Honors Biology Unit 1: Biochemistry
The basic unit of life is the cell, which is composed of various biomolecules including carbohydrates, proteins, lipids, and nucleic acids that perform essential functions.
Bonding:
Matter- Anything with mass and takes up space.
-Atoms: Smallest unit of matter.
→ Protons- Positive (Nucleus)
→ Neutrons- Neutral (Nucleus)
→ Electrons- Negative (Electron shell)
-Atoms are electrically neutral, meaning they have the same number of protons and electrons.
Elements: Made up of one type of atom
-Carbon -Phosphorous
-Oxygen -Nitrogen
-Hydrogen -Calcium
Atomic Interaction:
-Done by valence electrons (Outermost electrons)
→Shared electrons-Covalent Bond
→Donated/Received electrons-Ionic Bond
-Isotope: Atoms with the same number of protons, but different number of neutrons
-Carbon 12- 6p 6n 6e
-Carbon 13-6p 7n 6e
-Carbon 14-6p 8n 6e
-Carbon dating: Determines the age of fossils
→H2O
→Octet rule: Atoms want 8 valence electrons
Ionic Bonds: (Metals and Non-metals)
-Transferring electrons
-Ions are atoms with charges→ Two types
→Cation(+)
→Anion(-)
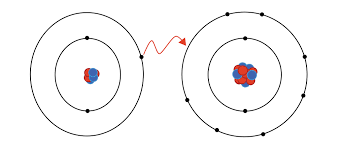
Covalent Bond: (2 Non-metals)→Sharing electrons
→Polar: Unequal sharing
→Non-polar: Equal sharing (That’s why oil and water don’t mix, because oil is non-polar while water is polar)
Water and its solutions:
Water:
-Forms hydrogen bonds
-Polar
-Solid form is less dense than the liquid form
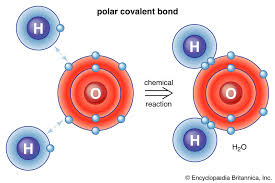
Water molecule:
-Polar molecule
→Polar covalent bond- Electrons are unequally shared between atoms
-Oxygen pulls the electrons closer to it as opposed to hydrogen
→O=Partial - H=Partial +
-Hydrogen bond: Attraction of a slightly positive hydrogen atom to a slightly negative atom (Oxygen, Nitrogen, or Fluorine)
Cohesion and adhesion:
-Cohesion: Attraction of water molecules to one another
-Adhesion: Attraction of water molecules to the sides of the container.
→Together, they cause surface tension (Liquid surfaces minimizing their surface area).
Capillary action:
-Cohesion and adhesion acting together to allow water to move against the force of gravity.
Heat capacity and living things:
-Water has a high heat capacity
Takes more energy to cause it to boil due to the hydrogen bonds
Living things are 60-70% water
Nearly every chemical reaction in living things takes place in water
Solutions:
-Mixture: Two or more elements that are not chemically combined and not in a specific ratio.
-Solution: Mixture in which all components are evenly distributed.
→Solute: Dissolved
→Solvent: What is doing the dissolving
Suspensions:
-Mixture in which molecules don’t dissolve; able to settle out of the mixture
Ex. Blood and muddy water
pH scale:
-Used to determine the concentration of H+ ions in a solution
-Low= more acidic High=less acidic
Acids and Bases:
-Acids are a compound that releases H+ ions in a solution
→pH below 7 Ex. Lemon juice and vinegar
-Bases are compounds that release OH- ions in a solution
→pH above 7 Ex. Bleach and soap
Buffers:
-Weak acids and bases that resist changes in pH
-The body is filled with them
→Keeps sharp changes from happening
-Bicarbonate buffer
-Phosphate buffer
Macromolecules:
Carbon:
-Essential to life
-All organic compounds contain carbon
Characteristics of carbon:
4 valence electrons
-Can form bonds with 4 atoms at once
-Variety of bonds with a variety of elements
Carbon forms a variety of molecules:
-Chains
-Rings
-single, double, triple bonds
→Forms bonds with: Hydrogen, Oxygen, Nitrogen, and Phosphorous
Macromolecules:
-Big organic molecules in living things
→ most are formed by polymerization (Converting monomers to polymers)
-Monomer: small units that join together to form polymers
-Dehydration synthesis: Taking out water in order to build a covalent bond
-Hydrolosis: Using water to destroy a covalent bond
Types of macromolecules:
-Carbohydrates-Quick energy
-Lipids-Energy storage
-Proteins-Build and maintain muscle tissue
-Nucleic acid-Storing genetic material
Carbohydrates:
-Sugar
-Starch
-Cellulose
→ Hydrogen, Oxygen, Carbon.
-In a 1:2:1 ratio
3 types:
-Monosaccharide: Glucose, Fructose, Galactose
-Disaccharide: Lactose, Maltose, Sucrose
-Polysaccharide: Starch, Glycogen, Cellulose
→ Starch and cellulose reside in plants, while glycogen is excess sugar in animals
Glycosidic bond: Bond between 2 sugars
-Simple carb test: Benedict’s
-Complex carb test: Iodine
Lipids: Fats
-Organic
-Non-polar
-Not water soluble
Elements:
-Carbon
-Oxygen
-Hydrogen
-Not in a 1:2:1 ratio
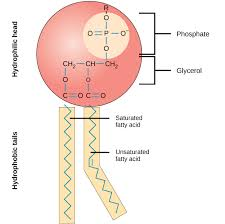
Monomers: Polymer→Triglyceride: 1 glycerol + 3 fatty acid chains
-Glycerol
-Fatty acid
Energy: Carbs and lipids are the biggest energy molecules
-Carbs: 4kcal/molecule
-Lipids: 9kcal/molecule
-Ester bond: Bond formed between glycerol and fatty acids
Functions:
-Energy storage
-Insulation
-Waterproofing
-Cell membrane
-Chemical messengers
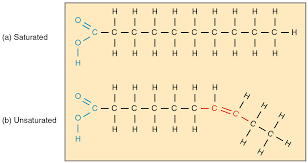
Saturated fats:
-Single bonds in the fatty acid chain
-Solid at room temperature
-Animal fats
Unsaturated fats:
-One or more double bonds
-Liquid at room temperature
-Plant-based fats
Steroid hormones:
-Estrogen
-Testosterone
Cell membrane:
2 ways:
-Cholesterol: Gives structure
-Phospholipid: Makes up the majority of the cell membrane
-Brown paper bag test: If translucent when held up to a light, lipids are present
Proteins:
-One of the largest most diverse macromolecules
Elements:
-Carbon
-Hydrogen
-Oxygen
-Nitrogen
Monomer: Amino acids (there’s 20 different types)
R-group-Caries with each amino acid. It’s what changes when the amino acid changes
Peptide bond: Carbon + Nitrogen covalent bond
Levels of organization:
-Primary structure: Linked amino acids
-Secondary structure: Alpha helix or Beta sheet
-Tertiary structure: 3-D structure (protein becomes functional)
-Quaternary structure- 2 or more tertiary proteins put together
Nucleic Acid: DNA and RNA
-Stores genetic info.
Monomer: Nucleotide
Elements:
-Carbon
-Hydrogen
-Oxygen
-Nitrogen
-Phosphorous
DNA: Adenine, Thymine, Cytosine, Guanine
RNA: Adenine, Uracil, Cytosine, Guanine
Enzymes:
Chemical reactions- Using one set of compounds to create a new substance
-Rearranging what atoms are connected to
-Reactants: What you start with
-Products: What you end up with
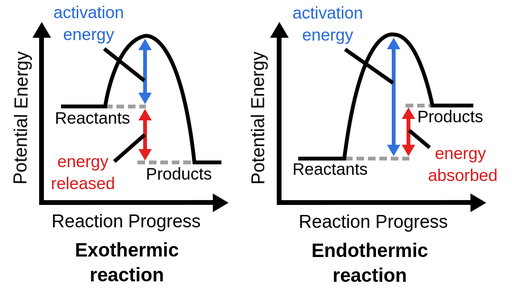
Energy:
-Exothermic: Energy is released
-Endothermic: Energy is absorbed
-Activation energy: The energy required to get a reaction started
Catalyst: Speeds up the rate of chemical reactions
-Not destroyed in the process
-Lower activation energy Ex. Heat
Enzyme: Biological catalyst→ Primarily found in cells
-Most are proteins
-Speed up reactions by lowering activation energy
-Very specific (Picky with what they’ll build or breakdown)
→One enzyme to one reaction
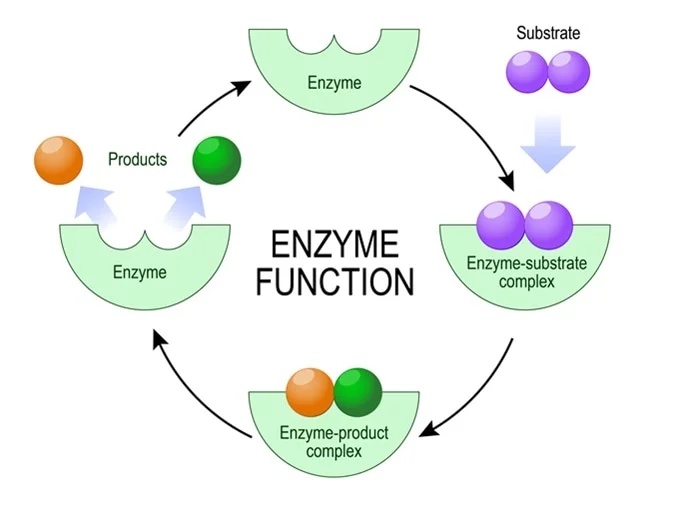
-Enzymes can be stopped from working
-They’re also picky with the conditions
Denaturing: Destroys the enzyme