Veterinary Medical Practices: Pharmacology
Terms & Terminology
Pharmacology
· Is the study of medicine which focuses on the uses, effects and modes of actions in drugs
· Is divided into the study of pharmacokinetics and pharmacodynamics
Pharmacokinetics
· Is the study of drug absorption, distribution, metabolism and excretion
· Affects the route of administration, dose, dose intervals and toxicities of drugs
Pharmacodynamics
· Is the study of the biochemical and physiological effects of drugs and their actions
Drug
· Is a substance used to treat, cure, prevent or diagnose a condition
· Classifications include:
- diagnostic
- thrapeutic
- preventive
Diagnostic Drugs
· Are used as a part of a test in order to identify and label a condition
· Help veterinarians determine proper treatment
· Examples include:
- iodine used to help detect health problems during x-rays
- dexamethasone suppression test is used to assess the adrenal gland function by injecting the drug and measuring the response
Therapeutic Drugs
· Are used in the treatment of a condition
· Examples include:
- antibiotics
- aspirin
- analegsics (pain relief)
Preventive Drugs
· Are used to prevent a condition
· Examples include:
- heartworm medicine
- flea and tick treatments
- vaccinations
Drug Sources
· Include:
- plants
- animals
- minerals
- synthesized
- biological
Clinic Corner: Plants are the oldest source of drugs.
Drug Names
· Include:
- chemical
- generic
- brand name
Chemical Name
· Provides scientific and technical information
· Describes the chemical structure of the drug
Clinic Corner: Chemical names are rarely used in clinical medicine because they are long and complex.
Generic Name
· Is the official identifying name of the drug
· Is sometimes referred to as the non-proprietary name
· Commonly describes the active ingredient
Clinic Corner: Generic names are always written using lower case letters and are the same in all countries.
Brand Name
· Is referred to as the proprietary name
· Is the name owned by the drug manufacturer
· Is registered with the U.S. patent office
Drug Categories
· Include:
- over-the-counter (OTC)
- prescription
- Veterinary Feed Directive (VFD)
- controlled substance
Clinic Corner: The FDA classifies OTC, prescription and VFD as marketing categories.
Over-the-Counter Drugs (OTC)
· Are available to anyone without a prescription
· Common examples include:
- Aspirin
- Benadryl®
- glucosamine
- hydrocortisone
Prescription Drugs
· Are regulated by the U.S. Food and Drug Administration (FDA)
· Are limited to use under the supervision of a licensed veterinarian
· Must be labeled with the following statement or legend:
- “Caution: Federal law restricts the use of this drug to use by or on the order of a licensed veterinarian.”
Clinic Corner: Because the caution statement or legend is required, prescription drugs are sometimes referred to as legend drugs.
Veterinary Feed Directive (VFD) Drugs
· Is intended for use in animal feeds
· Is permitted only under the supervision of a licensed veterinarian
Controlled Substances
· Are drugs considered to be dangerous due to potential human abuse or misuse
· Are classified as “controlled” by the FDA and enforced by the Drug Enforcement Agency (DEA)
· Common examples include:
- oxycodone
- morphine
- hydrocodone
- pentobarbital
Drug Compounding
· Is the preparation, mixing, assembling, packaging and/or labeling of a drug
· Is used to prepare a specific drug product to fulfill an individual patient’s need
· Examples include:
- flavoring
- dosing for weights
- changing pills into liquid
Withdrawal Period
· Reflects the amount of time necessary for an animal to _____________ an administered drug and the amount of time needed for the _____________ levels to decrease to a safe level
· Is printed on the product label or package insert
Extralabel Drug (Part 1)
· Also referred to as “off-label use”
· Is printed as:
- “Actual use or intended use of a drug in an animal in a manner which is NOT in accordance with the approved labeling.”
Clinic Corner: Veterinarians can use both approved animal and human drugs in an extralabel manner. When treating non-food animals a veterinary-client relationship must exist.
Extralabel Drug (Part 2)
· Examples include:
- phenobarbital (only approved for use in humans) to treat epilepsy in dogs
- ivermectin (antiparasitic only approved for cattle) in dogs and cats
- enrofloxacin (baytril) solution as a topical ear medication (only approved as injectable)
Clinic Corner: Extralabel drugs are used when a drug is not used in a way which is outlined with the approved FDA label directions. For example: the dosage, interval, route or species may differ.
New Animal Drug (NAD)
· Is defined as:
- “A drug intended for use in animals which is not Generally Recognized As Safe and Effective (GRASE) by qualified experts for the uses listed on the label.”
· Applications are required by manufactures to obtain marketing and selling approval from the FDA
Clinic Corner: The adjective “new” does not mean the drug just entered the market, but is a term outlined in the Federal Food Drug and Cosmetic Act.
Veterinary/Client/Patient Relationship (VCPR)
· Requires the veterinarian to:
- take responsibility for the health of the patient
- make a diagnosis and then keep up with the care of the patient with timely visits
- be available for follow-up evaluation or has arranged care and treatment
- provide oversight of treatment, compliance and outcome
- maintains patient records
Clinic Corner: The client’s, or owner’s, responsibility is to properly follow the veterinarian’s advice and instruction.
Governing Bodies & Regulations
Regulatory Agencies
· Governs how drugs are used in veterinary medicine
· Include both state and federal Levels
Federal Agencies
· Include:
- U.S. Food and Drug Administration (FDA)
- Center for Veterinary Medicine (CVM)
- Drug Enforcement Administration (DEA)
U.S. Food & Drug Administration (FDA) (Part 1)
· Is a scientific, regulatory and public health agency with jurisdiction encompassing the following:
- food products (other than meat and poultry)
- human and animal drugs
- therapeutic agents of biological origin
- medical devices
- radiation-emmiting products
- cosmetics
- animal feed
U.S. Food & Drug Administration (FDA) (Part 2)
· Enforces key legislative acts, such as:
- Pure Food and Drugs Act,1906
- Federal Food, Drug and Cosmetic Act, 1938
- Amendment to the Federal Food, Drug and Cosmetic Act, 1968
- Animal Medicinal Drug Use Clarification Act, 1994
- Animal Drug Availability Act, 1996
Pure Food & Drugs Act, 1906
· Prevents the manufacture, sale or transportation of misbranded or harmful foods, drugs or medicines
· Purpose was to protect the public against malpractice of food and drug products
Federal Food, Drug & Cosmetic Act, 1938
· Required manufacturers to provide evidence of drug safety before distribution
- when properly following label instructions
Clinic Corner: Animal drugs were regulated in three sections: new drugs, antibiotics and food additives if used on food-producing animals.
Amendment to the Federal Food, Drug, & Cosmetic Act, 1968
· Required manufactures of animal drugs to prove drug safety and effectiveness for intended use by:
- detecting unsafe drug residues in food
- providing acceptable drug withdrawal periods
Animal Medicinal Drug Use Clarification Act,1994
· Allowed veterinarians to prescribe extralabel drugs for animals under certain conditions
- approved new animal drugs
- approved human drugs
Animal Drug Availability Act,1996
· Amended the Federal Food, Drug and Cosmetic Act to provide new flexibility to the way FDA regulates new animal drugs and medicated feeds
- increased the number of approved new animal drugs on the market
The Center for Veterinary Medicine (CVM)
· Is a subdivsion of the U.S. Food and Drug Administration (FDA)
· Regulates the following:
- animal drugs
- animal feed
- veterinary devices
Clinic Corner: The CVM does not regulate the practice of veterinary medicine, vaccines for animals (USDA) and some flea and tick products (EPA).
Drug Enforcement Administration (DEA)
· Is a division of the U.S. departement of justice
· Enforces the controlled substance Act of 1970
Controlled Substances Act, 1970
· Is a federal drug policy which regulates the manufacture and distribution of controlled substances
· Categorizes drugs into five “schedules” or classifications based on their potential for abuse, status in international treaties and medical benefits
Records of Controlled Drugs
· Must include:
- orders
- receipts
- uses
- discards
- thefts
· Must be kept for two two years
State Agencies
· Include:
- Board of Veterinary Medicine(BVM)
- Board of Pharmacy(BOP)
Board of Veterinary Medicine (BVM)
· Protects the health and safety of the public and animals through the regulation of the practice of veterinary medicine
· Regulates in accordance with the state’s veterinary practice act
Board of Pharmacy
· Regulates wholesale and pharmacy distribution of veterinary drugs
· Regulates veterinarians which prescribe and distribute controlled substances
Veterinary Prescription Drugs
· Are to be used or prescribed only within the context of a veterinarian-client-patient relationship
- must be properly labeled before dispensing
- dispensing and treatment records must be filed
- should be dispensed in quantities required for the treatment, avoiding unlimited refills
Veterinary Prescription Orders
· Are issued by licensed veterinarians to authorized drug distributors for delivery of prescription drugs to:
- a client directly
- a pharmacist to dispense to a client
Drug Labels (Part 1)
· Must include:
- name, address and telephone number of veterinarian
- name, address and telephone number of client
- Animal Identification
- Date of treatment, prescribing or dispensing of drug
- name, active ingredient, quantity of the drug dosage and duration
Drug Labels (Part 2)
· Must include:
- route of administration
- number of refills
- cautionary statements
- expiration date
- slaughter withdrawal, or milk with-holding times
- signature
Common Veterinary Drugs
Common Veterinary Drugs
· Include:
- behavior modifying drugs
- non-steroidal anti-inflammatory drugs
- antibotics
- anti-parasitic drugs
- _____________ and sedatives
- corticosteroids
- anabolic steroids
- chemotherapeutics
Behavior Modifying Drugs
· Are used to treat behavioral problems, such as:
- fear
- anxiety
- aggression
- compulsive disorder
· Include:
- antidepressants
- anxiolytic drugs (i.e., anti-anxiety)
Clinic Corner: Hormones, such as synthetic progestin, have been used to aid in behavioral problems.
Non-steroidal Anti-inflammatory Drugs
· Are also known as NSAID
· Common uses include:
- pain relief
- osteoarthritis treatment in dogs
- pain management after surgery
Clinic Corner: Osteoarthritis is the permanent deterioration of the cartilage which surrounds the joints.
Antibiotics
· Are drugs used to inhibit the growth of bacteria
· Examples include:
- clavamox
- baytril
Anti-parasitic Drugs
· Are used to prevent or kill internal and external parasites
· Examples include:
- ivermectin
- pyrantel
- albendazole
Common Parasites
· Include:
Common Internal Parasites= pinworms
common external parasite= manage mites

Anesthetics & Sedatives (Part 1)
· Are used for restraint, minor procedures and general anesthesia
· Allow the patient to be unaware and not feel pain during surgery or other procedures or relax muscles throughout the body
Anesthetics & Sedatives (Part 2)
· Include:
Use 1st blank= provide pain control for specific locations
Use last blank= are used when an animal needs to be unconscious for surgical procedures
Corticosteroids
· Are used in pets to treat inflammation, allergies, itching, pain, trauma and skin, ear or eye problems
· Include:
- prednisone
- dexamethasone
- triamcincolone
- methylprednisolone
- prednisolone
Anabolic Steroids
· Are used to strengthen muscles and support growth, recover an animal’s appetite or treat certain types of anemia
· Examples include:
- winstrol v
· used to help underweight dogs
- equipoise
· used for race horses to increase red blood cell production
- finaplix
· pellets are used in cattle for increased meat production
Chemotherapeutics
· Are used to treat animals with cancer which has already spread to other areas of the body
· Attack cells in the process of growth and division, by:
- shrinking a large tumor
- killing microscopic cancer cells
- slowing the growth of cancer cells after surgery
Pharmacokinetics
· Is the physiological movement of drugs within the body and how drugs move into, through and out of the body
· Includes four steps, ADME:
- absorption
- distribution
- metabolism
- excretion
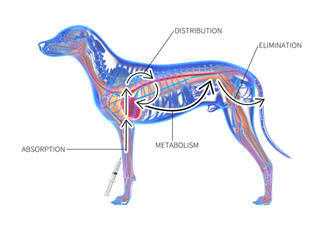
Drug Absorption
· Is the first step in the passage of a drug through the body
· Is the movement of a drug from the site of administration into the fluids of the body, which will then move to the site of action
Clinic Corner: Unless administered by an IV, a drug will have to cross several cell membranes before reaching circulation.
Bioavailability
· Is a subcategory of absorption
· Is the percentage of drug administered which actually enters systemic circulation
· IV and intra-arterial drugs immediately enter the blood and are 100 percent available
Clinic Corner: Orally administered drugs commonly have low bioavailability because they first have to be dissolved in gastric or intestinal fluids.
Drug Absorption Processes
· Which allow drugs to move across cell membranes include:
- passive diffusion
- facilitated diffusion
- active transport
- pinocytosis/phagocytosis
Passive Diffusion
· Is the movement of drug molecules from an area of high concentration to an area of low concentration until equilibrium is reached
- does not require energy or use of energy
Facilitated Diffusion
· Is a process by which molecules are transported across the plasma membrane by using a special carrier molecule
· Helps drugs move across the cell membrane
Clinic Corner: Carrier molecules are transport proteins. Transport proteins bind to the substance and alter its shape in order to bring the substance into or out of the cell.
Active Transport
· Uses both a carrier molecule and energy
- energy is needed because the drug molecules move against the concentration gradient
· Allows drugs to gather in high concentrations in a cell or body compartment
Factors Affecting Drug Absorption
· Include:
- lipid solubility
- drug pH
- molecular size
- patient factors (e.g., health, age, etc.)
Pinocytosis/Phagocytosis
· Is when a molecule is physically taken in or engulfed by a cell
- requires energy
- is important for the movement of larger molecules
· Pinocytosis
- cell membrane surrounds and engulfs liquid particles; also known as “cell drinking”
· Phagocytosis
- occurs when the cell membrane surrounds and engulfs solid particles; also known as “cell eating”
Lipid Solubility
· Is one of the most important determinants of drug absorbtion
- as a general rule, drugs which are highly lipid soluble are absorbed and distributed more rapidly
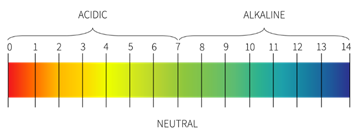
Drug pH
· Is the measurement of acidity or alkalinity of a substance
· Is based on a scale of 14, lower numbers are acid, higher are alkaline, 7 is neutral
Molecular Size
· Determines how rapid the absorption of the drug will be
- the smaller the molecular size of the drug, the faster it will absorb
Patient Factors for ADME
· Include:
- blood flow
- pain
- stress
- hunger
- fasting
- food consumption
- health
- age
- pH
Drug Distribution
· Is the movement of drugs from systemic circulation to the tissues and ends with the drug reaching the target tissue or site of action
· Factors include:
- volume of distribution
- protein binding
- tissue perfusion
- membrane permeability
Metabolism
· Is also known as biotransformation
· Is the chemical alteration of drug molecules into metabolites by body cells
- makes drugs more water soluble so they can be excreted from the body
· Sites include:
- liver (primary site)
- kidneys, small intestine, brain, lungs and skin
Metabolism Drug Interactions
· Include:
- inactivation
· active drug turns into an inactive metabolite
- activation
· inactive drug turns into an active metabolite
- modification
· activity after the conversion of an active drug to metabolite
- intoxication
· drug is moved into a normal cells metabolic pathway and then fails to react, resulting in cell death
Elimination
· Is the removal of a drug from the body
· Routes include:
- kidney
- liver
- intestine
- lungs
- milk
- sweat
- saliva
Drug Administration
Medical Treatment Steps
· Are assessed once a diagnosis has been made
· Include:
- diagnosis
- medication choice
- dosing regimen
The Six “Rights” of Proper Drug Administration
· Include:
- the right drug
- the right dose
- the right time
- the right route and technique
- the right patient
- the right documentation
The Right Drug
· Ensures the patient receives the drug which was prescribed
· Includes:
- reading the label three times
- never giving medication from a container that is unlabeled
The Right Dose
· Is the correct amount of drug administered at one time to achieve the desired effect
· Includes:
- loading dose
- maintenance dose
- total daily dose
Loading Dose
· Is the initial dose given to establish a therapeutic range
· Is often larger than subsequent doses
Therapeutic Range
· Is the drug concentration within the body to produce the desired effect in the animal with no signs of toxicity
· Factors include:
- properties of the drug
- Health of the animal
- absorption of the drug
- distribution of the drug
- Metabolism of the drug
- excretion of the drug
Maintenance Dose
· Is a dose which maintains or keeps the drug in the therapeutic range
· Is the amount of drug required to keep a desired concentration in the animal’s tissues
Total Daily Dose
· Is the total amount of the drug given within 24hours
- for example:
· 30 mg of a medication given four times a day = 120 mg total for the day
Dosage Regimen
· Represents the dosage interval, dosage, administration route and duration of treatment as a whole
- for example:
· 30 mg/kg three times daily (TID) orally (po) for 10 days
· Come from studies in normal animals but often require changes due to:
- disease
- age
- weight
- pregnancy
Dosage Interval
· Is how frequently the dose was given
- for example:
· once daily or twice daily
Tolerance
· Is a decreased response to a drug because the drug has been used repeatedly
- animal may require a larger dose
Effective Dose
· Is the amount of drug or dose which produces a desired effect
· Is determined by a positive result from at least 50% of the animals given the test dosage
Lethal Dose
· Is the quantity of a drug which will or may be sufficient to cause death in an animal
· Is determined by a giving a dose of a test drug which kills 50 percent of animals receiving it
The Right Time
· Is extremely important when correctly administering medication
· Includes:
- administering medication at specific times of the day
· for example:
− some medications must be given before or after meals
The Right Route & Technique
· Is how and where the medication goes into the body
· Must be followed by the prescription label on the medication and should be checked at least three times before administering the medication to the animal
Routes of Administration
· Include:
- parenteral
- oral
- local
- inhalation
- topical
Parenteral Administration
· Is given by route other than the GI tract and is injectable
· Is administered by needle or syringe
Common Injectable Routes
· Include:
- intramuscular (IM)
- intravenous (IV)
- subcutaneous (SQ or SC)
Intramuscular Injection
· Is injected directly into the muscle
· Is much easier to inject compared to intravenous or subcutaneous injections
· Is used for medication which are administered in small volumes
Intravenous Injection
· Is injected within the vein
· Can be administered in the following ways:
- bolus administration
- intermittent therapy
- continuous infusion of fluid
Bolus Administration
· Is when a drug is dilutes into a large volume of fluid, then rapidly injected using a syringe or needle
· Is most commonly used to rapidly increase or magnify a response, especially during emergency procedures
Intermittent Therapy
· Involves diluting a drug in a small amount of fluid and giving it to the animal during a period of 30 to 60 minutes
Continuous Infusion of Fluid
· Involves giving large volumes of fluid over long periods of time
· Is a dosing regimen used to deliver a constant amount of drug per unit time
· Is commonly used when short-acting drugs are need for long periods of time
Subcutaneous Injection
· Is given under the skin
· Is used when the drug is injected into the tissue layer between the skin and the muscle
· Absorbs more slowly than if it were injected into a vein
Other Injectable Routes
· Include:
- Intraperitoneal– into the lining of the abdominal cavity
- Intradermal– into the skin
- Intra-arterial– into the artery
- Intra-cardiac– into the heart
- Intra-articular– into the joint
- Intraosseous– into the bone
- intramammary– into the mammary gland
Inhalation Administration
· Is when the animal breathes the drug into the respiratory tract
- gas particles then enter the alevoli of the lung and diffuse across the membrane
- drug molecules then enter the blood
Topical Application
· Is applied on the surface of the skin or mucous membrane
· Can be an ointment, gel, cream, liniment, paste, lotion, powder, aerosol, etc.
- must first dissolve and then penetrate the skin
- absorbs much slower than any other route
Oral Route
· Sends the medicine directly to the GI tract and is more convenient to give
· Process includes:
- releasing from the tablet or liquid
- transport to the GI tract
- passage through the liver
Factors Influencing Administration
· Include:
- some drugs have different effects depending on the route of administration
- some can be given IM and not IV
- some may be destroyed by stomach acid
The Right Patient
· Protocol includes:
- always prepare medication for one patient at a time
- give the medication as soon as it is prepared
- do not talk to others while administering medication
- do not stop to do something else while in the middle of giving medication
- pay close attention at all times
The Right Documentation
· Protocol includes:
- each time a medication is administered, it must be documented
- documentation must be done at the time the medication is given
- always double check paperwork after administering medications and again at the end of the day
Dose Calculations
Calculating Doses
· Factors include:
- concentration
- strength
- volume
- weight of the animal
Concentration
· Is important for dosing in liquid drugs (oral and injectable)
· Refers to the amount of active drug (strength) in a given volume
· Concentration = Strength ÷ Volume
Strength
· Measures the amount of active ingredient of a drug per dosage unit
· In medications is available in different dosages
- for example:
· 10 mg per tablet or 50 mg per tablet
Volume
· Refers to the amount of liquid within a container
- for example:
· A bottle of medication holds 50 mL or an animal receives 10 mL of a medication
Weight of the Animal
· Is an important factor when calculating doses
- a prescription will be written as giving the correct amount of cc’s, number of pills, etc., per pound (lb.) or kilogram (kg)
Common Conversions
· Include:
- 1 liter = 1,000 mL
- 1mL= 1 cc
- 1 gram = 1,000 mg
- 1 kg = 2.2 lbs.
Recommended Dose Formula
· Weight of animal divided by prescribed dose = amount to give
- for example:
· a veterinarian prescribes a dose of medication which is 1 mL for every 10 lbs., this means a 10 lb. animal will receive 1 mL
· What if the animal weighs 45 lbs.?
45 lbs. x 1 mL = 45 lbs. mL = 4.5 mL
10 lbs. 10 lbs.
The animal will receive 4.5 mL
Convert to Kilograms
· Weight in kg = weight in lbs/ 2.2
2.2
20.45 kg = 45 lbs. 4.54 = 10 lbs.
2.2 2.2
· Weight of animal divided by prescribed dose = amount to give
20.45 kg = 1 mL = 20.45 kg mL = 4.5 mL
4.54 kg 4.54 kg
Concentration Dose Formula
· What if your veterinarian knows the weight of the animal and you are told to give 150 mg of a medication; the concentration on the bottle is 250 mg for every 5 mL?
· Use the following equation:
Prescribed dose/ strength × volume = amount to give strength
150 mg x 5 mL = 750 mg mL = 3 mL
250 mg 250 mg
The animal will receive 3 mL