Periodic table
Key Vocabulary
Observation: What you see happening (e.g., bubbles, colour change).
Inference: What you deduce from your observation (e.g., bubbles indicate gas production).
Sublimation: The transition of a solid directly to a gas or vice versa.
Inert: A substance that is unreactive.
Ionisation: The process of forming an ion.
THE PERIODIC TABLE - GROUPS 1, 7, 0, and Transition Metals
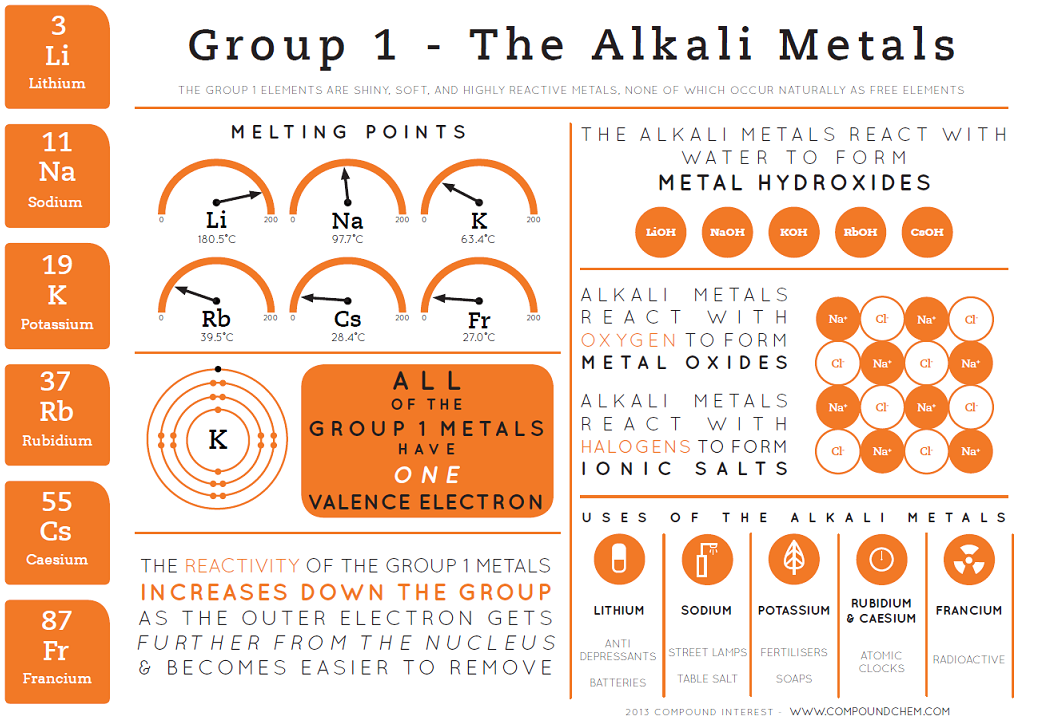
Group 1: The alkali metals:
Li (Lithium), Na (Sodium), K (Potassium)
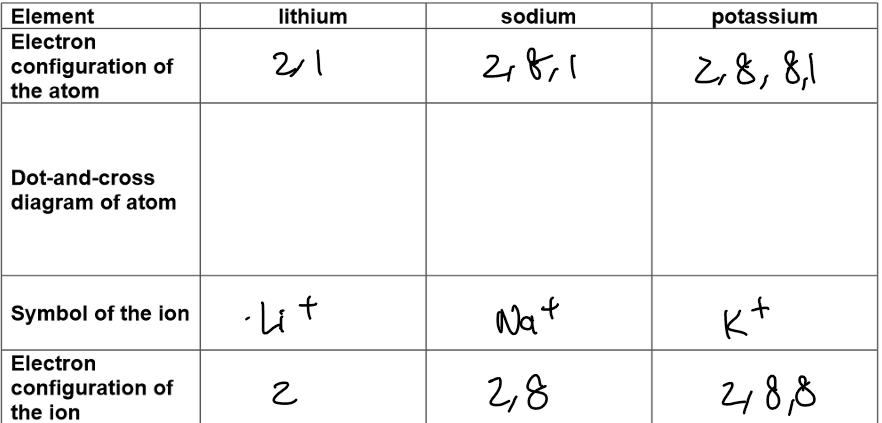
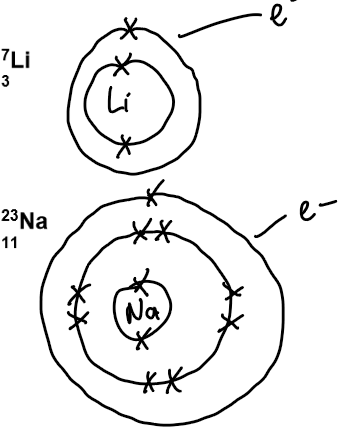
Electronic structure and ion formation:
Atoms have one electron in their outermost shell – hence Group 1.
All Group 1 elements have similar chemical properties because they all have one electron in their outermost electron shell.
They lose the electron in their outer shell to form positively charged ions, called cations, with a charge of +1.
Group 1 metals are stored under oil to prevent reactions with oxygen and moisture.
They are soft, can be cut with a knife, and tarnish quickly.
Low density, float on water as they react.
Rb and Cs are highly reactive, stored in sealed glass tubes.
Fr is rare, radioactive, and highly reactive.
Reactions of group 1 elements with cold water:
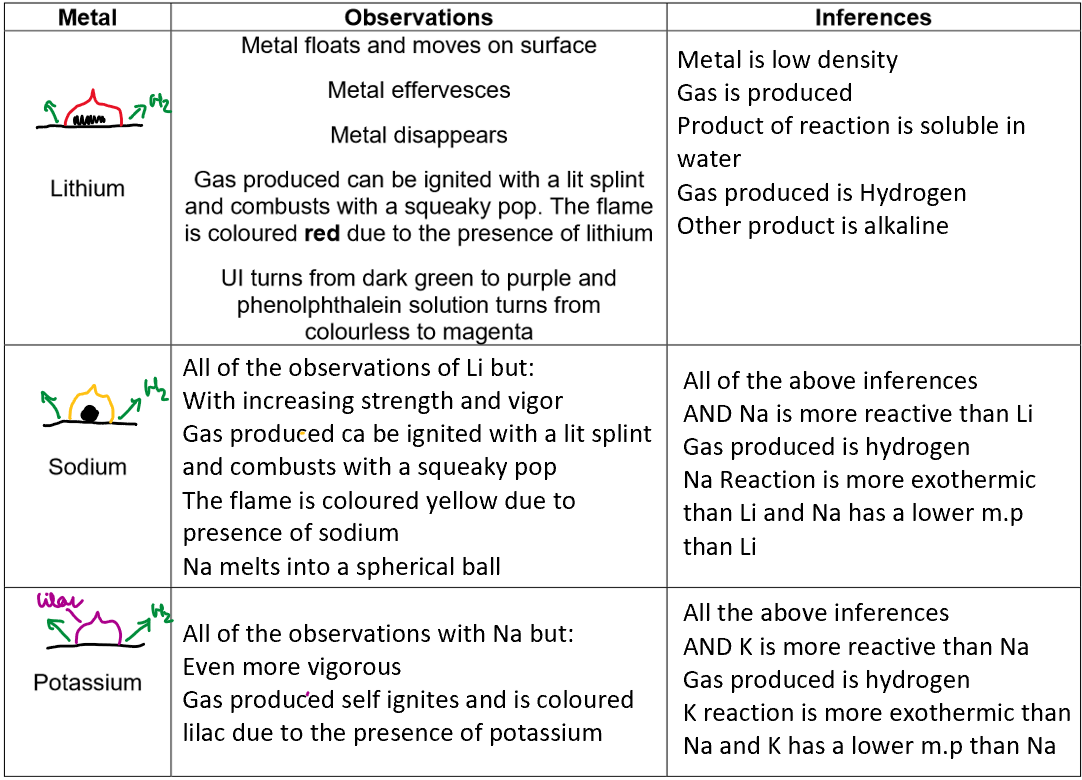
Conclusion:
General equation:
alkali metal + water → metal hydroxide + hydrogen
Lithium + water → lithium hydroxide + hydrogen
Sodium + water → sodium hydroxide + hydrogen
Potassium + water → potassium hydroxide + hydrogen
The metal hydroxides formed are ionic and dissolve in water, giving alkaline solutions with OH⁻ ions, resulting in a pH greater than 12. This is why Group I metals are called alkali metals.
Trends down group 1:
As you go down Group I (with increasing atomic number), alkali metals:
Have larger atoms due to an extra full shell of electrons per row.
Become more reactive because the outer electron is easier to lose. The electron is farther from the nucleus and more shielded, so the attraction between the positive nucleus and the negative outer electron is weaker.
Become denser, as atomic mass increases.
Are softer and easier to cut.
Have lower melting and boiling points due to weaker metallic bonding—the delocalised electrons are farther from the nucleus, weakening electrostatic attraction to the cations.
GROUP 7 – HALOGENS
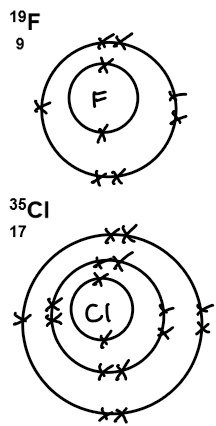
19F 35.5Cl 80Br 127I
9 17 35 53
• Safety: Toxic and reactive – handle with care, use in fume cupboards.
Physical Properties
Halogen | Formula | State (rtp) | Melting & Boiling Points | Density | Colour of Vapour |
|---|---|---|---|---|---|
Fluorine | F₂ | Gas | Very low | Very low | Pale yellow |
Chlorine | Cl₂ | Gas | Low | Low | Green-yellow |
Bromine | Br₂ | Liquid | Moderate | Medium | Brown |
Iodine | I₂ | Solid | Higher | Higher | Purple |
• Typical non-metals – poor thermal, electrical conductors, low mp.bp, brittle as solids.
• Elements exist as diatomic, simple molecular covalent molecules.
Halogens and Their Reactions
Safety:
Wear goggles and stay behind the safety screen.
Chlorine
Test: Turns damp blue litmus red, then bleaches it.
Reactions:
Fe + Cl₂ → FeCl₃
2Na + Cl₂ → 2NaCl
Bromine
Liquid bromine – volatile, forms brown vapour.
Bromine water – orange solution.
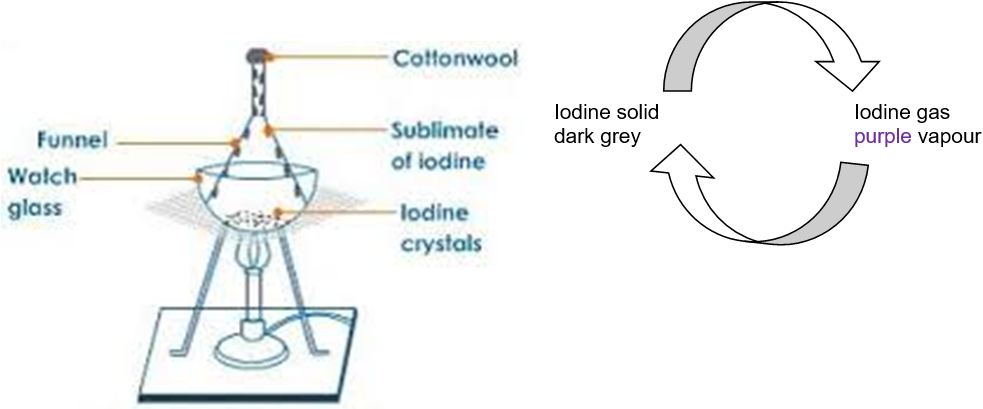
I₂(s) forms a purple vapour when heated, the purple vapour forms a shiny grey solid when cooled (no liquid phase).
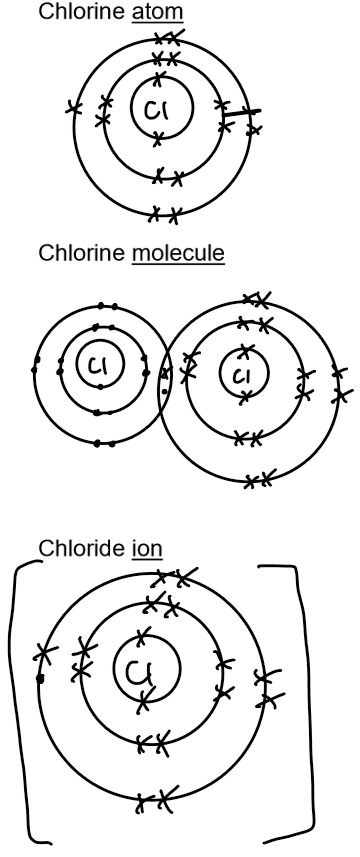
Electronic structure and ion formation
7 electrons in their outermost shell – hence Group 7.
Gain an electron to form negatively charged ions, called anions with a charge of -1.
Halogen atom | Electron arrangement of the halogen atom | Halogen element (covalent molecule) | Halide (negative ion) | Electron arrangement of the halide |
|---|---|---|---|---|
fluorine, F | 2, 7 | fluorine, F₂ | fluoride, F⁻ | 2, 8 |
chlorine, Cl | 2, 8, 7 | chlorine, Cl₂ | chloride, Cl⁻ | 2, 8, 8 |
bromine, Br | 2, 8, 18, 7 | bromine, Br₂ | bromide, Br⁻ | 2, 8, 18, 8 |
iodine, I | 2, 8, 18, 18, 7 | iodine, I₂ | iodide, I⁻ | 2, 8, 18, 18, 8 |
Dot and cross diagrams:
Trends down group 7:
As you go down Group VII (with increasing atomic number), the halogens become:
Larger – one more full electron shell is added each time.
Less reactive – harder to gain an extra electron. The outer shell is further from the nucleus with more shielding, so attraction for incoming electrons is weaker.
Denser – atoms have more mass.
Darker – fluorine is pale yellow, chlorine green, bromine a brown liquid, and iodine a dark grey solid.
Higher melting and boiling points.
Change from gases (F₂, Cl₂), to liquid (Br₂), to solid (I₂).
Changes in 5) and 6) are due to stronger intermolecular forces as molecules get larger.
Halogen Displacement Reactions
Safety:
Wear goggles and lab coat. Tuck chairs in. Follow teacher’s safety briefing.
Method:
Add 1 cm³ chlorine solution to 1 cm³ of each potassium halide in separate test tubes.
Stopper and shake each tube. Record colour changes.
Add 1 cm³ hexane, stopper again and shake. Record the upper hexane layer colour.
Repeat with bromine and iodine solutions.
Add starch solution to test tubes that may contain iodine.
Colours of the halogens in hexane:
Chlorine – pale green
Bromine – orange
Iodine – purple
Test for iodine:
Add starch – solution turns blue-black if iodine is present.
Results:
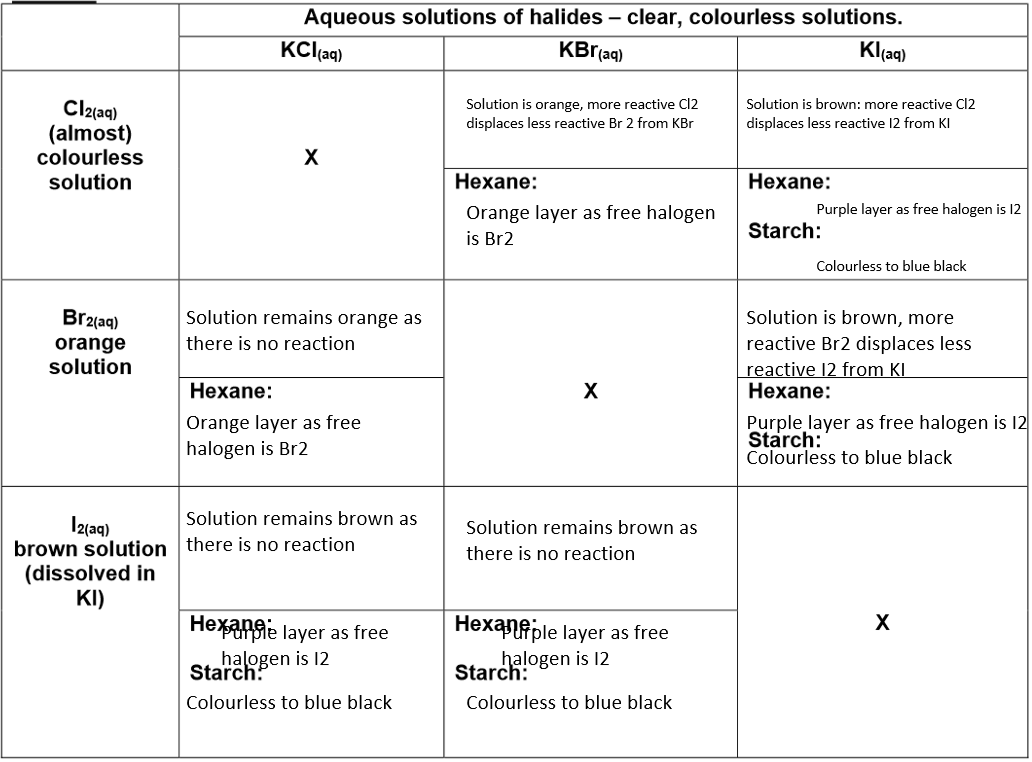
Displacement Reactions Involving Halogens
Halogen reactivity decreases down Group 7: F > Cl > Br > I
General equation:
More reactive halogen + less reactive halide → more reactive halide + less reactive halogen
1) Chlorine + Potassium Bromide
If chlorine solution (almost colourless) is added to potassium bromide (colourless), the solution turns orange-brown as bromine forms.
Chlorine displaces less reactive bromine.
Word equation: chlorine + potassium bromide → potassium chloride + bromine
Chemical equation: Cl₂ + 2KBr → 2KCl + Br₂
Ionic equation: Cl₂ + 2Br⁻ → 2Cl⁻ + Br₂
2) Chlorine + Potassium Iodide
If chlorine is added to potassium iodide, the solution turns brown as iodine forms.
Chlorine displaces less reactive iodine.
Word equation: chlorine + potassium iodide → potassium chloride + iodine
Chemical equation: Cl₂ + 2KI → 2KCl + I₂
Ionic equation: Cl₂ + 2I⁻ → 2Cl⁻ + I₂
3) Bromine + Potassium Iodide
If orange bromine is added to potassium iodide (colourless), the solution turns brown as iodine forms.
Bromine displaces less reactive iodine.
Word equation: bromine + potassium iodide → potassium bromide + iodine
Chemical equation: Br₂ + 2KI → 2KBr + I₂
Ionic equation: Br₂ + 2I⁻ → 2Br⁻ + I₂
Reactions of Group 7 elements with hydrogen:
Halogens react with hydrogen to form hydrogen halides

Properties of hydrogen halides:
colourless gas which is toxic
very soluble in water
Forming solutions containing H+ ions
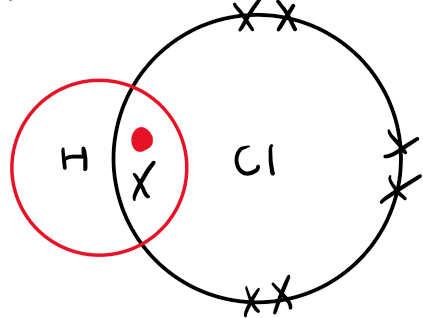
Dot and cross representation HCL:
Preparation of hydrogen chloride gas in the laboratory THE FOUNTAIN EXPERIMENT:
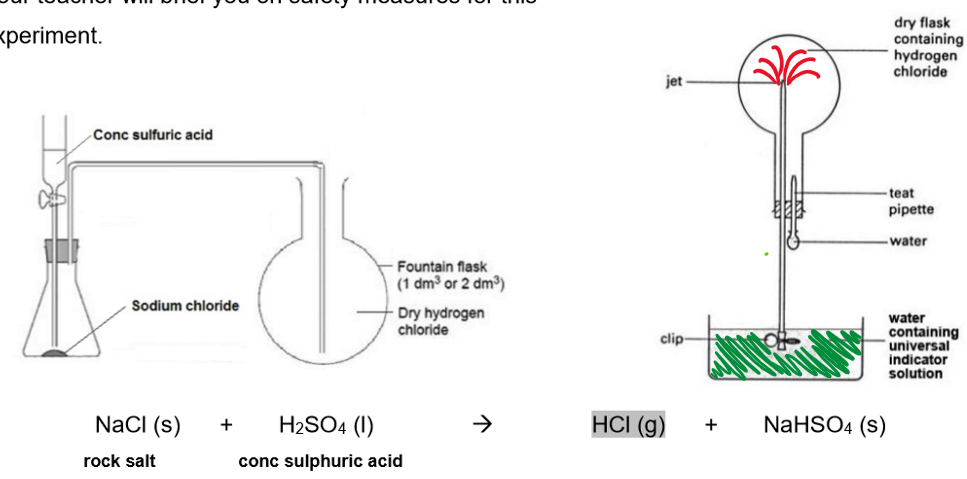
Method:
A small amount of water is injected into an upturned round-bottom flask filled with HCl gas. The pressure drops, causing water to rise from the trough into the delivery tube and flask.
Observations:
When the water (with universal indicator) enters the flask:
HCL gas dissolved in it
The solution formed is acidic
Universal indicator (UI) turns from green to red
Acidic Behaviour of HCl in Water:
HCl gas is covalent and doesn't show acidic properties on its own.
In water (a polar solvent), HCl ionises to form H⁺ and Cl⁻ ions, making the solution acidic.
In methylbenzene (a non-polar solvent), HCl does not ionise, so no acidic behaviour is observed.
Comparison Table:
Property of HCl gas | HCl in Water | HCl in Methylbenzene (Toluene) |
|---|---|---|
Dry blue litmus / UI solution | Turns red (acidic) | No colour change |
Mg ribbon | Fizzes (H₂ gas formed) | No reaction |
Limestone / CaCO₃ | Fizzes (CO₂ formed) | No reaction |
What do these results show?
Hydrogen chloride gas is a covalently bonded molecule. When added to water, the H-Cl bond breaks to form H⁺ and Cl⁻ ions. The H⁺ ions cause the acidity, and the resulting solution is called hydrochloric acid.
When added to methyl benzene or other organic liquids, the H-Cl bond does not break to form H⁺ and Cl⁻ ions, so it doesn’t behave as an acid.
Write equations for the reaction of HCl in water with:
i) Magnesium (Mg):
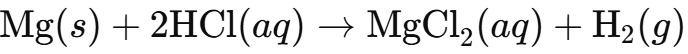
ii) Calcium Carbonate (CaCO₃):
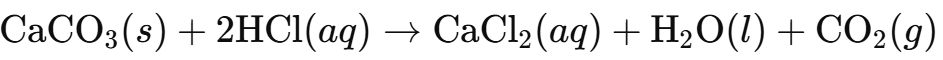
Tests for halides (precipitation of silver halides):
Method
1. Add 1cm3 of potassium halide solution to a test tube.
2. Add 1cm3 of nitric acid, HNO3(aq), and shake.
3. Add silver nitrate solution, AgNO3(aq), dropwise, with shaking, until you see a change.
Results:
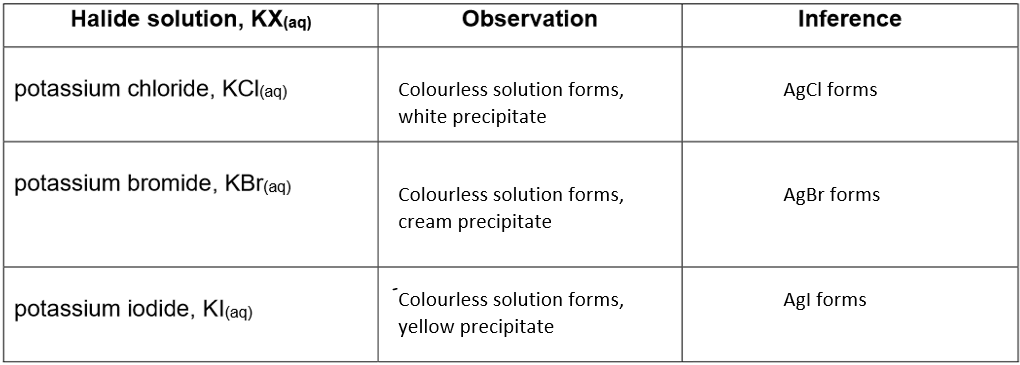
Conclusion
The halides formed can be identified through specific tests that detect the presence of chloride, bromide, or iodide ions.
The halides can be identified from the results of these tests, which typically involve the addition of silver nitrate solution to the sample.
1) Test for chloride (Cl⁻)
Word equation:
Silver nitrate + Chloride → Silver chloride + Nitric acid
Chemical equation:
AgNO₃ (aq) + Cl⁻ (aq) → AgCl (s) + NO₃⁻ (aq)
Ionic equation:
Ag⁺ (aq) + Cl⁻ (aq) → AgCl (s)
2) Test for bromide (Br⁻)
Word equation:
Silver nitrate + Bromide → Silver bromide + Nitric acid
Chemical equation:
AgNO₃ (aq) + Br⁻ (aq) → AgBr (s) + NO₃⁻ (aq)
Ionic equation:
Ag⁺ (aq) + Br⁻ (aq) → AgBr (s)
3) Test for iodide (I⁻)
Word equation:
Silver nitrate + Iodide → Silver iodide + Nitric acid
Chemical equation:
AgNO₃ (aq) + I⁻ (aq) → AgI (s) + NO₃⁻ (aq)
Ionic equation:
Ag⁺ (aq) + I⁻ (aq) → AgI (s)
SILVER PHOTOGRAPH EXPERIMENT
Silver halides are reduced to silver by light and are therefore used in photographic paper and film.
A piece of filter paper is soaked in silver nitrate solution and then potassium chloride solution is added to it. A white precipitate of silver chloride (AgCl) is formed on the paper.
A shape is cut out on a piece of card, placed on the filter paper, and left exposed to light. The exposed part of the filter paper turns silvery grey due to the formation of silver.
Chemical equation:
AgNO₃ (aq) + KCl (aq) → AgCl (s) + KNO₃ (aq)
Ionic equation:
Ag⁺ (aq) + Cl⁻ (aq) → AgCl (s)
The precipitate, AgCl(s), decomposes in sunlight causing a change in colour as silver (grey solid) is formed.

Uses of chlorine:
Sterilising drinking water and swimming pools
Manufacture of bleach
Manufacture of hydrochloric acid

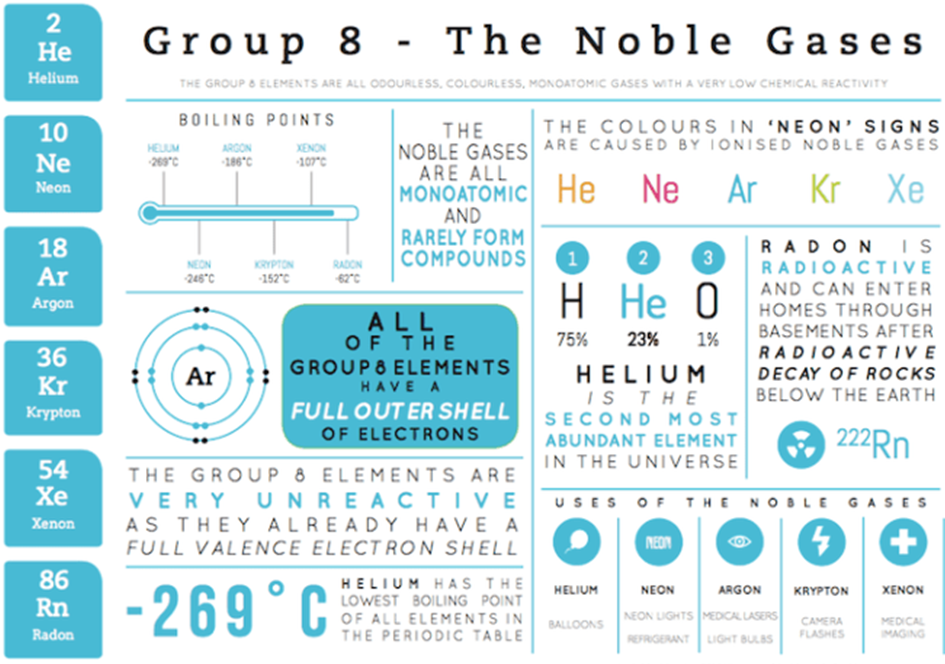
Group 0 - The Noble Gases
Properties:
All inert gases at room temperature with low melting and boiling points.
Density increases down the group as atomic mass increases.
Inert with a full outer shell of electrons, so they don’t form bonds.
Exist as monatomic gases.
M.p.s and b.p.s increase down the group due to stronger interatomic forces.
Uses:
Helium – Weather balloons and airships because it’s light and non-flammable.
Neon – Used in red advertising signs.
Argon – Used as an inert gas in light bulbs, preventing reactions with the filament.
Krypton – Used in lasers.
Xenon – Used in car headlights.
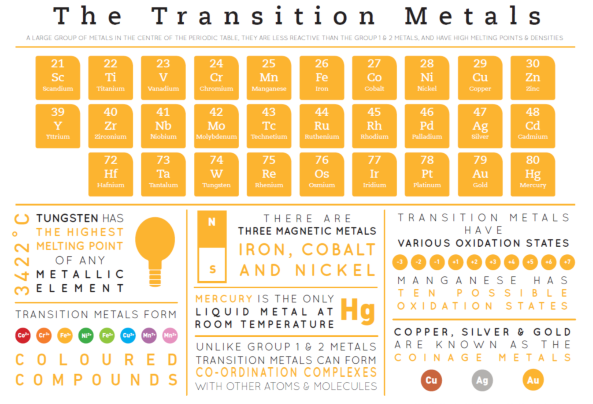
TRANSITION METALS
Location in the Periodic Table:
The transition metals are located in the d-block of the Periodic Table, between Group 2 and Group 3.
Properties:
High melting and boiling points.
Good electrical and thermal conductors.
Malleable, ductile, shiny, and sonorous.
Less reactive than Group I and II metals.
Reaction with Water:
Na-floats; moves on the surface of the water; effervesces, melts into a ball; reacts to form a colourless solution
Cu-metal sinks; no observable reaction
Ion Formation:
Transition metals can form more than one type of ion, depending on the reaction conditions.
Example: Fe²⁺ and Fe³⁺.
Coloured Compounds:
Transition metals form coloured compounds.
Examples: Copper(II) sulfate (blue), Iron(III) chloride (yellow).
Catalysts:
Fe in making ammonia (Haber process - used to make ammonia from nitrogen and from hydrogen)
V2O5 in manufacture in sulfuric acid (vanadium pentoxide)
Pt and Rh in catalytic converters
Ni in manufacture of margarine
MnO2 in decomposition of hydrogen peroxide (manganese (IIII) oxide)