Molecules/Atoms Review
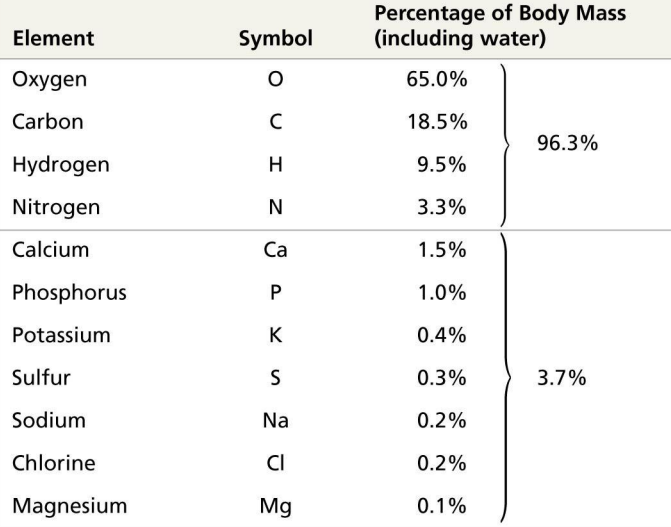
Elements in Human Body
N - nuetral : 1.7 x 10-24 grams = 1amu
E - Negative : 0amu
P - Positive : 1.7 x 10-24 grams = 1amu
Mass = P + N
Atomic = P in nucleus
Isotopes “2 atoms of an element that differ in nuetrons”
Radioactive Isoptopes
decay spontaneously
Radiometric Dating
half-life - parent isotope decaying into daughter isotope at fixed rate , half life is the amount of time needed for one half of the parent to decay into daughter
Chem behavious in valence shells
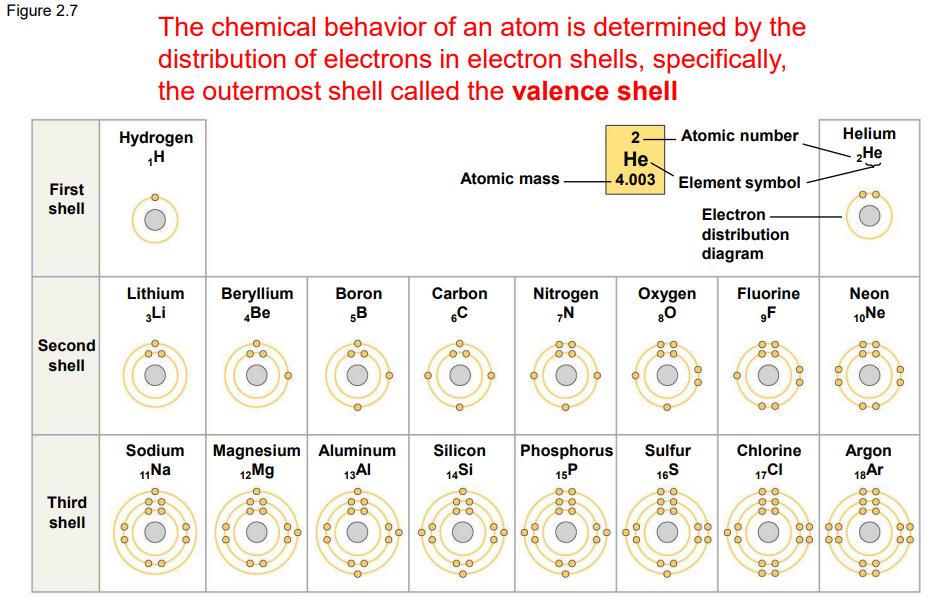
Atoms with incomplete valence shells share/transfer electrons or “Chemical Bonds”
Covalent bond - sharing between 2 atoms where the shared electrons count towards each part of the atoms valence shell
Non-polar ; electrons shared equally
Polar ; one atom is more electronegative
Ionic Bonds - atom taking electrons from other atom
“salts”
Hydrogen Bonds - basically hydrogen bonding between 2 diff molecules
Chem reactions make and break chem bond
starting molecules = reactans
final molecules = products
Most reactinos are reversible
products of the forward reaction becoms reactans for the reverse reaction
Chemical equilibrium is reached when the forward and reverse reactions occur at the same rate
At equilibrium, the concentrations or reactants/products dont change
Law of conservation of mass – no matter is created or destroyed in a chemical reaction, atoms are just rearranged.