increasing vibrational levels have increasing number of nodes. As electrons are promoted to higher vibrational levels, the internuclear separation can increase. The ground state is at v = 0. At v = max, the bond will break
Jablonski diagram
Shows electronic states of a molecule and the transitions between them. Used to illustrate different types of electronic transition
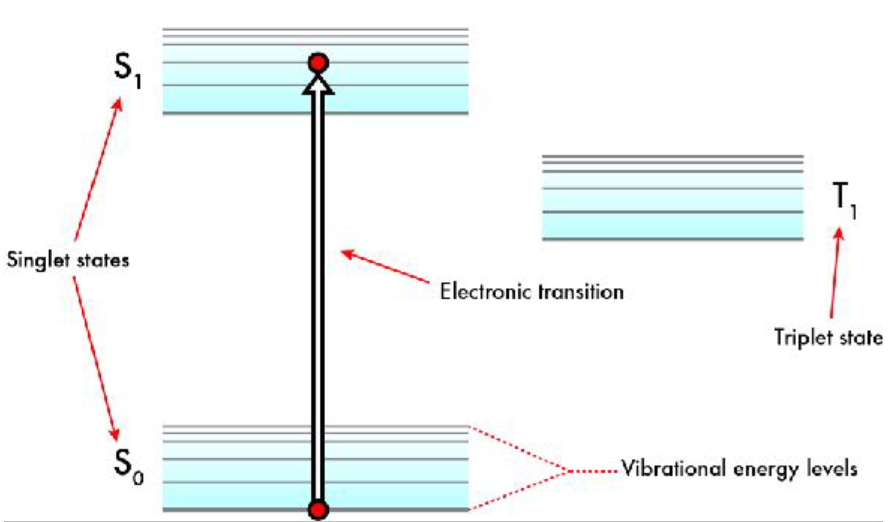
No unpaired electrons = singlet state
2 unpaired electrons with opposing spin in different energy levels = excited singlet state
2 electrons with same spin in 2 different energy levels = excited triplet state
Internal conversion
When electrons are excited to a higher electronic state, they experience internal conversion (vibrational relaxation) in which they lose energy and “bounce” down from a higher vibrational state until it reaches the ground vibrational state of the excited electronic state
Absorbance
Electron absorbs a photon and is promoted to a higher electronic state. It undergoes internal conversion and then returns to the ground electronic state, emitting the energy as heat (non-radiative emission)
The absorption spectrum is indicative of the excited state energy levels available to the ground state electron
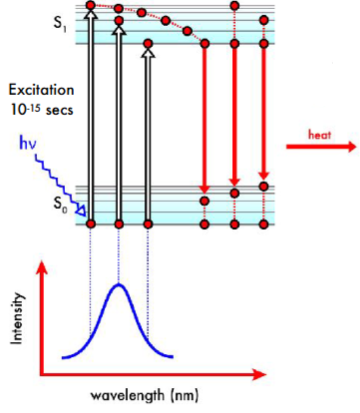
A chromophore is a molecule that absorbs light
Fluorescence
Electron absorbs a photon and is promoted to a higher electronic state. It undergoes internal conversion and then returns to the ground electronic state, emitting energy as a photon
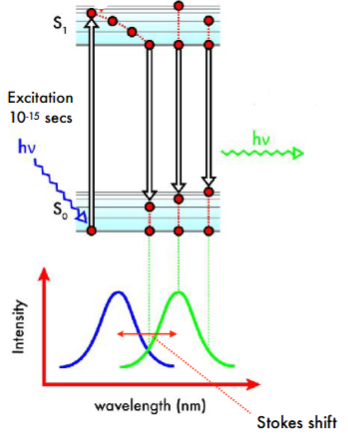
Due to energy loss during internal conversion, the wavelength that is absorbed is shorted than the wavelength emitted, resulting in stokes shift, the rightward shift of the emission spectrum relative to the absorption spectrum
The emission wavelength and intensity of the fluorophore is sensitive to its chemical environment, however, fluorescence is much more sensitive than absorbance
A fluorophore is a molecule that fluoresces
Phosphorescence
Electron absorbs a proton and is promoted to a higher electronic state. It undergoes internal conversion and intersystem crossing, in which the electron flips its spin and an excited triplet state is achieved. Then the electron returns to the ground electronic state, re-flipping its spin, and emitting energy as a photon
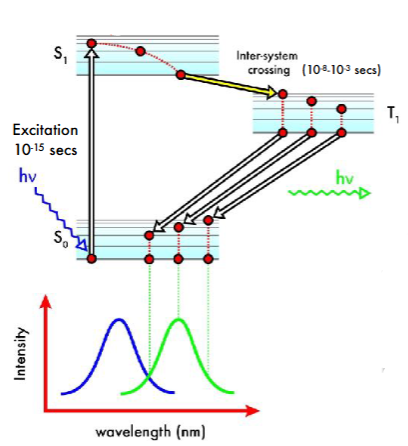
Intersystem crossing is very slow and so phosphorescence lasts longer than fluorescence. Stokes shift still occurs due to internal conversion
A phosphor is a molecule that phosphoresces
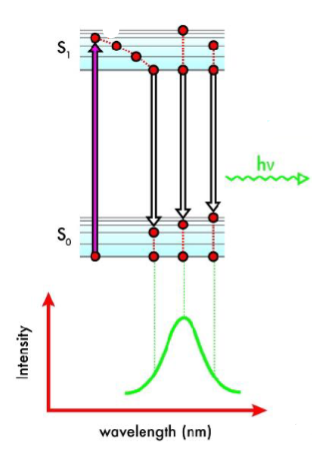
Chemiluminescence
Electron is promoted to a higher electronic state via a chemical reaction (instead of a photon). The electron undergoes internal conversion and returns to the ground state, emitting energy as a photon
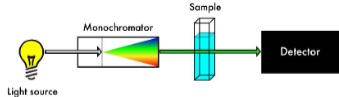
Spectrometers
Absorbance is measured at 180o to the light source
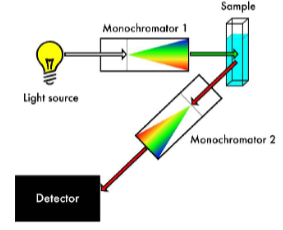
Fluorescence measured at 90 degrees to excitation beam
Fluorescence efficiency
Not all absorbed photons are emitted by a fluorophore. Energy can be lost through heat or be transferred to a quenching molecule. Fluorescence efficiency is defined by the quantum yield () of a fluorophore
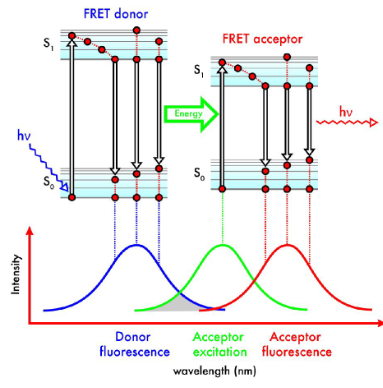
Forster resonance energy transfer (FRET)
FRET is a type of non-radiative energy transfer. Following the excitation of a donor fluorophore, emissions would be observed from an acceptor fluorophore. This is strongly distant dependent