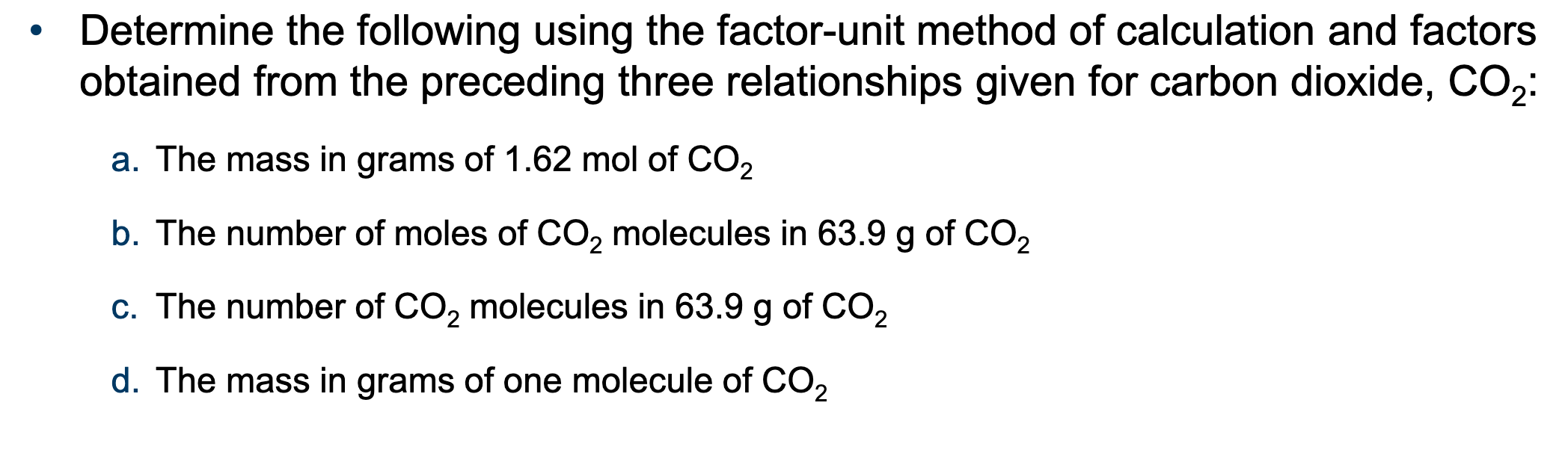Lesson 3: Atoms and Molecules
Symbols and Formulas
Each element is represented by a unique name and symbol.
Symbols are based on elemental properties or derived from scientists, places, astronomical bodies, or mythological characters.
Elemental symbols: One capital letter or a capital letter followed by a lowercase letter.
Compound Formula
Consists of the symbols of the atoms in the molecule.
Each elemental symbol represents one atom of the element.
Subscripts indicate the number of atoms of an element present in the compound.
At least 2 atom types in compound (ex. triatomic - 3 types of atoms)
Example:
Nitrogen dioxide: (one nitrogen atom, two oxygen atoms).
Sulfuric acid: H2SO4 (two hydrogen atoms, one sulfur atom, four oxygen atoms).
Atomic Structure
Atoms are composed of three subatomic particles: protons, neutrons, and electrons.
Protons(+, defines element) and neutrons(+- is a filler):
Located in the nucleus.
Tightly bound together.
Electrons(-):
Located outside the nucleus.
Move rapidly throughout a large volume of space surrounding the nucleus.

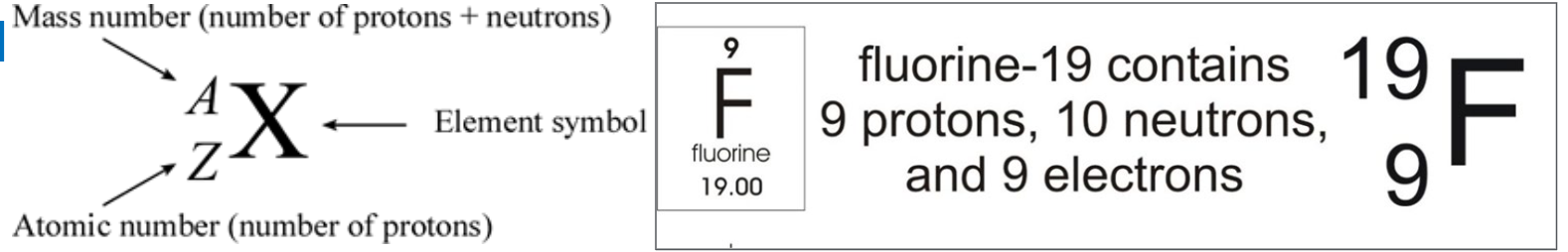
Atomic and Mass Numbers
Atomic number:
Equal to the number of protons in the nucleus.
Symbol: Z
Located above the element symbol in the periodic table.
Mass number:
Equal to the sum of protons and neutrons in the nucleus.
Symbol: A
Isotopes
Atoms with the same number of protons but different numbers of neutrons.
Same atomic number (Z) but different mass numbers (A).
Example: Chlorine-35 and Chlorine-37.
All isotopes of the same element have:
Same number of electrons outside the nucleus.
Same number of protons in the nucleus.
Masses of Atoms and Molecules
Atomic Mass:
Numbers beneath the element symbol in the periodic table.
Provide a means of comparing the masses of atoms.
Atomic weight of elements is the average relative mass of the atoms in the isotope mixture.
Atomic Mass Unit (u):
Used to express the relative masses of atoms.
the mass of a carbon-12 atom.
One carbon-12 atom has a relative mass of 12 u.
An atom with a mass equal to twice the mass of a carbon-12 atom would have a relative mass of 24 u.
Molecular Weight:
Relative mass of a molecule expressed in atomic mass units.
Calculated by adding together the atomic weights of the atoms in the molecule.
Example: Water ()
MW = (2 × atomic weight of H) + (1 × atomic weight of O)
MW =
Example Problem: Molecular weight of urea,
Avogadro’s Number and the Mole Concept
Avogadro’s number: Number of atoms or molecules in a specific sample of an element or compound.
Mole (mol): Number of particles in a sample with a mass in grams equal to the atomic or molecular weight.
1 mol =
Example: 1 mol S atoms = S atoms = 32.1 g S
Relationships:
1 mol S atoms = S atoms
S atoms = 32.1 g S
1 mol S atoms = 32.1 g S
The Mole and Chemical Formulas
Chemical formulas represent numerical relationships among atoms in a compound.
Example: represents a 2:1 ratio of hydrogen to oxygen atoms.
Relationships:
molecules contain H atoms and O atoms
1 mol contains 2 mol H atoms and 1 mol O atoms
18.0 g contains 2.0 g H and 16.0 g O
Mole Calculations:
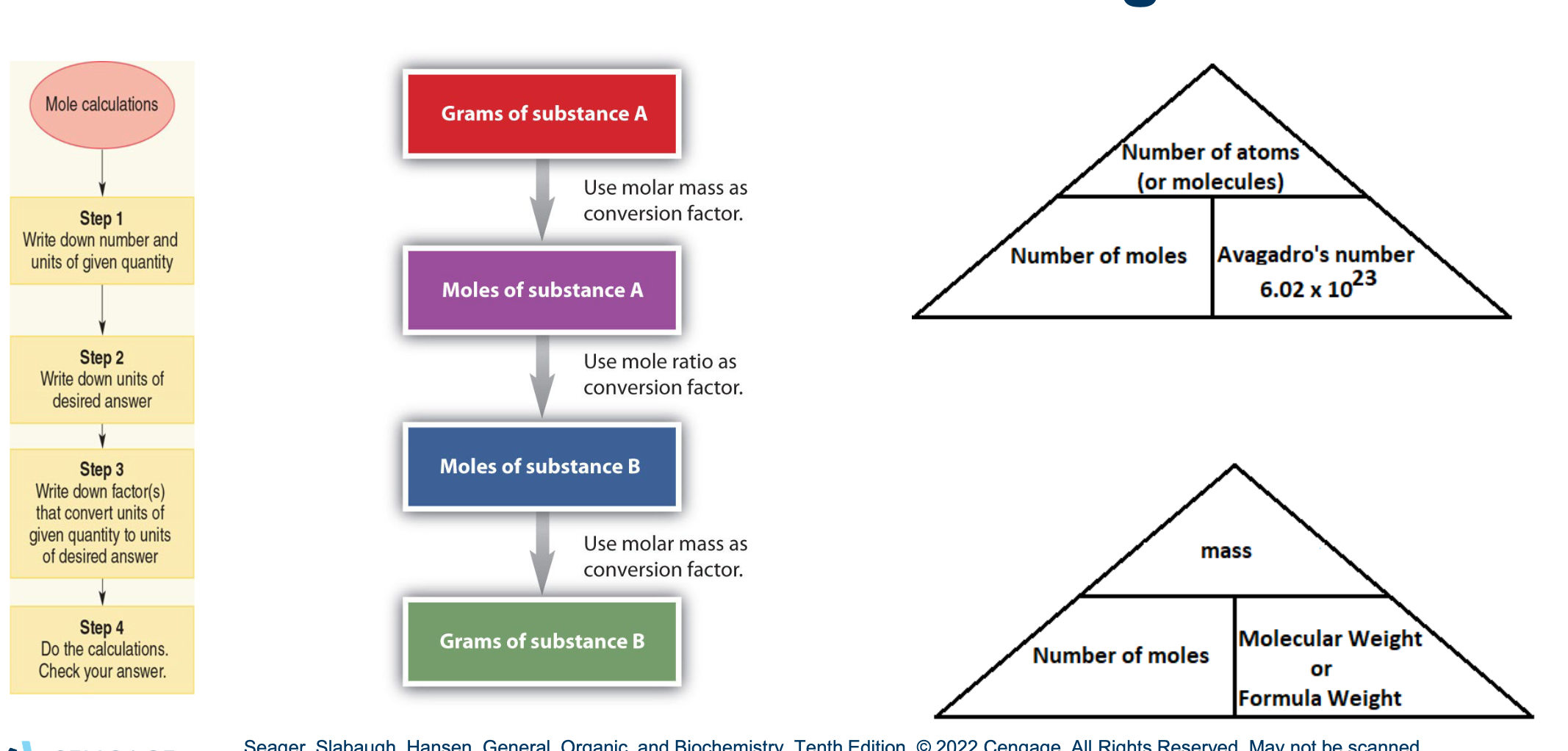
Mole Calculation Example
Calculate the number of moles of Ca in a 15.84 g sample of Ca.
Example - Factor-Unit Calculations for Sulfur (S)
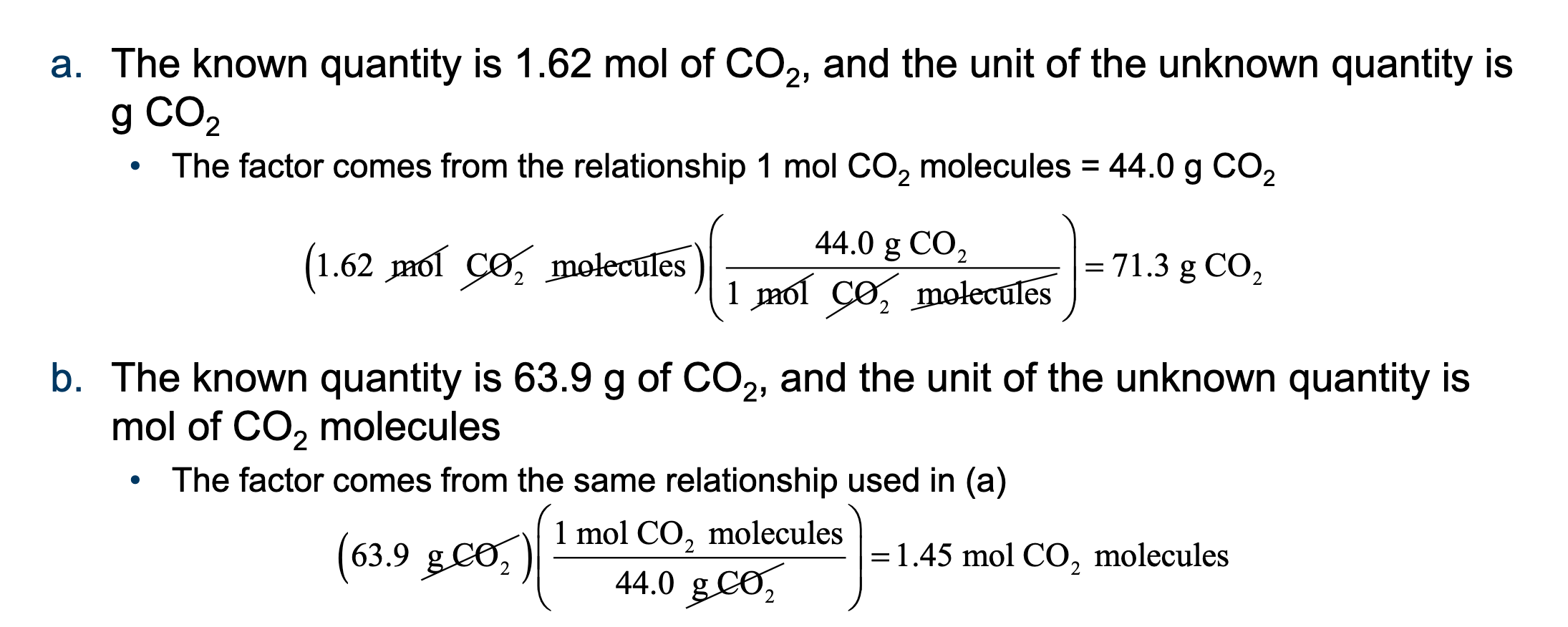
Determine the following using the factor-unit method:
The mass in grams of 1.35 mol of S:
The number of moles of S atoms in 98.6 g of S:
The number of S atoms in 98.6 g of S:
The mass in grams of one atom of S:
The Mole Concept Applied to Compounds
One mole of any compound is a sample with a mass in grams equal to the molecular weight of the compound.
1 mol CO26.02 × 10^{23} CO2CO2
Relationships for factor-unit calculations:
1 mol CO26.02 × 10^{23} CO2molecules
6.02 × 10^{23} CO2CO2
1 mol CO2CO2
1 mol contains 1 mol C atoms
1 mol contains 2 mol O atoms
1 mol C atoms = 12.01 g C
Mole Calculation Examples

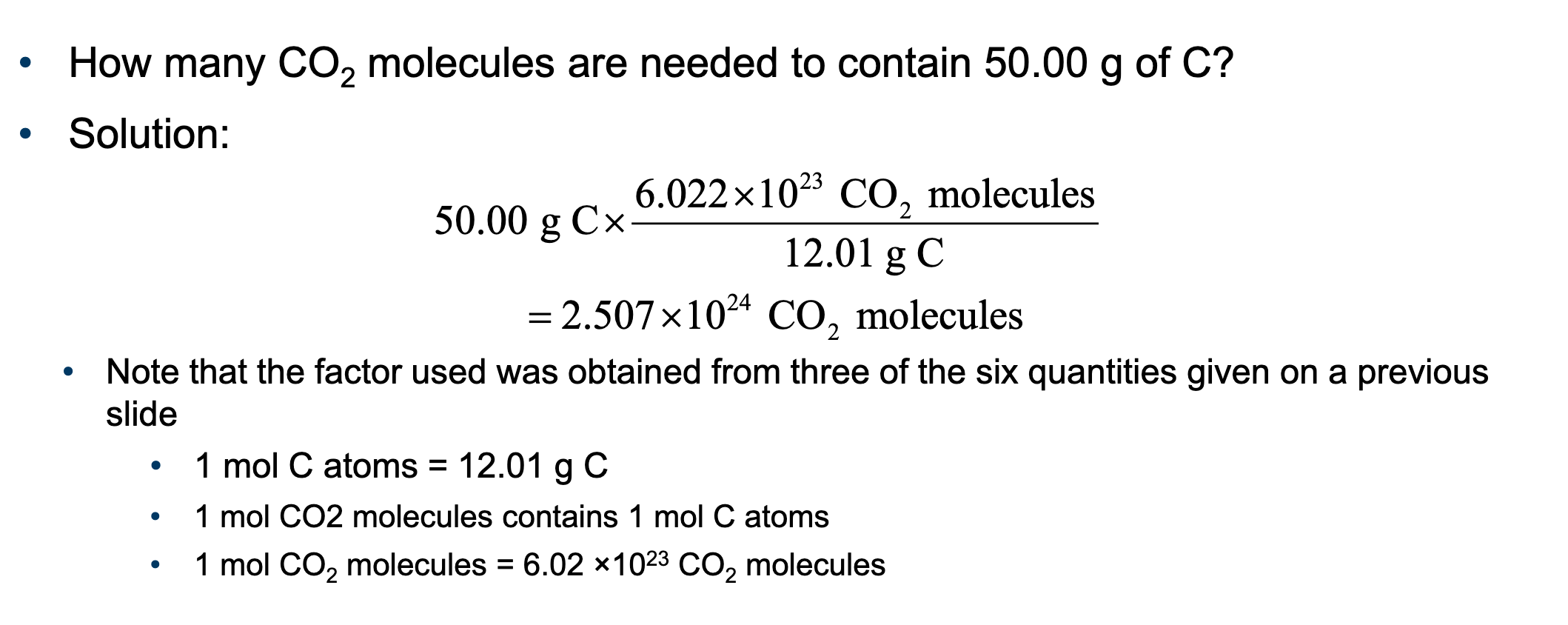

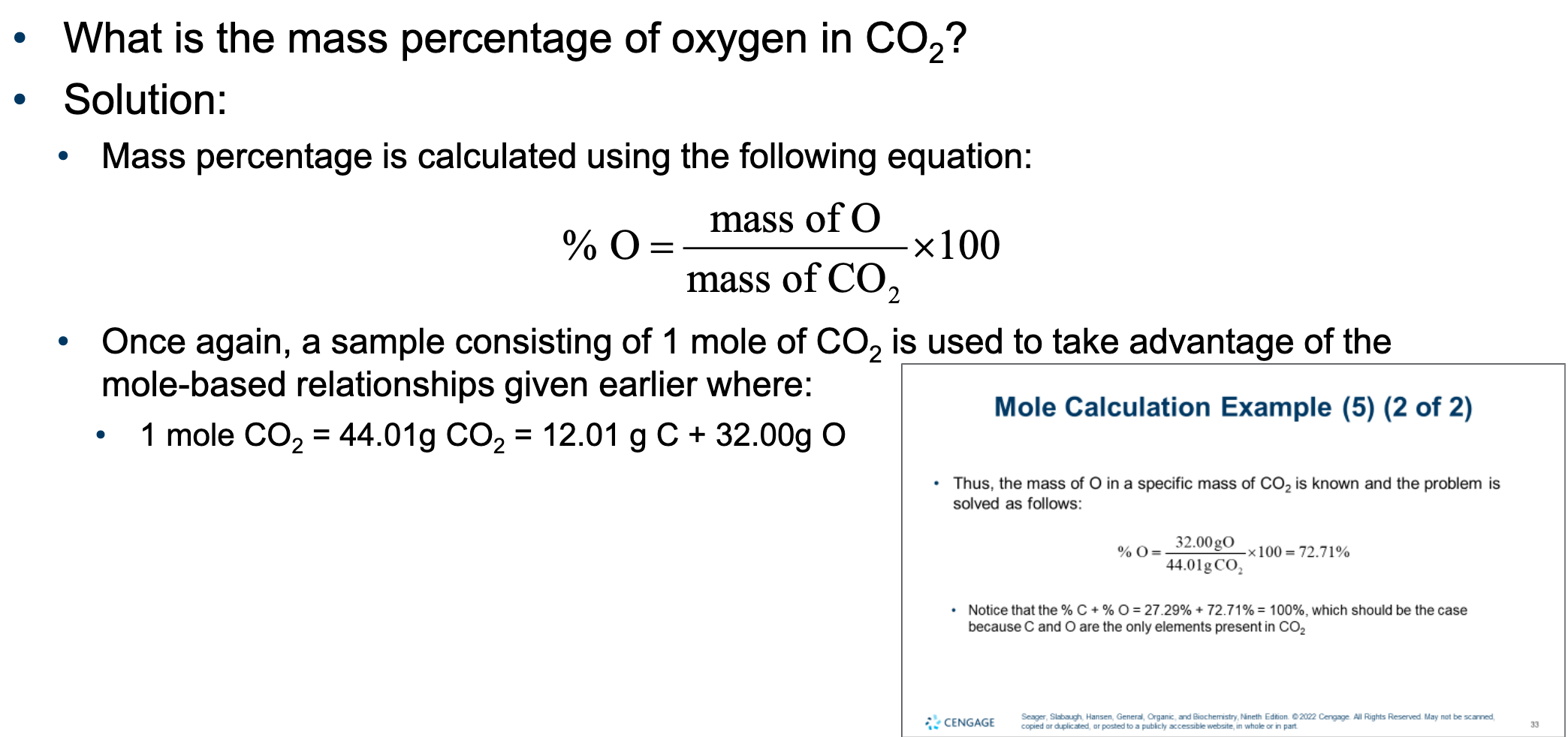
Example - Factor-Unit Calculations for Carbon Dioxide