Chemical bonds, ionic, covalent and metallic
Chemical bonds
There are three types of strong chemical bonds: ionic, covalent and metallic:
For ionic bonding the particles are oppositely charged ions.
For covalent bonding the particles are atoms which share pairs of electrons.
For metallic bonding the particles are atoms which share delocalised electrons.
Ionic bonding occurs in compounds formed from metals combined with non-metals.
Covalent bonding occurs in most non-metallic elements and in compounds of non-metals.
Metallic bonding occurs in metallic elements and alloys.
Ions
Ionic bonding
Ionic bonding is the strong electrostatic attraction between oppositely charged ions formed when electrons are transferred from a metal to a non-metal.
This is because when metal atoms lose electrons to form positive ions, and non-metal atoms gain electrons to form negative ions.
Ionic compound
Ionic compounds form a giant ionic lattice (3d structures) with strong electrostatic forces of attraction acting in all directions between the ions.
Formula of ionic compounds
Ionic compounds must have an overall charge of zero.
So the positive charges must balance the negative charges.
Properties
High melting and boiling points
Ionic compounds have high melting and boiling points because there are strong electrostatic forces of attraction between oppositely charged ions in a giant ionic lattice, which require a large amount of energy to overcome.
Electrical conductivity (solid)
When solid, ionic compounds do not conduct electricity because the ions are held in fixed positions in the lattice.
Although the ions can vibrate, they cannot move freely, so charge cannot flow.
Electrical conductivity (molten)
When molten (liquid), ionic compounds can conduct electricity because the ionic lattice breaks down and the ions are free to move.
The movement of charged ions allows electric current to be carried.
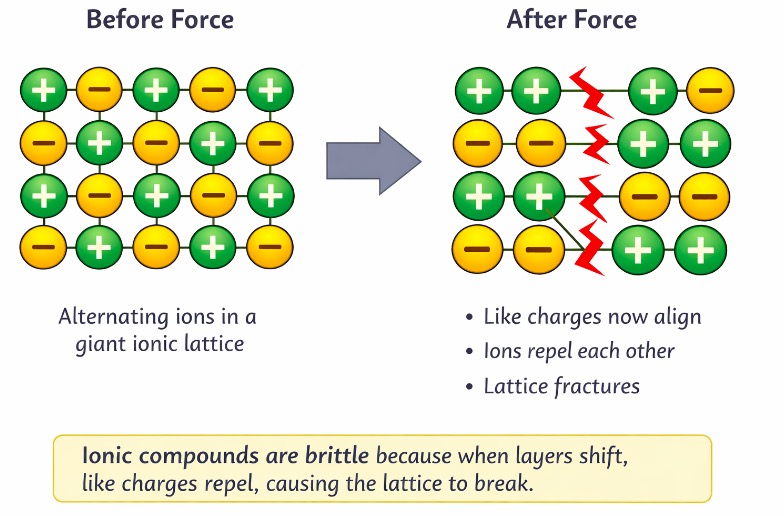
Shape and strength
Strong forces act in all directions
Makes ionic compounds:
Hard
Brittle (layers shift → like charges repel (electrostatic repulsion) → lattice break.
Covalent
Covalent bonding
Covalent bonding = sharing of a pair of electrons between non-metal atoms.
When they share their electrons, they form covalent bonds
This is so that they can achieve a full outer shell.
Covalent bonding forms a molecule.
They are held together by shared electrons.
Single bond=each atom shares one pair of electrons.
Double bond=each atom shares two pairs of electrons.
Small covalent molecules - substances made of a few atoms joined by strong covalent bonds, but they are held together as molecules by weak intermolecular forces.
Low melting and boiling points
→ Only weak intermolecular forces need to be overcome, not the strong covalent bonds.
As the temperature increases, the molecules vibrate more and the weak intermolecular bonds(forces) stretch(melting point).
As the temperature increases even more, the weak intermolecular bonds(forces) break(boiling point) and the molecules turn into a gas.
Do not conduct electricity
→ They have no delocalised electrons or ions that can move.
Giant covalent structures - huge continuous network of atoms held together by strong covalent bonds in all directions.
Very high melting and boiling points
→ A vast amount of energy is required to break the many strong covalent bonds in the giant structure.Generally hard (except graphite)
→ Atoms form rigid, tightly bonded networks.Do not conduct electricity
→ Most have no charged particles or delocalised electrons (diamond, SiO₂).Graphite conducts electricity
→ Each carbon has one delocalised electron, free to move between layers.
Large molecules are very long chains of repeating units (monomers) joined by strong covalent bonds along the chain.
The chains are held next to each other by intermolecular forces that are stronger than in small molecules but weaker than in giant structures.
Higher melting and boiling points than small molecules
→ Polymer chains are long → more intermolecular forces → more energy needed to separate them.Usually solids at room temperature
→ Many intermolecular forces between long chains.Do not conduct electricity
→ Polymers have no delocalised electrons or ions.Flexible
→ Chains can slide over each other because forces between chains are weaker than covalent bonds.
(POLYMERS ARE AN EXAMPLE OF THIS)
Metallic
(METAL AND METAL)
Metallic bonding is the strong electrostatic attraction between:
positive metal ions (a giant lattice of cations) and
a sea of delocalised electrons that are free to move through the structure in the outer shell.
This forms a giant metallic lattice/structure.
1. High melting and boiling points
Reason:
Metals contain strong electrostatic forces of attraction between positive ions and delocalised electrons.
A large amount of energy is needed to overcome these forces.
2. Good electrical and thermal conductors
Reason:
Metals have delocalised electrons that are free to move:
Electricity: electrons flow through the structure, carrying charge.
Heat: electrons transfer kinetic energy rapidly.
3. Malleable and ductile (can be bent or hammered into shape)
Reason:
Layers of positive ions can slide over each other without breaking the strong metallic bond because the delocalised electrons hold the structure together even when the layers move.
4. Dense
Reason:
Metal ions are packed closely together in a regular lattice.
Alloys (TRIPLE)
Mixture of metals or metal + carbon.
Different-sized atoms distort layers → prevent sliding.
Harder and stronger than pure metals.
States of Matter
Particle model:
Solid: fixed positions, vibrate, closely packed.
Liquid: close but can move past each other.
Gas: far apart, move randomly and rapidly.
Changes of state:
Melting, freezing, evaporation, condensation, sublimation.
These are physical changes: no new substances formed.
Energy changes:
Energy is needed to break intermolecular forces (melting/boiling).
Energy released when forming forces (freezing/condensing).
Structure and Bonding — Summary Area Sizes (Triple)
Coarse particles: 2500–10,000 nm
Fine particles: 100–2500 nm
Nanoparticles: 1–100 nm
Nanoparticles have a very large surface area : volume ratio.
Nanoparticles (TRIPLE ONLY)
Nanoparticles
Very small particles: 1–100 nm.
Contain only a few hundred atoms.
Very large surface area : volume ratio → more reactive.
Uses:
Catalysts
Drug delivery systems
Electronics
Antibacterial coatings
Sun creams
Cosmetics
Risks / Concerns:
Not fully understood
May be toxic
Potential environmental impact
Must be thoroughly tested before widespread use
Limitations of models:
Dot-and-cross: doesn’t show lattice shape or scale.
2D diagrams: inaccurate distances.
Ball-and-stick: bonds shown as sticks (not real), gaps exaggerated.
Space-filling: no clear bond angles.