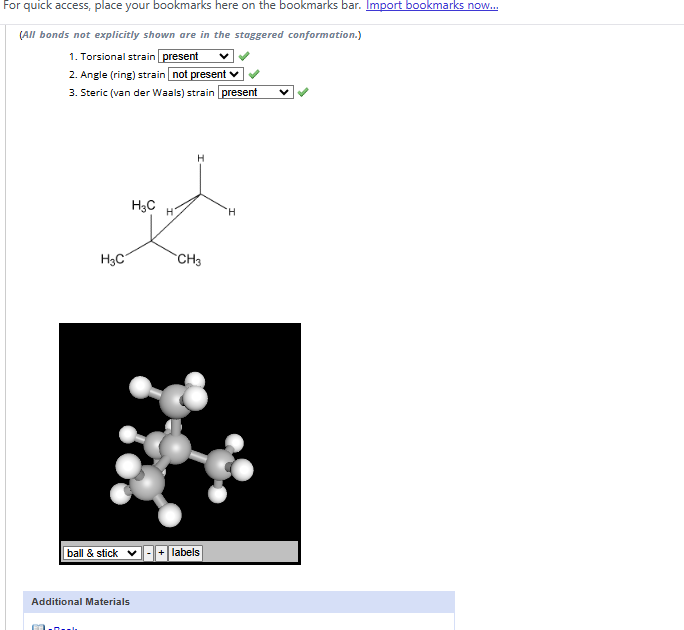
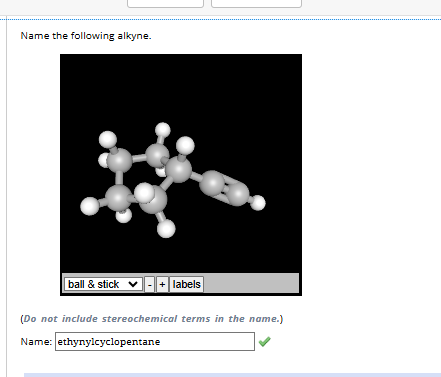
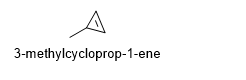
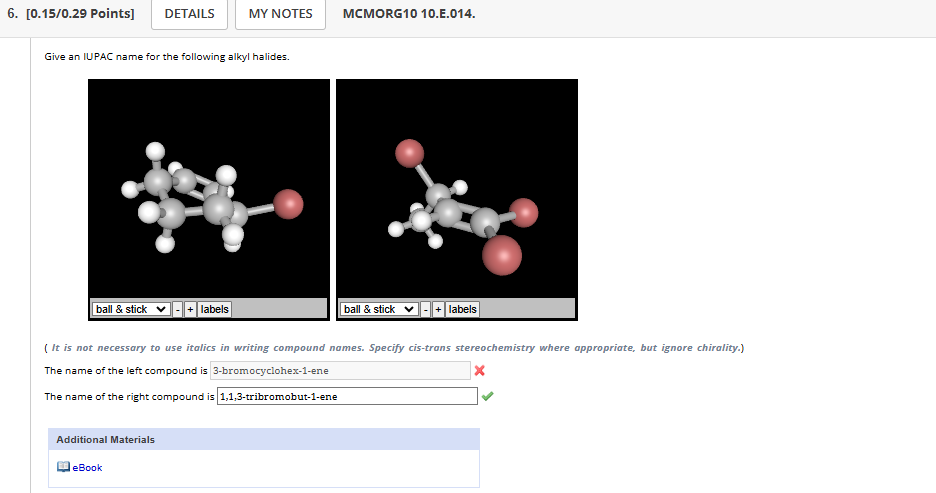
4.1-4.12
4.1
Molecular handedness arises from stereoisomers called enantiomers, which are not identical to their mirror images.
Molecules like CH3X and CH2XY can superimpose on their mirror images and are not considered chiral.
CHXYZ cannot be superimposed on its mirror image, similar to how left and right hands relate.
Enantiomers result when a tetrahedral carbon is bonded to four different substituents.
Lactic acid has two enantiomers: (+)-lactic acid and (−)-lactic acid, found in sour milk.
Only the (+) form of lactic acid occurs in muscle tissue.
Enantiomers cannot be superimposed on each other due to differing group arrangements.
4.2
A chiral molecule is not identical to its mirror image and cannot overlap with its enantiomer.
A molecule is achiral if it possesses a plane of symmetry, where one half mirrors the other (e.g., a coffee mug).
Propanoic acid is achiral due to its symmetry.
Lactic acid is chiral as it lacks a symmetry plane.
Chirality typically arises from a tetrahedral carbon bonded to four different groups, known as a chirality center.
Recognizing chirality centers can be challenging:
5-bromodecane is chiral.
Methylcyclohexane is not chiral due to its symmetry.
2-Methylcyclohexanone is chiral due to its non-symmetrical bonding.
Various examples demonstrate chirality and chirality centers in organic compounds.
4.3
The study of chirality began in the early 19th century with Jean-Baptiste Biot's investigations into plane-polarized light.
Ordinary light consists of electromagnetic waves oscillating in many planes perpendicular to its direction of travel.
A polarizer allows only light waves oscillating in one plane to pass, resulting in plane-polarized light.
Biot discovered that plane-polarized light passing through certain organic solutions can rotate the plane of polarization.
Substances that rotate polarized light are called optically active while others are not.
The rotation can be measured using a polarimeter, which consists of a sample tube and an analyzer.
Optically active molecules can rotate polarized light either to the left (levorotatory, -) or to the right (dextrorotatory, +).
Rotation extent depends on sample concentration and path length; doubling either increases the rotation observed.
Specific rotation, [α]D, standardizes measurements: [α]D = Observed rotation / (Pathlength × Concentration).
Examples of specific rotations include: (+)-lactic acid [α]D = +3.82, (−)-lactic acid [α]D = -3.82.
Other compounds with specified rotations include Penicillin V (+233), Sucrose (+66.47), and Morphine (-132).
The specific rotation of a pure stereoisomer is a fixed value, equal and opposite to its enantiomer.
Observed rotation of a mixture of enantiomers falls between the specific rotations of the two.
An observed rotation of 0° indicates a 50% composition of each enantiomer.
More useful to measure enantiomeric excess (ee), which ranges from 0-100%.
Enantiomeric excess can be calculated using:
ee = (Observed rotation / Specific rotation of pure enantiomer) × 100%
In 1848, Louis Pasteur studied tartaric acid salts, discovering distinct crystals that were nonsuperimposable mirror images.
Original 50:50 mixture was optically inactive, while sorted piles were optically active with equal but opposite specific rotations.
Pasteur described these as enantiomers, with identical physical properties but different optical activities.
4.4
Structural drawings visually represent stereochemistry; written methods indicate the 3D arrangement at chirality centers.
Cahn–Ingold–Prelog rules rank the four groups attached to a chirality center:
Rule 1: Rank atoms based on atomic number; highest = 1st, lowest (usually hydrogen) = 4th.
Rule 2: If first atoms are equivalent, compare second, third, or fourth atoms until the first difference is found.
Rule 3: Multiple-bonded atoms count as single-bonded equivalents.
Establish stereochemical configuration by orienting the molecule with the lowest ranking group (4) pointing away:
Clockwise sequence (1 → 2 → 3) = R configuration.
Counterclockwise sequence = S configuration.
Example: For (−)-lactic acid, functions indicate R configuration; for (+)-lactic acid, the opposite.
Configuration does not directly relate to optical activity; both (S)-glyceraldehyde and (S)-alanine show different activity despite having S configuration.
Absolute configuration assignments became certain in 1951 through X-ray diffraction
4.5
Molecules like lactic acid, alanine, and glyceraldehyde have one chirality center and two stereoisomers.
Molecules with more than one chirality center increase complexity; a molecule with n chirality centers can have up to 2n stereoisomers.
Threonine (2-amino-3-hydroxybutanoic acid) has two chirality centers (C2 and C3) and four possible stereoisomers.
Stereoisomers can be grouped into pairs of enantiomers: 2R,3R is a mirror image of 2S,3S, and 2R,3S of 2S,3R.
Diastereomers are stereoisomers that are not mirror images, akin to hands from different people; they share some similarities but differ in configurations at some chirality centers.
Enantiomers have opposite configurations at all chirality centers, while diastereomers have opposite configurations at some but the same configurations at others.
The 2S,3R stereoisomer occurs naturally in plants and animals and is essential for humans.
In cases where two diastereomers differ at only one chirality center, they are termed epimers (e.g., cholestanol and coprostanol differ at C5).
4.6
Tartaric acid has four stereoisomers.
2R,3R and 2S,3S are nonsuperimposable mirror images, representing a pair of enantiomers.
2R,3S and 2S,3R are superimposable and identical due to a plane of symmetry, making them achiral.
Compounds that are achiral but have chirality centers are called meso compounds.
Tartaric acid has three stereoisomeric forms: two enantiomers and one meso form.
The meso form has a symmetry plane through the C2–C3 bond.
Physical properties of tartaric acid stereoisomers are:
(+) tartaric acid: melting point 168–170 °C, [α]D +12, density 1.7598 g/cm³, solubility 139.0 g/100 mL.
(−) tartaric acid: melting point 168–170 °C, [α]D −12, density 1.7598 g/cm³, solubility 139.0 g/100 mL.
Meso tartaric acid: melting point 146–148 °C, [α]D 0, density
4.7
Increasing chiral carbons make typical skeletal structures difficult to draw.
Carbohydrates have numerous chirality centers, necessitating a quick representation method for stereochemistry.
Emil Fischer proposed Fischer projections in 1891 to represent tetrahedral carbons on flat surfaces.
In Fischer projections:
Tetrahedral carbon atoms are shown by two crossed lines.
Horizontal lines indicate bonds coming out of the page; vertical lines indicate bonds going into the page.
Example: (R)-glyceraldehyde is illustrated as a Fischer projection.
Comparing Fischer projections helps determine if they represent the same or different enantiomers.
Allowable movements of Fischer projections:
180° rotation retains the same substituent groups.
90° rotation inverts meanings, changing the molecule (e.g., (R)- to (S)-glyceraldehyde).
One group can be held steady while the others rotate without changing stereochemistry.
Assigning R,S designations involves three steps:
Rank the four substituents.
Place the lowest-ranked group at the top of the projection.
Determine the direction of rotation for the remaining groups to assign R or S.
Carbohydrates with multiple chirality centers stack in Fischer projections, with the carbonyl carbon at the top.
Fischer projections do not accurately depict the true three-dimensional conformation of molecules; they may resemble shapes similar to curled bracelets.
4.8
Pasteur's work with tartaric acid involved crystallizing two optically active forms: 2R,3R and 2S,3S.
The initial form was a 50:50 mixture of two chiral enantiomers, known as a racemate or racemic mixture (± or d,l).
Racemates exhibit no optical rotation; the (+) rotation of one enantiomer cancels the (−) rotation of the other.
Pasteur successfully separated the racemic tartaric acid into its enantiomers (+) and (−).
Fractional crystallization was effective for tartaric acid, but it is not suitable for most racemates, necessitating other methods.
The common method for resolving racemates of chiral carboxylic acids involves an acid-base reaction with an amine base to create ammonium salts.
For example, the reaction of racemic lactic acids with achiral methylamine yields a racemic mixture of ammonium salts.
When reacting with a chiral amine like (R)-1-phenylethylamine, the two salts formed are diastereomers, differing in properties.
This allows for their separation, and subsequent acidification isolates the pure en
4.9
Isomers are compounds with the same chemical formula but different structures.
There are two fundamental types of isomers:
Constitutional Isomers: Atoms are connected differently.
Types include:
Skeletal isomers
Functional isomers
Positional isomers
Stereoisomers: Atoms are connected in the same order but have different spatial arrangements.
Types include:
Enantiomers
Diastereomers
Cis-trans isomers (a subclass of diastereomers, which are non-mirror-image stereois
4.10
Chirality in molecules often arises from four different substituents bonded to a tetrahedral atom.
Common chirality centers include carbon, nitrogen, phosphorus, and sulfur atoms.
Trivalent nitrogen (with its lone pair) is tetrahedral and can theoretically create enantiomers, but typically undergoes rapid inversion, making isolation difficult.
Trivalent phosphorus (phosphines) has slower inversion, allowing isolated stable chiral forms, such as (R)- and (S)-methylpropylphenylphosphine.
Divalent sulfur compounds are achiral, whereas trivalent sulfur compounds (sulfonium salts) can be chiral and configurationally stable.
An example of a stable chiral sulfonium salt is S-adenosylmethionine, which acts as a biological methyl donor and is involved in many metabolic pathways.
S-adenosylmethionine has stable stereochemistry at sulfur and is configurationally stable at room temperature, while its R enantiomer is not biologically active.
4.11
Prochirality: A molecule can transition from achiral to chiral in one chemical step.
Example: 2-butanone is prochiral and converts to chiral 2-butanol with hydrogen addition.
Enantiomeric Production: Which enantiomer forms depends on the reaction at the carbonyl group's face (Re or Si).
Ranking Groups: Involves ranking three groups attached to the sp2-hybridized carbon to determine the Re (clockwise) and Si (counterclockwise) orientation.
Sp3-Hybridized Prochirality Centers: Changing one group on an sp3-hybridized atom (like the carbon in ethanol) can create a chirality center.
Pro-R and Pro-S: Identifying which atom's replacement results in R or S chirality gives the terms pro-R and pro-S.
Biological Reactions: Many involve prochiral compounds, such as the addition of H2O to fumarate to yield (S)-malate.
Stereochemistry Studies: Understanding prochirality helps elucidate detailed mechanisms in biochemical reactions, including those in the citric acid cycle.
4.12
Different enantiomers of a chiral molecule have the same physical properties but usually differ in biological properties.
Chirality can affect the biological properties of drugs.
Example: Fluoxetine (Prozac) is an effective antidepressant but has no activity against migraines; the pure S enantiomer is effective for migraine prevention.
To have biological effects, a substance must fit into a receptor with a complementary shape.
Biological receptors are chiral, allowing only one enantiomer to fit; mirror-image enantiomers do not fit properly.
Interaction between chiral molecules and receptors can be visualized like fitting a left hand into a right-handed glove.
Prochiral substrates can undergo selective reactions in chiral environments.
Example: Reaction of ethanol with NAD+ shows how chiral environments influence selectivity.
Chiral receptors make certain substituents chemically distinct based on their arrangement in the environment.
Drugs approved by the FDA can come from various sources but often exist as single enantiomers rather than racemates.
Penicillin V is an example of a chiral drug with specific configurations required for activity.
Ibuprofen is sold as a racemate (R and S mixture), but only the S enantiomer is effective against pain and inflammation.
The presence of the inactive R enantiomer can impact the efficacy of the S enantiomer, making synthetic approaches for producing single enantiomers desirable.