Properties of Water
Properties of Water
Water is essential for life and possesses unique properties due to its molecular structure and the way its molecules interact:
Polar vs. Nonpolar Molecules
Polar Molecules: These molecules have an unequal distribution of charges, leading to a partial positive charge on one end and a partial negative charge on the other. For example, water (H₂O) is polar, with oxygen being more electronegative, pulling electrons closer to itself.Nonpolar Molecules: These molecules have an equal distribution of charge and do not have charged ends. An example includes oil (e.g., hexane), which is hydrophobic.
Hydrophobic vs. Hydrophilic Substances
Hydrophilic Substances: These are substances that interact well with water due to their polar nature. They can dissolve in water and include substances like salts and sugars. For instance, table salt (NaCl) is hydrophilic as it dissociates in water.Hydrophobic Substances: These substances do not interact well with water and are typically nonpolar. Examples include fats and oils, which do not mix with water and instead form separate layers.
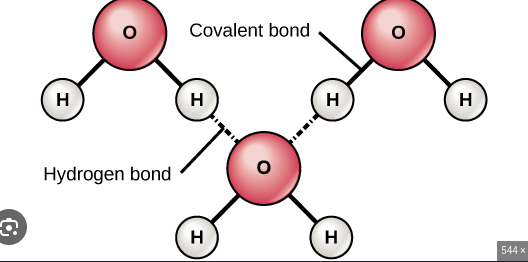
Hydrogen Bonds in Water
Hydrogen bonds form between the slightly positive hydrogen atoms of one water molecule and the slightly negative oxygen atom of another water molecule. This can be diagrammatically represented as:H|O -- H|H
Effects of Water's Properties
Cohesion: Water molecules are attracted to each other due to hydrogen bonding, allowing phenomena such as surface tension.
Life Promotion: Enables insects to walk on water and helps water transport in plants.
Adhesion: Water molecules can stick to other surfaces, which is crucial in capillary action in plants.
Life Promotion: Helps water rise through plant tissues against gravity.
Capillary Action: A combination of adhesion and cohesion allows water to flow in narrow spaces.
Life Promotion: Essential for water movement in the small tubes (xylem) of plants.
Temperature Stabilization: Water can absorb a lot of heat without a significant change in temperature due to its high specific heat.
Life Promotion: Helps moderate Earth’s climate and maintain stable temperatures in living organisms.
Specific Heat: Water can absorb a lot of heat without a significant change in temperature. Its specific heat is high compared to most substances.
Life Promotion: Helps moderate Earth’s climate and maintains stable temperatures in living organisms.
Density of Ice vs. Water: Water expands and becomes less dense as it freezes, causing ice to float.
Life Promotion: Insulates aquatic life in winter, preventing bodies of water from freezing solid.
Universal Solvent: Water can dissolve more substances than any other liquid, due to its polarity.
Life Promotion: Facilitates biochemical reactions and nutrient transportation in living organisms.