How are Anions & Cations Identified?
How are Anions & Cations Identified
Lab Handout:
Materials within the Group (lab bench) Kit
Aluminum Granules
In 125 mL Dropper Bottles
0.2 M Oxalic Acid
0.2 M Potassium Nitrate
0.2 M Potassium Sulfate
0.1 Silver Nitrate
0.2 M Barium Nitrate
0.2 M Iron (III) Nitrate
0.2 M Nickel (II) Nitrate
Saturated Barium Hydroxide
6 M Sodium Hydroxide
6 M Ammonia
3 M Sulfuric Acid
6 M Hydrochloric Acid
6 M Nitric Acid
1 M Lead (II) Nitrate
1% Dimethyl Glyoxime
0.1 M Potassium Ferrocyanide
1 vial of Red Litmus Paper
1 vial of Blue Litmus Paper
24 well plates
Materials within TA bin
In 125 mL Dropper Bottles
1 of the Unknown A
1 of the Unknown B
1 of the Unknown C
1 of the Unknown D
1 of the Unknown E
1 of the Unknown F
Common Group Glassware Drawers:
28+ test tubes
2 test tube racks
Drawers in Front of Room:
21+disposable plastic pipets
Well plates
Introduction:
Cations (positive ions) and anions (negative ions) make ionic compounds, known as salts (some examples include, Ca(NO3)2, AgNO3, NaI, and Na2CO3). Some salts have high solubility, while others are low. Solubility is the maximum amount of the substance that will dissolve in in a certain quantity of solvent at a given temperature to form a saturated solution.
Ions from soluble salts separate from each other when dissolved in water and their properties can be studied separately. Below is an example of two aqueous (soluble in water) salts being mixed. The mixture of soluble potassium iodide and lead (II) nitrate results in the formation of a solid precipitate, lead (II) iodide.
lead (II) nitrate (soluble) + potassium iodide (soluble) → lead (II) iodide (insoluble) + potassium nitrate (soluble) molecular equation: Pb(NO3)2(aq) + 2KI(aq) → PbI2(s) + 2KNO3(aq)
ionic equation: Pb2+(aq) + 2NO3-(aq) + 2K+(aq) + 2I-(aq) → PbI2(s) + 2K+(aq) + 2NO3-(aq)
net ionic equation: Pb2+(aq) + 2I-(aq) → PbI2(s)
The (s) in the equations indicates formation of a solid precipitate. The net ionic equation has the advantage of emphasizing the fact that any (aq) Pb2+ salt will react with any soluble iodide to result in the precipitate. Solubility guidelines for common ionic compounds in water are available at the end of this handout or the textbook in chapter 4.
Qualitative analysis involves the identification of ionic species based on the species’ unique reactivity or solubility. But what happens when you have a mixture of ionic species and you wish to separate and identify each ion in the mixture? When this is the goal, a flow chart can be developed to selectively separate and confirm the identity the ions in a mixture of ions. The solubility of the ions of interest when introduced to other ions can produce recognizable products. Thus, by selective precipitation or reaction, the identity of the ions present can be confirmed.
These reactions might involve formation of gases, so if the identity of the gas can be confirmed, the ion from which it originated can be confirmed present. There are other confirmatory tests that can be performed that also yield uniquely colored complex ions that are used as well for separation and/or confirmation. The formation of precipitates based on solubility rules is commonly used for ion confirmation as well. Precipitation (double-displacement) reactions are useful in the identification of ions because the precipitates vary in color and texture. For example, cations (Ag+ and Pb2+) can be isolated as chloride precipitates by using a hydrochloric acid solution with low concentration. Dissolving precipitates with a reagent can also be used for identification. For example, copper (II) hydroxide can be dissolved by concentrated ammonia, NH3. However cobalt (II) hydroxide will not dissolve using ammonia, so with this difference, the precipitates may be identified.
Most labs depend on quantitative techniques, where determining specific quantities or concentrations are essential to achieve the principal goals of the experiment. Qualitative analysis of cations and anions by chemical reactions involves the identification of these ions based on qualitative (visual) means. Knowing the amount or concentration of ion present is not required when confirming its identity is the only goal of the experiment. Another goal of this experiment is the systematic separation and identification of each individual cation and anion. These ions can be isolated from one another and identified by the various means described above by following a flow chart. Cations and anions are initially separated into small groups by adding appropriate reagents which will precipitate, form complex ions, form gases, etc, resulting in one ion uniquely identified by its product at the terminus of each branch. If a gas is formed, it must be collected or identified by a second reagent. If a solid, the product can be separated by filtration or more typically, by centrifugation, which accelerates the settling of a solid suspended in a liquid. The remaining liquid above the precipitation is referred to as the supernatant. The liquid can then be decanted, which is to gradually pour liquid, without disturbing the precipitate, into a different container. The remaining precipitate or supernatant can be tested by further means.
In this investigation, there will be two sets of ions that you will use a series of chemical reactions to separate and identify.
Anion Set Cation Set |
0.2 M H2C2O4 0.2 M KNO3 0.2 M K2SO4 0.1 M AgNO3 0.2 M Ba(NO3)2 0.2 M Fe(NO3)3 0.2 M Ni(NO3)2 |
You will eventually be performing qualitative analysis tests on an unknown solution that can contain one or more of the ions simultaneously with the known mixture, so you can compare the results and determine which ion(s) are present in your group’s unknown. The “unknown” solution would contain ions at approximately the same concentrations present as your known mixture used in your flowchart testing.
First, the individual reaction of each ion of interest with the reagents provided must be analyzed. This type of individual analysis can be done in test tubes or in well plates to keep volumes small. Based upon the reactions noted, a flowchart for selectively separating the ions can be developed. Refer to internet resources like http://www.public.asu.edu/~jpbirk/ for the properties of some metals and nonmetals as you identify products and write your net ionic reactions for your flow charts. After a working flow chart is determined using a known solution with all ions of interest, an unknown solution containing a combination of one or more of the original ions can be analyzed and the ions present (and absent) can be identified.

Objectives upon completion of this section:
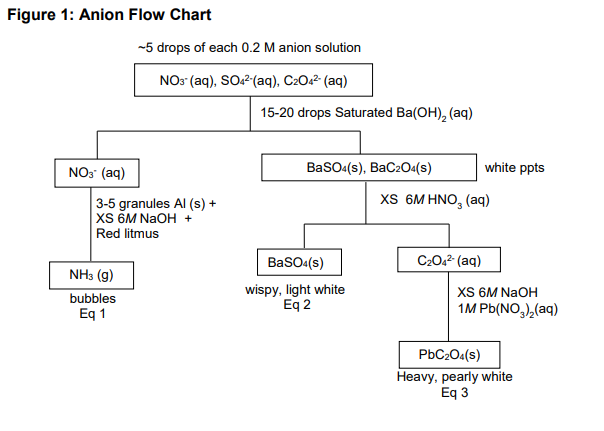
Analyze the flow chart (Figure 1) that will be followed to separate a mixture of all three anions of interest. Follow the recommended procedures to allow you to recognize the presence of each of these ions. Record all procedures (volumes and concentrations of added reagents, separation and gas collection procedures, etc) and all observations for each step of the analysis in your lab notebook.
Consult valid references to be able to explain and write balanced net ionic reactions for each of the reactions that occur in the flow chart.
Safety: NaOH, and Ba(OH)2, are highly corrosive. H2C2O4, K2SO4, and KNO3 are toxic. You must wear personal protective equipment at all times.
Before you come to lab, write the following tables and figures into your notes and write a rough proposal using the procedural considerations below.
Take the following procedural considerations into account for proposal writing:
During the course of the experiment, will you record any observations or sources of potential error you notice in your lab notebook?
Figure 1: Anion Flow Chart

Procedure Considerations:
You will be testing a qualitative analysis scheme to separate and confirm the identity of three anions in a mixture using the suggested flow chart (Figure 1). As you write your proposal, consider the specific procedural considerations provided below.
Make the known mixture:
How much of each anion solution should you place in your mixture? Record observations.
Initial reaction of the flow chart:
What should you add to the mixture to separate the insoluble barium salts from the nitrate? How many drops will you add? Record observations.
What separating technique can be used to quickly separate the solid from the supernatant? Record observations.
Before decanting the supernatant, add 2-3 additional drops of reagent. How will you know if the reaction was complete? If not complete, add more reagent and repeat the previous step.
If all of the barium salts are successfully precipitated, decant the supernatant into an empty test tube.
Testing of the supernatant:
What is the only anion that should be present in the supernatant? Should any significant amount of the other two ions be present in this solution?
What should you add to the test tube with the supernatant to produce the gas of interest? What gas is supposed to be produced? What ion would be confirmed present?
How much aluminum should be added? Was the solution required to be acidic or basic?
Develop a technique to plug the test tube and suspend the wet red litmus paper above the test tube without allowing it to touch the liquid or any liquid that may be on the side of the test tube. Is the gas of interest an acid or a base? What should happen to the wet red litmus paper when it comes into contact with the gas of interest?
Use your considerations above to determine your reagent volumes and gas detection setup. Record all observations.
Testing of the Precipitate:
What two anion salts are present in the precipitate?
According to the flow chart (Figure 1), which precipitate dissolves in acidic solution? How will you know you have added enough acid? Will you record how many drops you added and what the solution looks like when you stop?
What separating technique can be used to quickly separate the remaining solid from the supernatant? Record observations.
What anion have you successfully separated in the solid phase? What anion is present in aqueous solution in the supernatant?
Final Step:
When performing the final precipitation, what must the pH be? Under what pH condition is the solution currently? How will you know you have successfully neutralized the solution and are in excess at the desired pH? (Keep in mind you should already know how much of each reagent you have already added and have litmus or pH paper as well.)
Once your conditions are correct, how much reagent will you add to completely precipitate the final product?
What separating technique can be used to quickly separate the solid from the supernatant? Record observations.
Add 2-3 additional drops of reagent. How will you know if the reaction was complete? If not complete, add more reagent and repeat the previous step.
At this point, three separate test tubes should contain a unique precipitate or confirmatory test result for each of the three anions originally present. If you haven’t already, make sure to record your observed results for each step for the selective precipitation of each ion.
Before you Proceed to the Next Section:
Write net ionic equations for all confirmatory tests and precipitates formed (as indicated in Figure 1).
Confirm your results with your TA: Identify the precipitate or substance present for a confirmatory test for each of the test tubes.
Discuss your answer to the following question with your TA (or group, if you’re TA is not readily available): What would be the same and what would be different if you ran the flow chart with one or more of the anions absent? How could you identify the anions present in a mixture if the mixture only contained 1 or 2 of the original 3 ions?
Clean up and Waste Collection:
NaOH, and Ba(OH)2, are highly corrosive. H2C2O4, K2SO4, and KNO3 are toxic. Please use paper towel to clean up any spills immediately. Any spills left behind might result in points being deducted (at the discretion of your instructor).
Rinse all solids from spot plate analysis and/or test tubes into your waste beaker before washing with soap and water.
Transfer all waste from waste beaker to waste bottle at the end of lab.
Rinse everything well with tap water followed by a quick distilled water rinse to prevent ion contamination for future stages of the experiment.
Objectives upon completion of this section:
Individual Analysis
Observe individual cation solutions when exposed to various reagents.
Identify products using solubility rules to predict and explain the reactions that occur.
Safety: HNO3, H2SO4, HCl, NaOH, and NH3OH, are highly corrosive. Hg2(NO3)2, Ba(NO3)2, Fe(NO3)3, Ni(NO3)2, DMG, K4[Fe(CN)6], and KSCN are toxic. You must wear personal protective equipment at all times.
Before you come to lab, write the following tables and figures into your notes and write a rough proposal using the procedural considerations below.
Table 1: Individual CATION Reactions
Cation in tt or spot plate→ Reagent added↓ 0.1 M Ag+ ~10 drops 0.2 M Ba2+ ~10 drops 0.2 M Fe3+ ~10 drops 0.2 M Ni2+ ~10 drops |
6M HCl Observe initially, at ~1-2 d & XS at ~10 drops |
3M H2SO4 Observe initially, at ~1-2 d & XS at ~10 drops |
6M NH3 Observe initially, at ~1-2 d & XS at ~10 drops |
Table 2: Individual CATION Confirmatory Tests
Cation Conditions (Basic) (Acidic) (Basic) (Acidic) |
(in spot plate or test tube) → 10 drops 10 drops 10 drops 10 drops |
0.2M Fe3+ 0.2M Fe3+ 0.2M Ni2+ 0.2M Ni2+ |
+ 10 drops + 10 drops + 10 drops + 10 drops |
Confirmatory Test Reagents 6M NH3 6M NH3 6M NH3 6M NH3 |
(add to individual cation) + 15 drops + 15 drops |
↓ 6M HNO3 6M HNO3 |
0.1M K4[Fe (CN)6] Observe initially, at ~1-2 d & XS at ~10 drops |
0.1M DMG Observe initially, at ~1-2 d & XS at ~10 drops |
Take the following procedural considerations into account for proposal writing:
During the course of the experiment, will you record any observations or sources of potential error you notice in your lab notebook?
You will be observing the reaction of individual cations using the suggested individual analysis tables (Tables 1 and 2).
Study the specific procedural considerations provided below.
Based on the results of this table, you will be developing a flow chart with a reaction scheme to allow for separation and confirmation of each ion.
Individual Cation Analysis Procedure Considerations:
You will be observing the reaction of individual ions with specific reagents using Tables 1 and 2 and the specific procedural considerations provided below.
Look at the volume requirements suggested in Tables 1 and 2. Would these tests be best performed in a spot plate or in test tubes?
The results of individual analysis will help with the identification of the same ions when mixed together. Will you make careful notations of solution color, precipitate color and texture, evolution of gas, etc?
For the Table 2 cations, is there a uniquely recognizable confirmatory test result that can be used to identify each ion in question? Will you carefully note the specific conditions under which the confirmatory test must be performed (e.g.: basic/acidic/neutral conditions, specific reagents use, specific procedure etc.)?
The quantities and concentrations of added reagents are important for future analysis. Will you carefully record all volumes and concentrations of cation and reagent solutions you use?
Before you Proceed to the Next Section:
Identify any precipitates, complex ions, or gaseous products that resulted from positive reactions or confirmatory tests.
Clean up and Waste Collection:
HNO3, H2SO4, HCl, NaOH, and NH3OH, are highly corrosive. Hg2(NO3)2, Ba(NO3)2, Fe(NO3)3, Ni(NO3)2, DMG, K4[Fe(CN)6], and KSCN are toxic. Please use paper towel to clean up any spills immediately. Any spills left behind might result in points being deducted (at the discretion of your instructor).
Rinse all solids from spot plate analysis and/or test tubes into your waste beaker before washing with soap and water.
Transfer all waste from waste beaker to waste bottle at the end of lab.
Rinse everything well with tap water followed by a quick distilled water rinse to prevent ion contamination for future stages of the experiment.
Clean your bench top. Put all equipment back exactly where you found it.

Objectives upon completion of this section:
Consider various separation techniques on the videos in CANVAS. If unsure, check with your TA prior to continuing.
Use the results of the individual analysis tables (Tables 1 and 2) to develop a flow chart for separation and identification of the four cations in the cation set.
This section may be completed in lab or between the first and second lab meetings.
Take the following considerations into account as you build your flow chart:
You will be developing a qualitative analysis scheme that confirms the presence or absence of each of the cations in your cation set.
What ions, reagents (and their concentrations and volumes) will you be using?
Which of the confirmatory test reagents will you use for the proposed cations?
Flow chart must be electronically generated.
All chemical species (ions, complex ions, precipitates, reagents) should be represented with the correct chemical formula. This includes charges, where applicable.
Identify the species in each box as solid (s) and aqueous (aq) as seen in Figure 1.
Each added reagent should also include its molarity.
For each precipitate or complex ion, include a brief description with its color.
Number each precipitate or complex ion; write a balanced net ionic reaction for each. There should be at least six balanced equations, each numbered corresponding to the number on the flow chart.
Don’t forget you have access to the website: http://www.public.asu.edu/~jpbirk/ (provided in the prelab) to confirm the observations you made from Part 2, and use it to check your net ionic equations!
Before you Proceed to the Next Section:
Your flow chart must be approved by your TA.

Objectives upon completion of this section:
Known Cation Mixture Analysis: Observe the results of the flow chart analysis to separate the 4 cations in a mixture by selective precipitation upon exposure to various reagents.
Unknown Cation Mixture Analysis: Identify which of the cations are present in an unknown mixture based on observations from unknown mixture flow chart analysis compared to individual analysis and known mixture flow chart results.
Safety: HNO3, H2SO4, HCl, NaOH, and NH3OH, are highly corrosive. Ag(NO3), Ba(NO3)2, Fe(NO3)3, Ni(NO3)2, DMG, K4[Fe(CN)6], and KSCN are toxic. You must wear personal protective equipment at all times.
Before you come to lab, write a rough proposal using the procedural considerations below and the flow chart you developed in Part 3.
Take the following procedural considerations into account for proposal writing:
During the course of the experiment, will you record any observations or sources of potential error you notice in your lab notebook?
Known Cation Mixture Analysis
How much of each cation solution should you place in each test tube?
Based on your approved flowchart:
What reagents will you use to separate each of the cations?
What order will they be added? What amounts will you use?
Will you make sure after any additions step that your solution is well mixed? What could happen if you did not completely mix your reagent and solution before moving on to the next step?
What separating technique can be used to quickly separate solids from the liquid?
What extra step makes sure each cation has been fully reacted/separated?
At each final precipitate or confirmatory test will you record any observations and compare them to the results of the individual analysis.
Show your TA the precipitates or confirmatory test results, one for each cation in your mixture, and provide the identity of each before collecting your group’s unknown.
Unknown Cation Mixture Analysis
Write down the unknown letter you are testing.
Will you follow the same basic procedural steps and reagent volumes as followed for your know analysis?
What would be different about your results if a cation was not present in the mixture?
Would you need to continue to add a reagent if there was no sign of an expected reaction? Would you need to perform a separation step if no solid formed?
Show your TA the precipitates or confirmatory test results, one for each cation in your mixture, and provide the identity of each that you can confirm as present in your group’s unknown. Confirm as well which cation or cations were not present in the unknown.
Clean up and Waste Collection:
HNO3, H2SO4, HCl, NaOH, and NH3OH, are highly corrosive. Ag(NO3), Ba(NO3)2, Fe(NO3)3, Ni(NO3)2, DMG, K4[Fe(CN)6], and KSCN are toxic. Please use paper towel to clean up any spills immediately. Any spills left behind might result in points being deducted (at the discretion of your instructor).
Rinse all solids from test tubes into your waste beaker before washing with soap and water.
Transfer all waste from waste beaker to waste bottle at the end of lab.
Rinse everything well with tap water followed by a quick distilled water rinse to prevent ion contamination for future stages of the experiment.
Clean your bench top. Put all equipment back exactly where you found it.
Before you Leave Checklist:
Write net ionic equations for all reactions that formed precipitates.
Confirm identity of the unknown with your TA.
Solubility Rules for Ionic Compounds
Substance Rules |
Na+, K+, NH4+, NO3-, CH3COO- Mainly water soluble. |
SO42- All sulfates are soluble. EXCEPT: CaSO4, SrSO4, BaSO4, PbSO4, Hg2SO4, and Ag2SO4 |
Cl-, Br-, I- All compounds containing chloride, bromide, and iodide are soluble. EXCEPT: AgX, Hg2X2, PbX2, HgBr2, and HgI2. PbCl2 is slightly insoluble. |
Ag+ AgNO3 is soluble, but all others containing Ag+ are not. |
OH- All hydroxides are insoluble. EXCEPT: Alkali metals, NH4OH, Ba(OH)2, Sr(OH)2, and Ca(OH)2 |
S2- All sulfides are insoluble. EXCEPT: Alkali & Alkaline metals and (NH4)2S |
CrO42- All ionic chromates are insoluble. EXCEPT: K2CrO4, Na2CrO4, (NH4)2CrO4, MgCrO4, CaCrO4, Al2(CrO4)3, NiCrO4 |
CO32-, PO43-, SO32-, SiO32- All compounds containing carbonate, phosphate, sulfite, and silicate are insoluble. EXCEPT: KX, NaX, and NH4X |