AP Chem REMEMBER
Carbon and hydrogen form a NONPOLAR bond
VSPER: name, what it looks like, and bond angle
Formal charge (# val e - # lone e - # bonds)
ideal gas assumptions
Solubility of salts (SNAP ions will always be soluble in water: Sodium (Na+), Nitrate (NO3-), Ammonium (NH4+), Potassium (K+))
any group 1 ion is soluble
anything with acetate (C2H3O2-) is soluble
if it’s not one of these, assume it’s insoluble in water, unless told otherwise
how each type of radiation affects molecules (microwave - rotation, infrared - vibration, ultraviolet/visible - transitions in electronic energy levels)
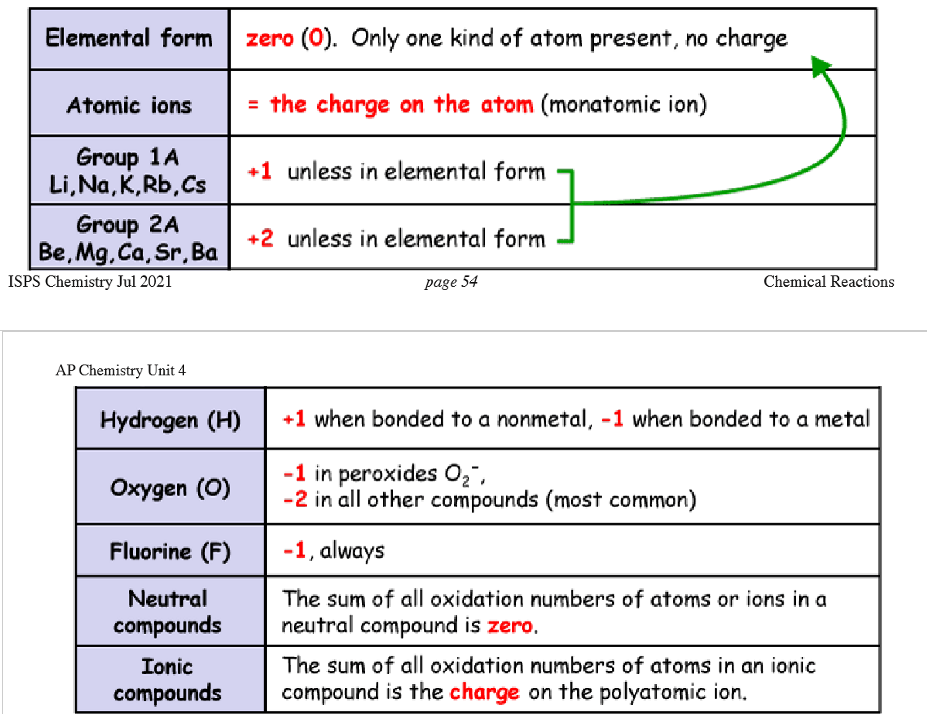
Oxidation Numbers Rules
These strong acids which completely dissociate in water, they completely ionize
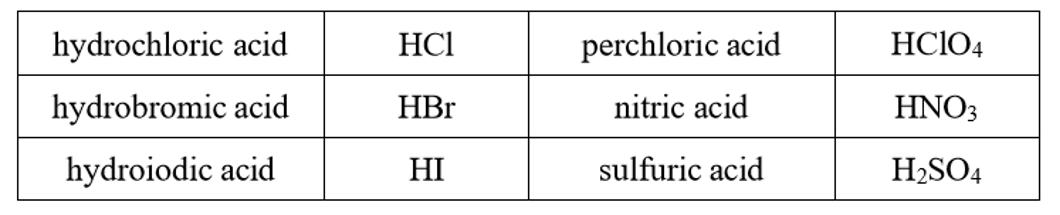
When writing the acid base reaction, they only work one way because the conjugate bases are too weak to work the other way