Ch9b Periodic Properties of the Elements
Electron Configuration Irregularities
Description of electron spins:
Methodology of representing electron spins using tables.
Emphasis on the necessity of familiarity with the methodology for accurate interpretation.
Irregular Electron Configurations:
The phenomenon of irregular configurations arises due to close energy levels of the 4s and 3d orbitals.
Energy effects can significantly influence electron arrangement.
Certain transition metals exhibit irregular electron configurations where:
The nth orbitals may become partially filled before the (n-1)th.
These configurations are derived from experimental observation rather than prediction.
Notable Exceptions:
The first documented irregularity is seen in the case of chromium
Electron configuration norms according to established rules:
For chromium, the predicted configuration is 4s² 3d^4 according to the Aufbau principle.
However, the actual observed configuration is 4s^1 3d^5 and achieves half-filled d orbitals.
Significance of Half-Filled and Fully-Filled Orbitals:
The stability of half-filled or fully-filled configurations is attributed to enhanced electron spin maximization.
Example with copper, normally predicted as 3d^9, actual configuration is 3d^10 similarly achieves stability.
This stability arises from energy differences which favor these configurations over the partially filled orbitals, as elements prefer to achieve maximum spin
Listing of Transition Metals with Irregular Configurations:
Transition metals exhibiting these phenoms include:
Chromium, Molybdenum, Tungsten, Niobium, Copper, Silver, Gold, Platinum,
Copper, Silver, Gold (exhibits preference for fully filled d orbitals).

Additional Observations:
Palladium displays a unique phenomenon where it opts for an electron configuration of 4d^10 5s^0 instead of expected fill.
Rule of thumb: Half-filled and fully filled orbitals are more favorable.
Properties of Elements on the Periodic Table
The properties of the elements follow a periodic pattern.
Elements in the same column have similar chemical properties.
The elements in a period show a pattern that repeats.
Elements with electron configurations closest to noble gases are most reactive.
Valence Electrons and Electron Configuration Patterns:
Elements in the same group exhibit identical valence electron counts leading to similar chemical properties.
Example configurations:
Alkali metals:
Lithium: 2s^1, Sodium: 3s^1, Potassium: 4s^1, Rubidium: 5s^1
Noble Gases and Stability:
Noble gases maintain stable configurations with full electron shells (e.g., Helium = 2, the rest = 8). Krypton = 18 when incorporating d-electrons.
The stability of noble gases contributes to their low reactivity.
Reactivity and Ion Formation:
Sodium's (ALKALI METAL) electron loss leads to the formation of a Na+ ion, gaining a stable electron configuration like neon.
Chlorine (HALOGEN) gains an electron to form Cl- achieving a configuration of Argon.
Insights into formation of anions and cations based on periodicity and inherent charge predictability.
These atoms form ions that will result in an electron configuration that is the same as the nearest noble gas.
Atomic Size and Trends
Concept of Atomic Size:
Atoms have measurable sizes determined by radius.
Application of Heisenberg's uncertainty principle indicates the necessity of indirect measurement strategies.
Simplified measurement method: measure nuclear distances post-crystallization and divide by 2 to approximate atomic radii.
Measurement Techniques:
Atomic radii can be determined through various methods:
Nonbonding interactions via crystal structures yielding bulk radius measurements (Van der Waals)
Covalent radius measurements through bond lengths in diatomic molecules (Covalent bonding radius)
Atomic radius is an average radius of an atom based on measuring large numbers of elements and compounds.
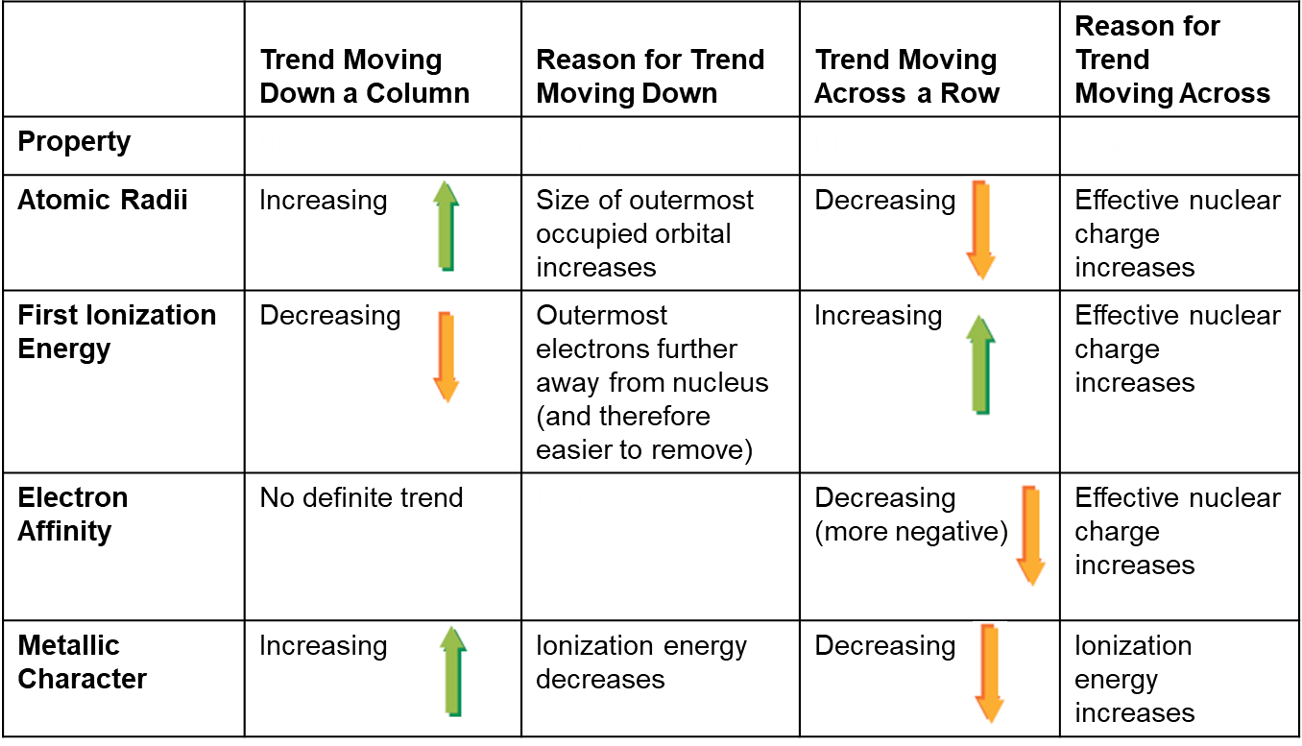
Trends in Atomic Radius:
Observational trends when plotted with atomic number indicate:
Atomic size increases down a group in the periodic table. As we move down a column in the periodic table, the principal quantum number (n) of the electrons in the outermost principal energy level increases, resulting in larger orbitals and therefore larger atomic radii.
Atomic size decreases across a period from left to right. As we move to the right across a row in the periodic table, the effective nuclear charge (Zeff) experienced by the electrons in the outermost principal energy level increases, resulting in a stronger attraction between the outermost electrons and the nucleus, and smaller atomic radii.
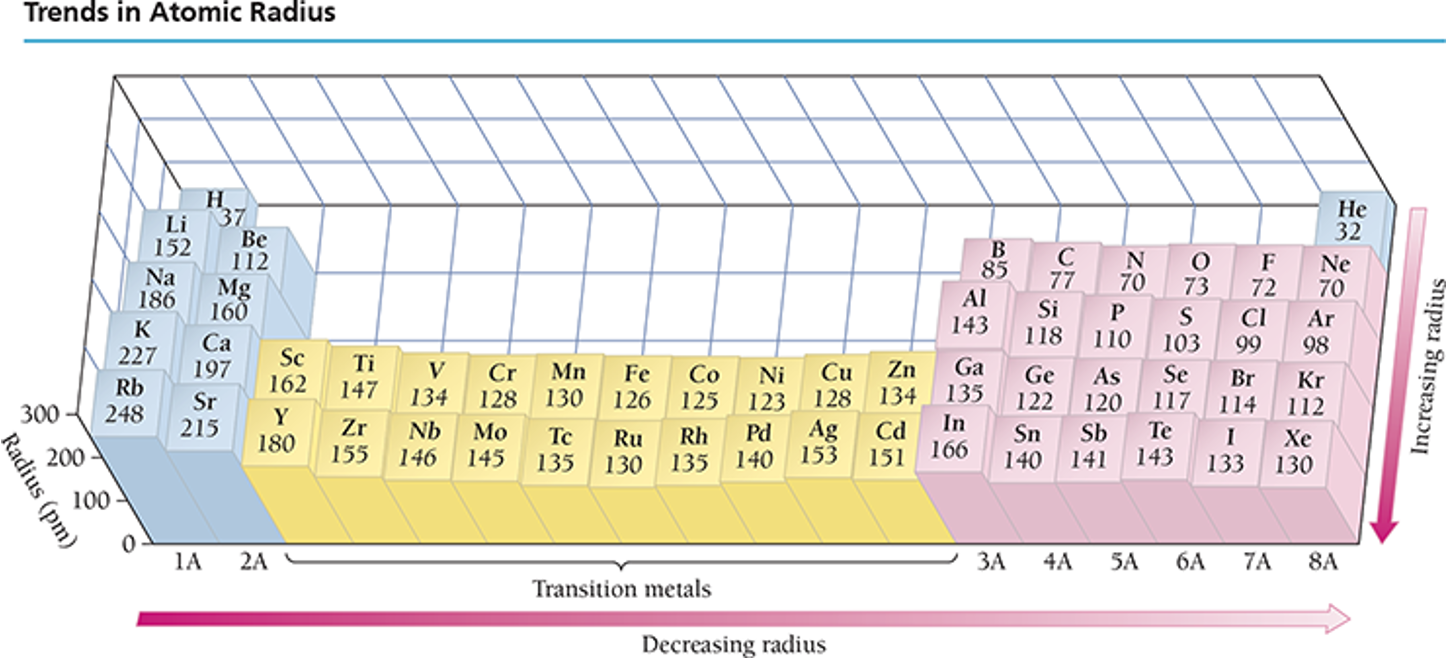
Group Trends and Periodic Trends:
Groups show increasing atomic sizes:
Example: Rubidium as the largest in alkali metal group (Elemental context: K > Na > Li).
Periods exhibit decreasing atomic sizes from left to right:
Example: Lithium to Neon.
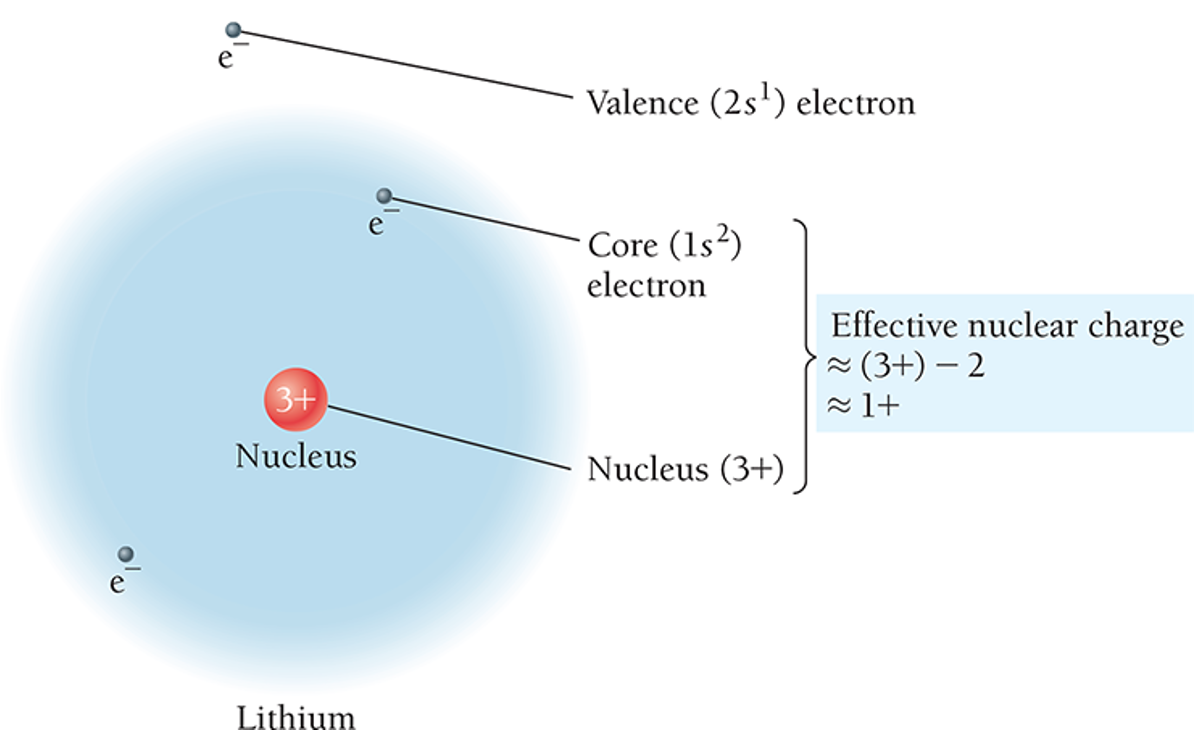
Effective Nuclear Charge
Defined as the net positive charge experienced by valence electrons.
Calculated using the atomic number (number of protons) minus the number of electrons in the lower energy levels (shielding effect).
Zeff = Z - S
Z is the nuclear charge, and S is the number of electrons in lower energy levels.
Variation in resulting effective charge influences atomic behavior in multi-electron systems where electrons repel one another while simultaneously being attracted to the nucleus.
Example with Lithium:
Configuration of Lithium: 1s^1 2s^1 results in 2 core electrons shielding the valence electron.
Effective nuclear charge felt by the valence electron is calculated as:
Meaning this valence electron experiences an effective charge of +1 leading to notable atomic behavior.
Cation/Anion Electron Configurations
Cations form when the atom loses electrons from the valence shell.
Al atom = 1s22s22p63s23p1 or [Ne] 3s23p1
Al3+ ion = 1s22s22p63s23p1
Anions are formed when nonmetal atoms gain enough electrons to have eight valence electrons.
Filling the s and p sublevels of the valence shell
S atom = 1s22s22p63s23p4 or [Ne] 3s23p4
S2- ion = 1s22s22p63s23p6 or [Ne]
When transition metals form cations, the first electrons removed are the S valence electrons, even though other electrons were added after.
Electrons may also be removed from the sublevel closest to the valence shell after the S valence electrons
The iron atom has two valence electrons:
Fe atom = 1s22s22p63s23p64s23d6 or [Ar]4s23d6
Fe2+ ion = 1s22s22p63s23p63d6 or [Ar]3d6
Fe3+ ion = 1s22s22p63s23p63d5 or [Ar]3d5
Assigning Valence Electrons
For main group elements (block s and p)
Electrons in the largest n (s and p sublevels)
Example: Sr = [Kr] 5s2 valence electrons: 5s2 (2)
As = [Ar] 4s23d104p3 valence electrons: 4s24p3 (5)
For transition elements (block d and f)
Electrons in largest n (s and p) and (n-1)d
Example: Fe = [Ar] 4s23d6 valence electrons: 4s23d6 (8)
W = [Xe] 6s24f145d4 valence electrons: 6s25d4 (6)
Magnetic Properties
Electron configurations that result in unpaired electrons mean that the atom or ion will have a net magnetic field; this is called paramagnetism (para = “along” or “against”).•
Will be attracted to a magnetic field
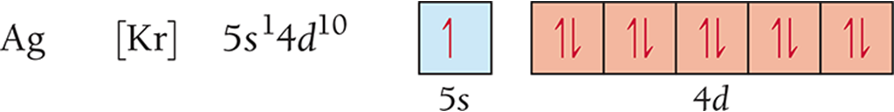
Electron configurations that result in all paired electrons mean that the atom or ion will have no magnetic field; this is called diamagnetism (dia = “through”).
Slightly repelled by a magnetic field
Ionic Radius
Ions in the same group have the same charge.
Ion size increases down the column.
Higher valence shell (n), larger ion
Cations are smaller than neutral atoms; anions are larger than neutral atoms.
Cations are smaller than anions.
Except Rb+ and Cs+, bigger or same size as F− and O2-
Larger positive charge = less electrons = smaller cation
For isoelectronic species
Isoelectronic = same electron configuration
Larger negative charge = more electrons = larger anion
For isoelectronic species
CATIONS
When atoms form cations, the valence electrons are removed.
The farthest electrons from the nucleus are the p or d electrons in the (n − 1) energy level.
This results in the cation being smaller than the atom.
These “new valence electrons” also experience a larger effective nuclear charge than the “old valence electrons,” shrinking the ion even more.
Traversing down a group increases the (n − 1) level, causing the cations to get larger.
Traversing to the right across a period increases the effective nuclear charge for isoelectronic cations, causing the cations to get smaller.
ANIONS
When atoms form anions, electrons are added to the valence shell.
These “new valence electrons” experience a smaller effective nuclear charge than the “old valence electrons,” increasing the size.
The result is that the anion is larger than the atom.
Traversing down a group increases the n level, causing the anions to get larger.
Traversing to the right across a period increases the effective nuclear charge for isoelectronic anions, causing the anions to get smaller.
Trend | Explanation | Applies to |
|---|---|---|
Down a group → radius ↑ | More shells (n increases) | Atoms, cations, anions |
Across a period → radius ↓ | Zeff increases, pulls e⁻ tighter | Atoms, cations, anions |
Neutral → anion | Radius ↑ | Gains e⁻, more repulsion |
Neutral → cation | Radius ↓ | Loses e⁻, less repulsion |
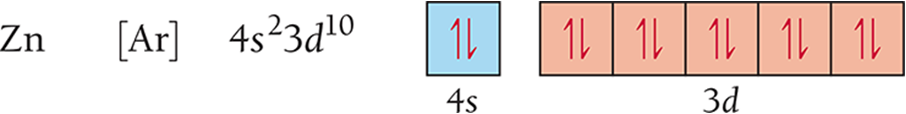
Ionization Energy (IE)
Definition: Energy required to remove an electron from an atom or ion.
Minimum energy needed to remove an electron from an atom or ion
Gas state
Endothermic process
Valence electron easiest to remove, lowest IE
M(g) → M1+(g) + 1e- IE1
M1+(g) → M2+(g) + 1e- IE2
First ionization energy IE = energy to remove electron from neutral atom
Second IE = energy to remove electron from 1+ ion, etc.
Trends:
Generally increases across a period (left to right).
Generally decreases down a group (top to bottom).
Example: Ionization energy of helium is the highest, while cesium has the lowest.
The larger the effective nuclear charge on the electron, the more energy it takes to remove it.
The farther the most probable distance the electron is from the nucleus, the less energy it takes to remove it.
First IE decreases down the group.
Valence electron farther from nucleus
First IE generally increases across the period.
Effective nuclear charge increases
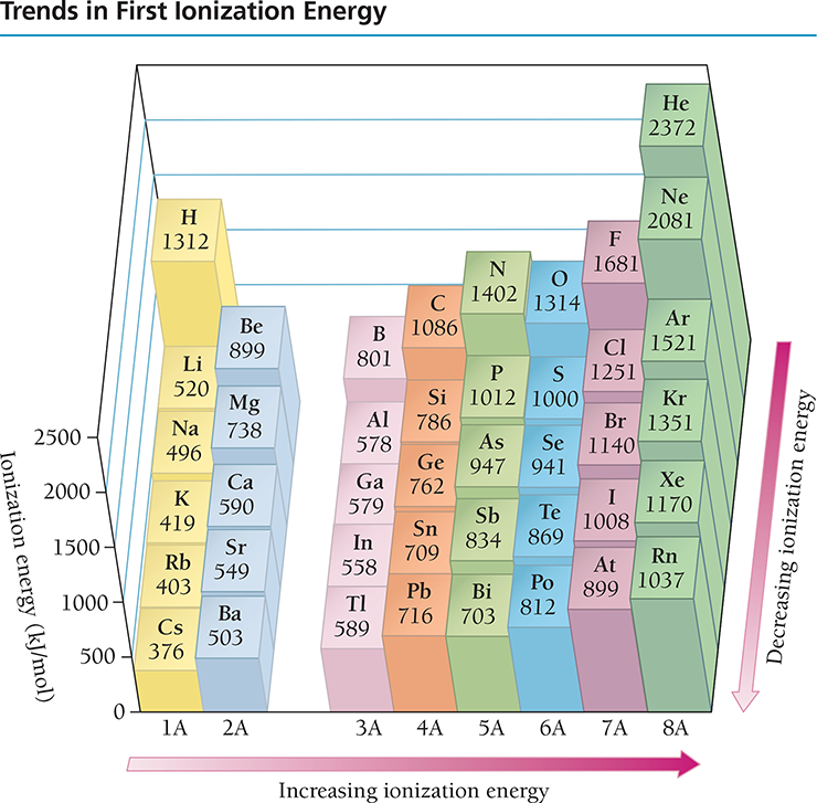
SUCCESSIVE IONIZATION ENERGY
Removal of each successive electron costs more energy.
Shrinkage in size due to having more protons than electrons
Outer electrons closer to the nucleus; therefore harder to remove
There’s a regular increase in energy for each successive valence electron.
There’s a large increase in energy when core electrons are removed.
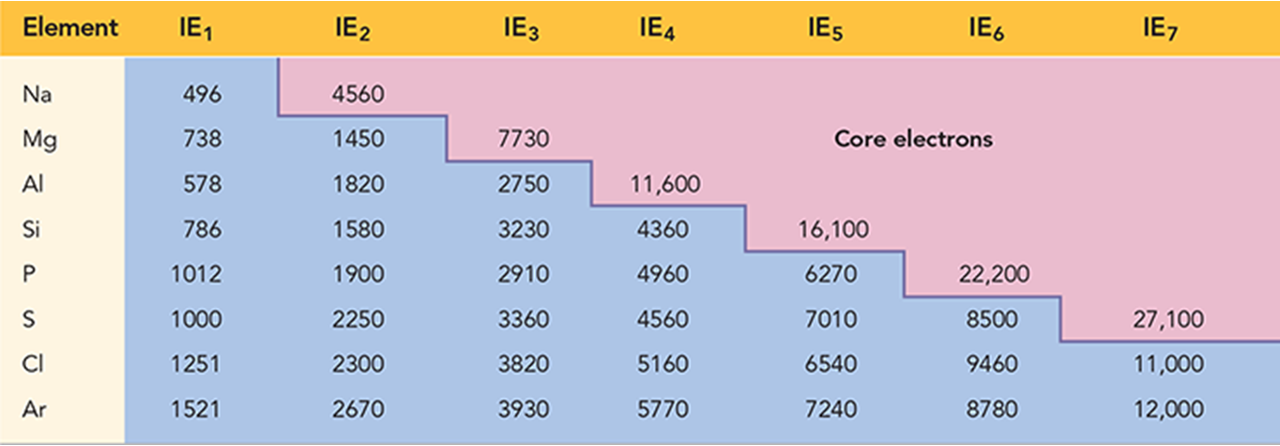
Electron Affinity (EA)
Energy is released when a neutral atom gains an electron.
Gas state
M(g) + 1e- à M1-(g) EA
Electron affinity is defined as exothermic (−) but may actually be endothermic (+).
Electron affinity of some alkali earth metals and all noble gases are endothermic.
The more energy that is released, the larger the electron affinity, the more stable the anion.
The more negative the number, the larger the EA
General Trends
Across a period → same distance, stronger magnet (more protons) → pulls electrons hard → more negative EA.
Down a group → magnet gets “further away” (new shells), feels more shielded → pulls added electron less → less negative EA.
Exceptions
Alkali metals decrease electron affinity down the column.
But not all groups do
Generally irregular increase in EA from second period to third period (mainly nonmetals groups 15-17)
This irregularity comes from:
Electron–electron repulsion in smaller 2nd-period atoms (less room in small orbitals → incoming electron repelled).
Larger 3rd-period atoms have more space in their orbitals → added electron experiences slightly less repulsion → EA can appear more negative than expected.
EA generally” increases across period
Becomes more negative from left to right
Not absolute
Highest EA in any period = halogen
Group 15 (Nitrogen family: N, P, As…)
Observation: EA is lower (less negative) than expected.
Reason:
Group 15 elements have half-filled p orbitals (ns² np³).
Example: N = 1s² 2s² 2p³ → three unpaired electrons in p orbitals.
Adding an extra electron means it must pair with an existing electron in one of the p orbitals.
Pairing electrons in the same orbital costs extra energy due to electron–electron repulsion.
✅ Result: EA is less negative → the atom is less eager to gain an electron than the trend across the period would suggest.
2⃣ Group 2A (Alkaline earth metals)
Observation: EA is very low (slightly positive or small negative).
Reason:
Group 2A elements have a full s subshell (ns²).
To gain an extra electron, it must go into the next higher energy p orbital (a new sublevel).
Adding an electron to a new higher-energy level is not very favorable energetically.
✅ Result: EA is small or even positive → the atom doesn’t “want” an extra electron.
3⃣ Group 8A (Noble gases)
Observation: EA is also very low (usually positive).
Reason:
Noble gases have full valence shells (ns² np⁶).
Adding an extra electron forces it into a new orbital of the next energy level, far from the nucleus.
Strong electron–electron repulsion in the full shell resists the extra electron.
✅ Result: EA is very small or positive → extremely low tendency to gain an electron.
Metallic Character
Metallic character is how closely an element’s properties match the ideal properties of a metal.
More malleable and ductile, better conductors, and easier to ionize
Metallic character decreases left to right across a period.
Metals found at the left of the period, and nonmetals to the right
Metallic character increases down the column.
Nonmetals found at the top of the middle main-group elements, and metals found at the bottom
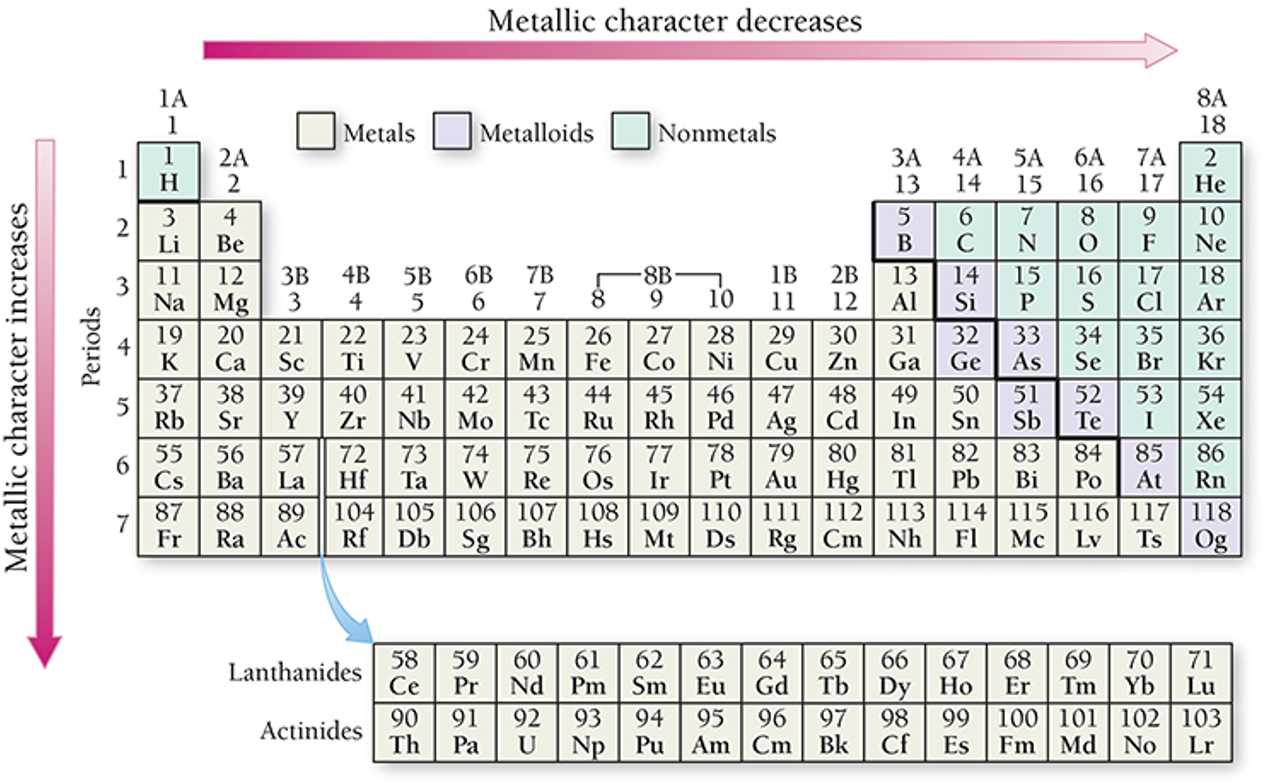
Metals generally have smaller first ionization energies, and nonmetals generally have larger electron affinities.
Except for the noble gases
Quantum mechanics predicts the atom’s metallic character should increase down a column because the valence electrons are not held as strongly.
Quantum mechanics predicts the atom’s metallic character should decrease across a period because the valence electrons are held more strongly and the electron affinity increases.
Periodic Trends Summary