Properties
Dmitri Mendeleev created the periodic table.
Valence electrons are bonding electrons and occupy the outermost principle energy level while core electrons occupy complete principle energy levels as well as d and f orbitals.
Trends
Atomic Radii
The nonbonding radius (Van der Waals radius) is 1/2 the distance between adjacent nuclei. The bonding radius (covalent radius) is half the distance of two atoms bonded together. The bond length of a covalent bond is the time of their atomic radii.
Atomic radii increases down and decreases across the table. Down the table shells get added, which increase the radius, and across the table the effective nuclear charge holds the electrons closer, which decreases the radius.
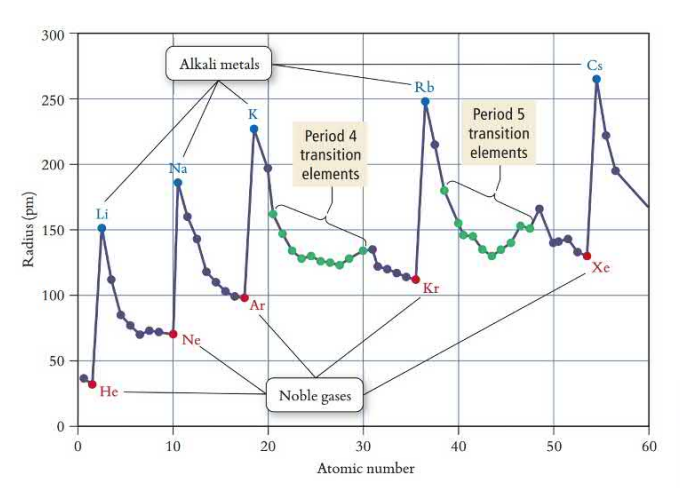
d-block elements do not follow the atomic radii trend because their outermost shell is not the d-orbital, it is the s-orbital. The electrons added are shielded more than the outermost electron.
Effective Nuclear Charge
The charge of the nucleus felt by any electron can be calculated using Slater’s rule. The lower the effective nuclear charge, the larger the atomic radii.
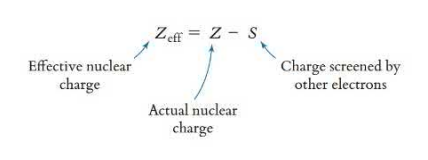
All electrons in the same principle energy level as the electron you are solving for are multiplied by 0.35. Any electrons higher, if they exist, are not counted. Electrons in one energy level lower than the one being calculated are multiplied by 0.85 is the electron we are solving for is in the s or p orbital, and 1.00 if the electron we are solving for is in the d or f orbital. Any electrons of a lower energy level than what has been calculated are multiplied by 1.00.
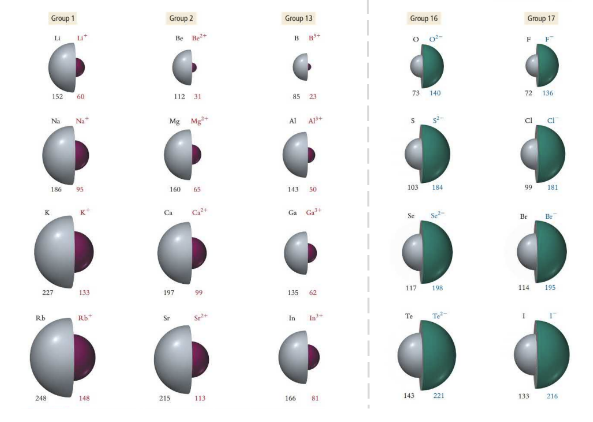
Ionic Radii
When electrons are removed and a cation is formed, the ionic radii is smaller than the atomic radii. When an electron is gained and an anion is formed, the ionic radii is larger than the atomic radii.
Ionization Energy
Ionization energy is the energy required to remove an electron. IE decreases down and increases across the table. Ionization energy increases as the effective nuclear charge increases. The first time removing an electron (first IE) is always endothermic, heat is required. Each successive ionization energy requires more energy to be inputted due to increases effective nuclear charge felt by the electrons.
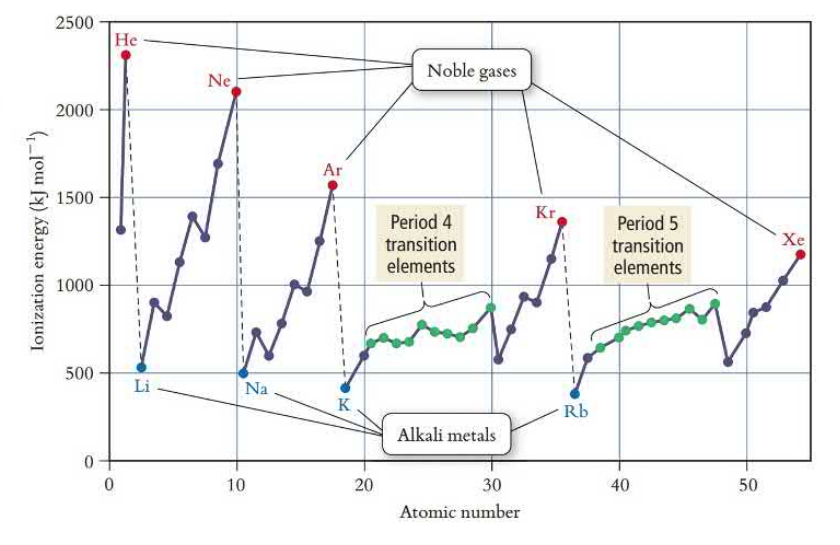
Exceptions to the trend occur due to increased electron repulsion when a new subshell is added or when electrons are paired. The repulsion makes it easier for electrons to be removed.
Electron Affinity
Electron affinity is the energy released when an electron is gained. EA decreases down and increases across the table. The decrease in EA down is not the most definite trend.
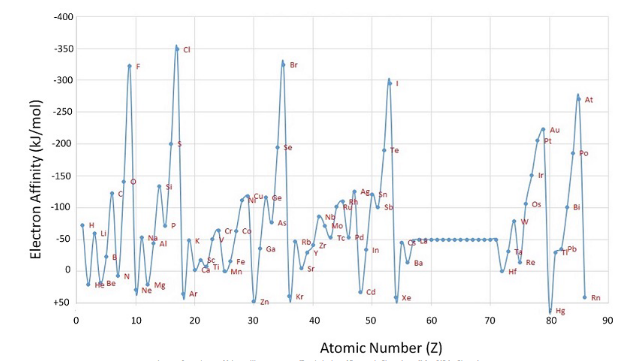
Exception occur when an electron from a full subshell is removed and when an electron from an unpaired subshell is removed, as seen in groups 2, 15, and 18. This is due to the loss of stability created by removing and electron from a full subshell/unpaired full subshell/full valence shell.
The first EA can be any value and any successive ones will be positive, meaning energy has to be used in order for an electron to be gained.
Electronegativity
Linus Pauling developed the electron negativity scale which is a numeric value between 0 and 4 that represents an elements power to attract electrons when apart of a compound. EN decreases down and increases across the table.
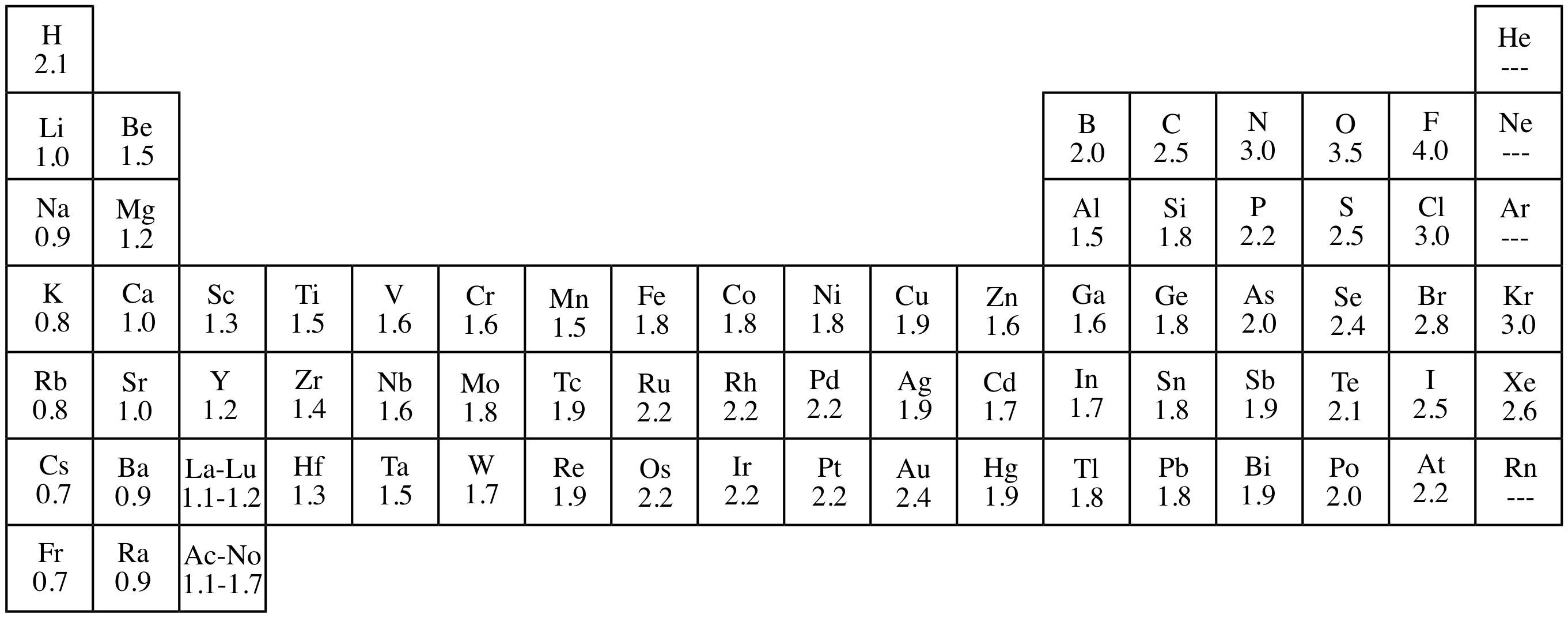
Ionic bonds occur when the EN difference is greater than 2. This generally happens when metals and non-metals bond. The metal is oxidized (small EN) and the non-metal is reduced (large EN).