Phenol
Phenol and its uses
-OH group bonded to an aromatic ring
Alcohols and phenols have similar reactions, but the proximity of the delocalised ring influences some reactions
Used in manufacture of disinfectants, detergents, aspirin, etc.
Carbolic acid - Joseph Lister in 19th century
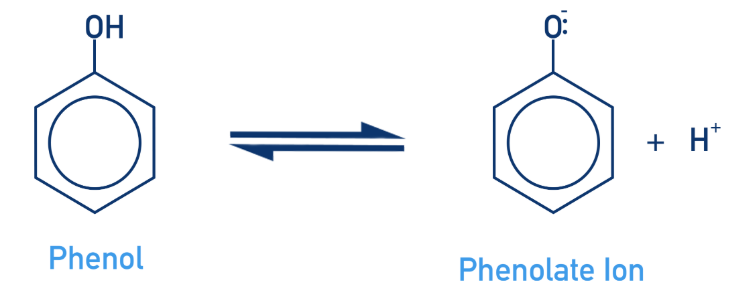
Phenol as a weak acid
Less soluble in water than alcohol
Partially dissociates, forming the phenoxide ion and a proton (H+)
The highly electronegative oxygen atom from the ion attracts negative charge strongly
More acidic than alcohols, but less than carboxylic acid
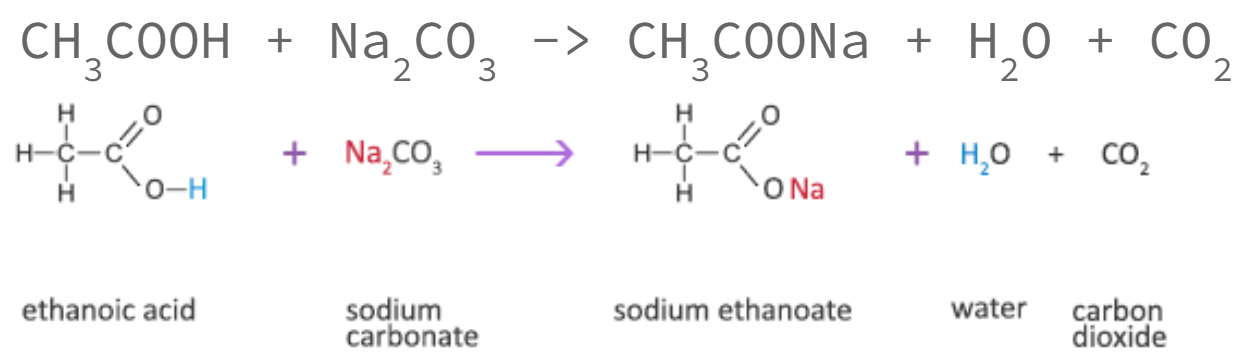
To distinguish between phenols and carboxylic acid: Na2CO3
phenol has no reaction
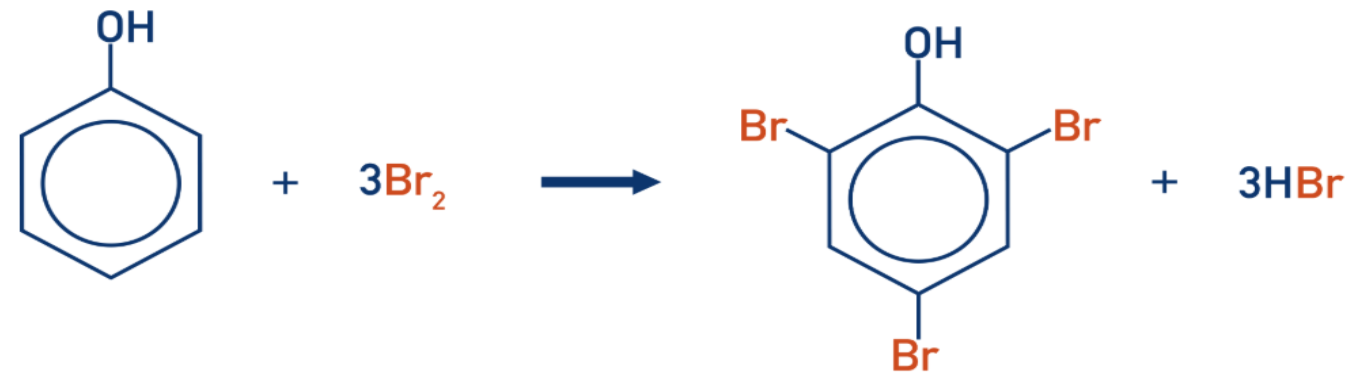
Electrophilic substitution - bromination
Phenol reacts with an aqueous solution of bromine
Reaction decolourises the bromine water
Forms a white precipitate of 2,4,6-tribromophenol
With phenol, a halogen carrier is not required and reaction is carried out at room temperature
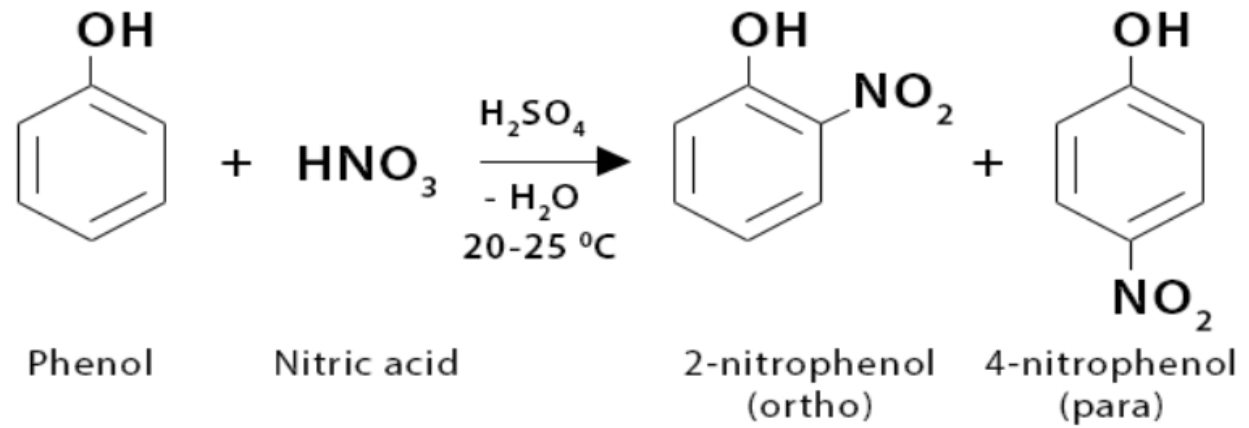
Electrophilic substitution - nitration
Phenol reacts readily with dilute nitric acid at room temperature
A mixture of 2-nitrophenol and 4-nitrophenol is formed
Gives a mixture of ortho (1,2) and para (1,4) nitrophenols
Reactivity compared to benzene
Reactivity of phenol > reactivity of benzene
A lone pair of electrons from the p-orbital of the -OH group gets donated to the π-system of phenol
The electron density of the benzene ring in phenol is increased
Electrophiles are more attracted to phenol than benzene
Br2 and HNO3 react more readily with phenol than with benzene
Phenol can be nitrated with dilute nitric acid; concentrated acid is not necessary
Halogen carrier catalyst is not required for bromination - increased electron density can polarise Br2 molecules