L2 Role of the kidneys in homeostasis and clinical cases
Introduction
Instructor: Dr. Jon Mabley
Email: j.g.Mabley@brighton.ac.uk
Focus on the role of the kidneys in homeostasis and clinical cases related to renal function.
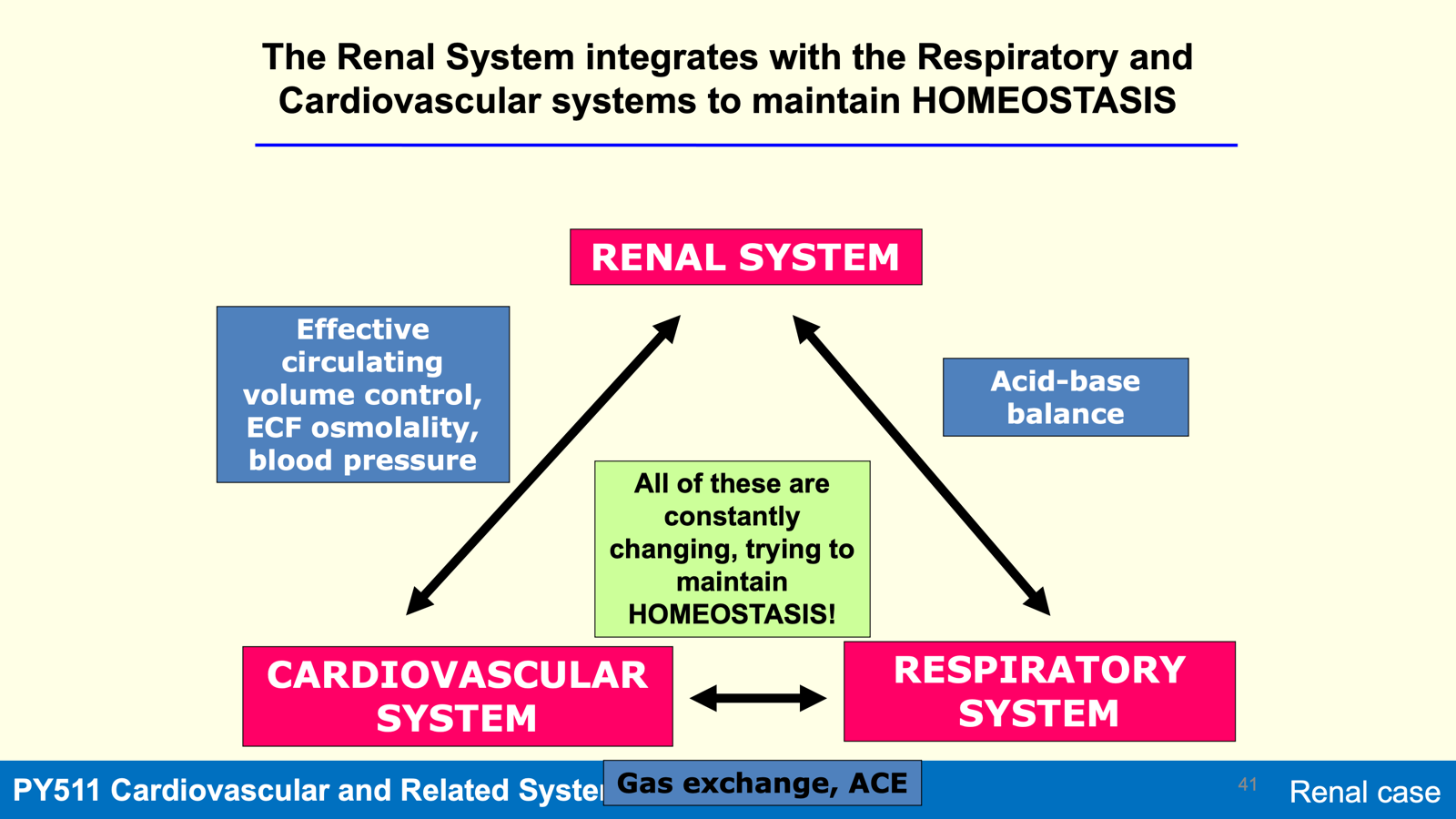
Integration of Renal, Cardiovascular, and Respiratory Systems
The Renal System interacts with the Cardiovascular and Respiratory Systems to maintain homeostasis, which includes:
Acid-base balance
Gas exchange
Effective circulating volume control
Extracellular fluid (ECF) osmolality
Blood pressure
Homeostasis is a dynamic process where all systems are constantly changing to adapt to physiological needs.
Renal Function and Dysfunction
Vital renal functions include:
Excretion of endogenous waste products - removal of waste generated from metabolism.
Excretion of drugs and their metabolites - clears medications from the body.
Water and electrolyte balance - maintains hydration and concentrations of ions.
Acid-base balance - regulates pH in the body.
Production of hormones
a. Erythropoietin - stimulates red blood cell production in response to low oxygen levels.
b. Renin - involved in the regulation of blood pressure.
Consequences of renal dysfunction:
Decreased renal excretory function leading to:
Uraemia - buildup of waste products in the bloodstream.
Drug toxicity - accumulation of medications.
Inability to maintain electrolyte and water balance leading to metabolic consequences.
Reduced hormone function may result in conditions like anaemia and hypertension.
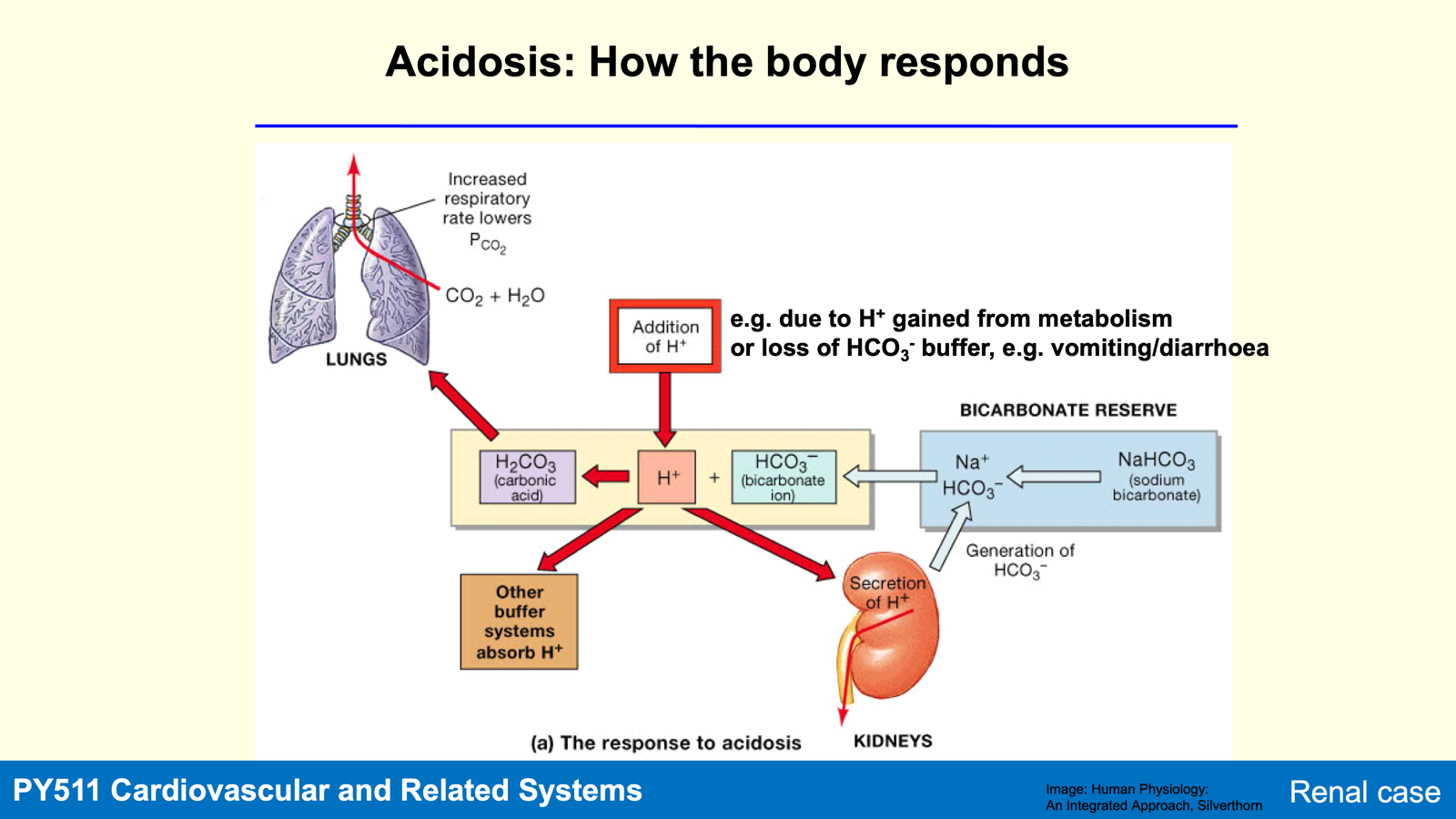
Role of Kidney in Acid-Base Balance
Acid-base balance is controlled by the kidneys through:
The offset of H+ gain by H+ loss.
Interaction between the renal system and cardiovascular and respiratory systems is crucial.
The kidneys play a key role in maintaining pH homeostasis of extracellular fluid (ECF).
Importance of pH Control
The normal pH range for ECF is 7.35 to 7.45:
Acidaemia: If plasma pH falls below 7.35, resulting in acidosis.
Alkalemia: If plasma pH rises above 7.45, resulting in alkalosis.
Alterations in pH outside of these boundaries impact all body systems:
pH < 6.8 or > 8.0 can result in death.
Acid-Base Balance Mechanisms
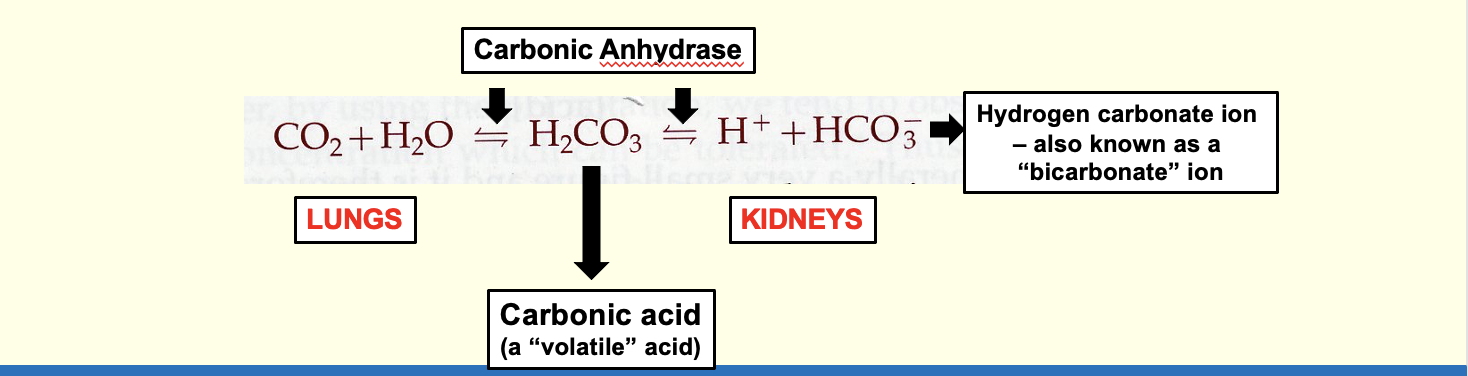
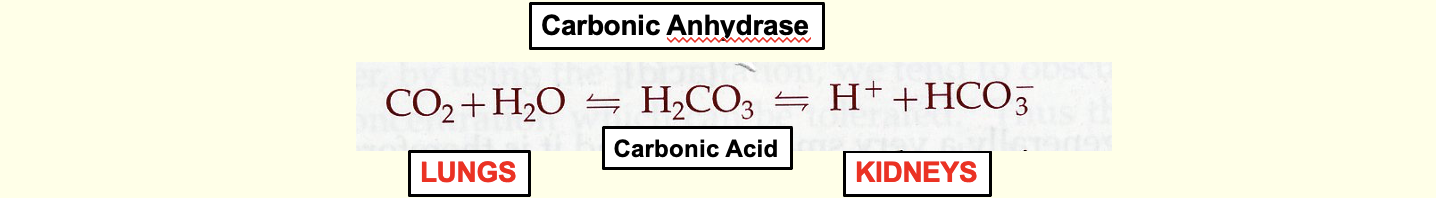
The kidneys are crucial for maintaining acid-base homeostasis in conjunction with the respiratory system.
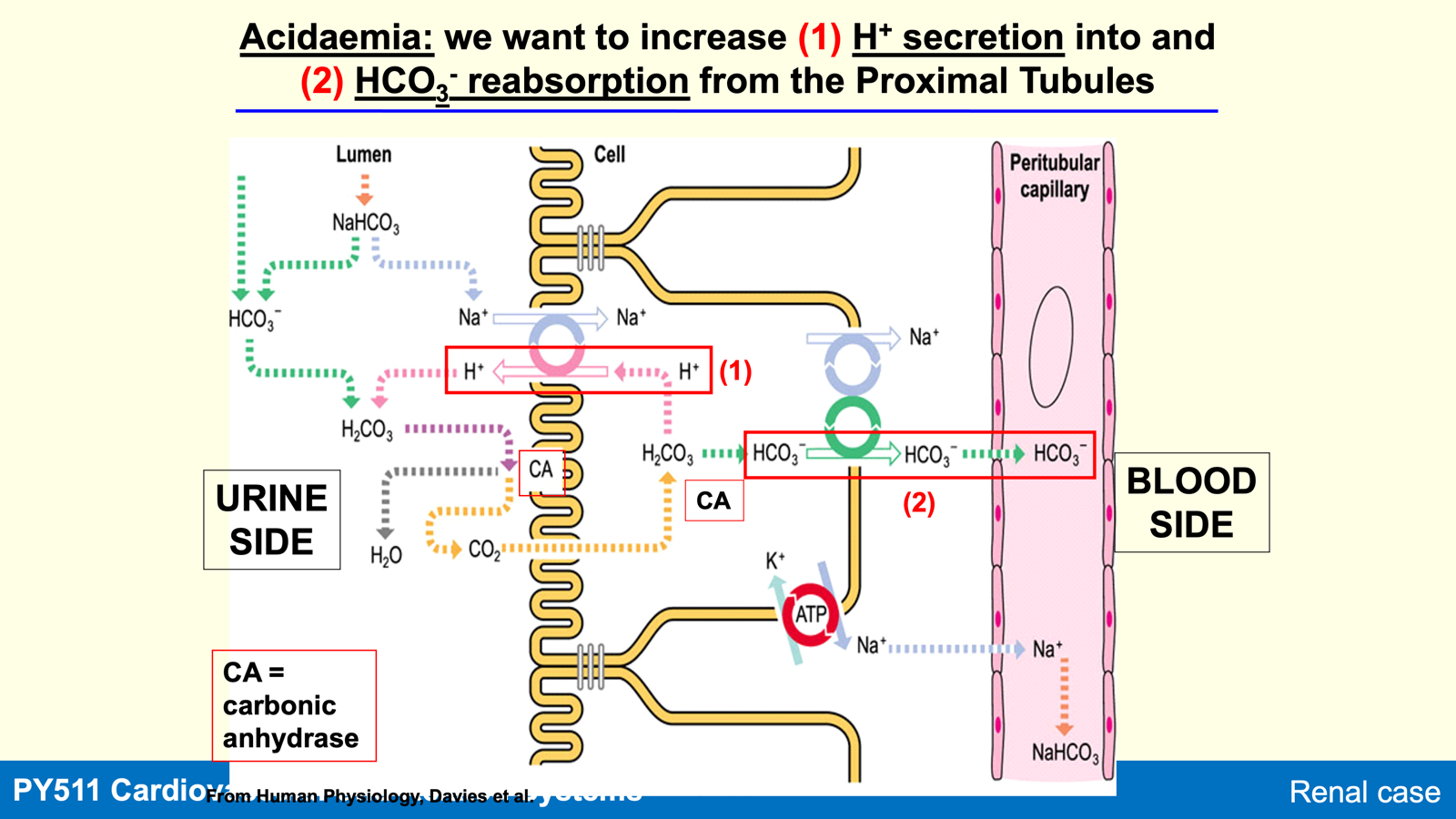
Carbonic Anhydrase facilitates the conversion between carbonic acid and bicarbonate (HCO3-) in both lungs and kidneys.
The lungs excrete CO2, a byproduct of metabolism that forms carbonic acid in water.
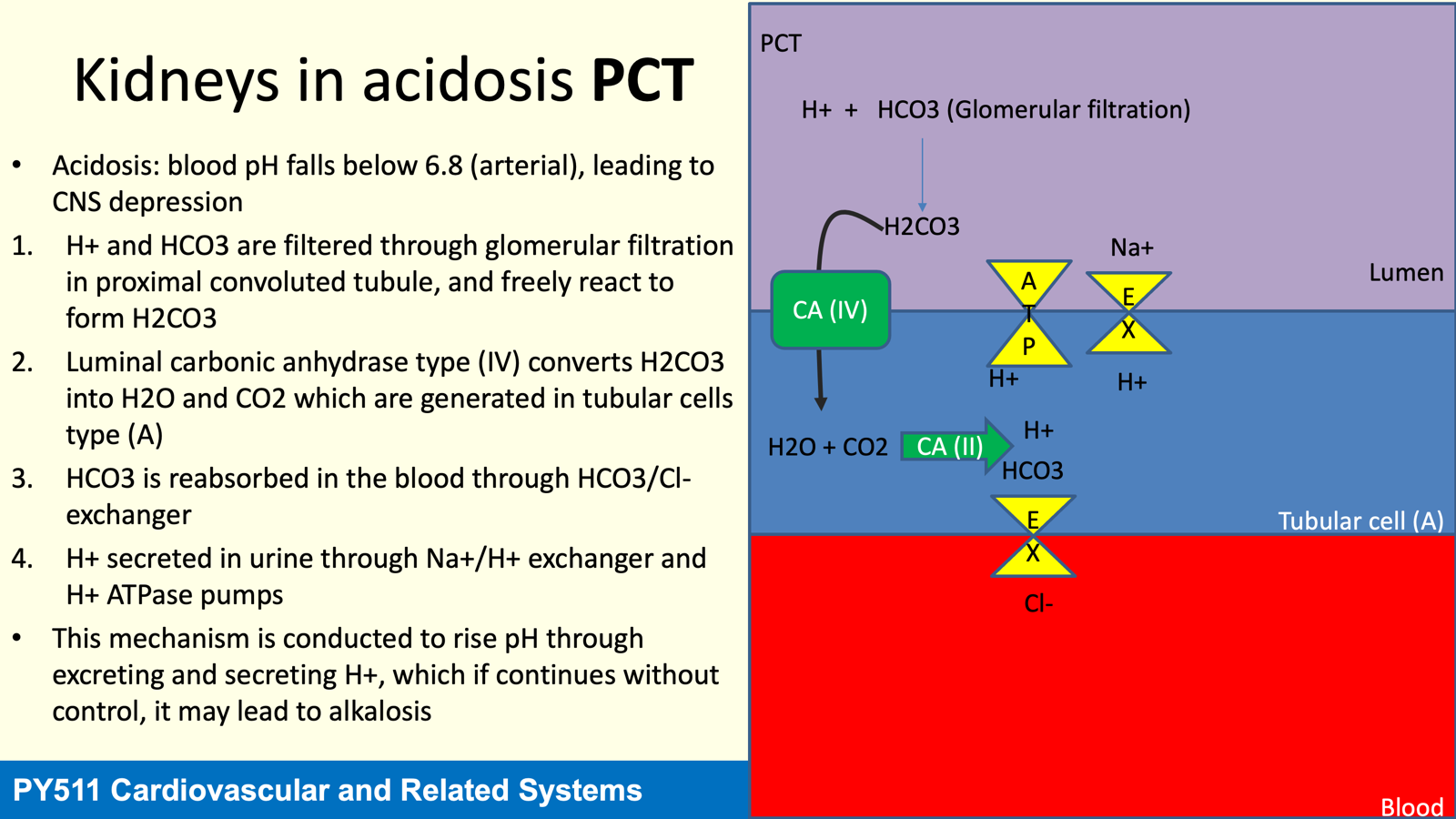
The kidneys also handle non-volatile acids, e.g. lactic acid, which cannot be excreted by the lungs.
The kidneys must reabsorb almost all filtered HCO3- as it acts as a physiological buffer to balance acid-base status.
Failure in these processes can lead to acidosis or alkalosis.
Relationships Among pH, HCO3-, and CO2
Relevant statistics in healthy states:
Blood pH should be approximately 7.4.
Urine pH expects around 6.0.
Blood concentration of HCO3- should be about 24 mM.
Blood partial pressure of CO2 (pCO2) is roughly 40 mmHg.
Plasma osmolality should be 285 mOsm/kg water with urine osmolality varying based on hydration from 50 to 1200 mOsm/kg.
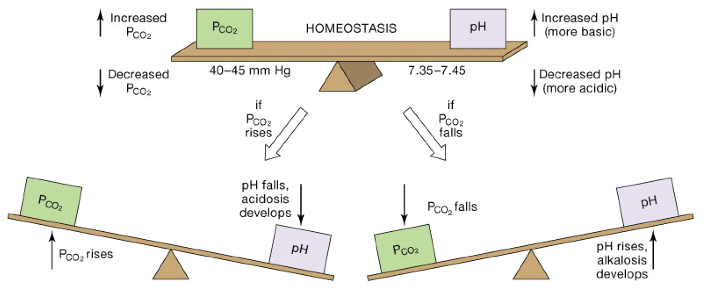
Inverse relationships are observed:
As pCO2 increases, pH decreases leading to acidosis.
As pCO2 decreases, pH increases leading to alkalosis.
Respiratory Regulation of Acid-Base Balance
Homeostasis is maintained via the HCO3-/CO2 buffer system:
Increased pCO2 levels result in lower pH.
Decreased pCO2 levels result in higher pH.
Changes in pH are sensed by peripheral chemoreceptors which communicate with brain's respiratory centers:
Hyperventilation decreases pCO2 and increases pH.
Hypoventilation increases pCO2 and decreases pH.
Renal Regulation of Acid-Base Balance
The kidneys are pivotal in regulating body fluid pH:
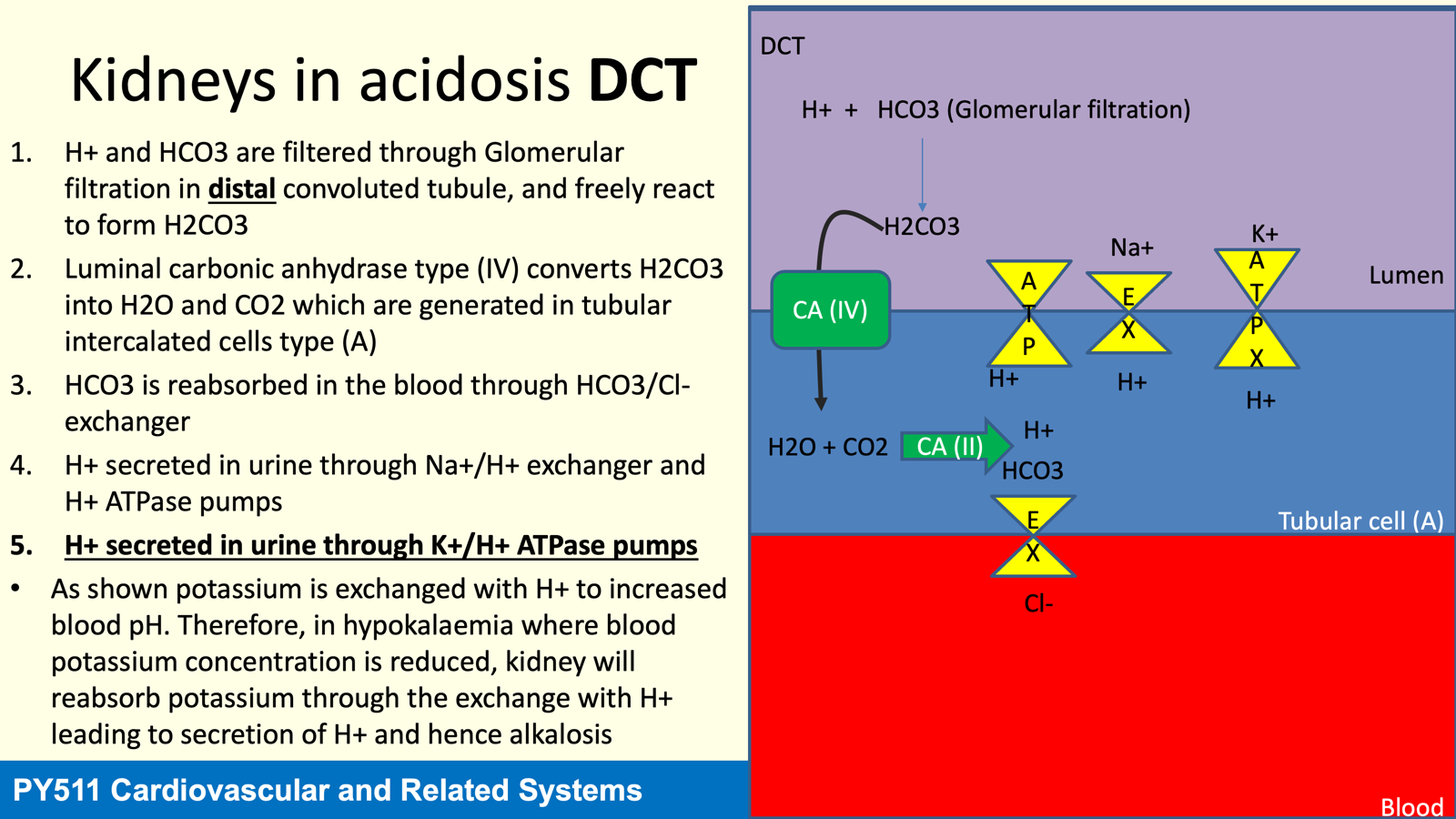
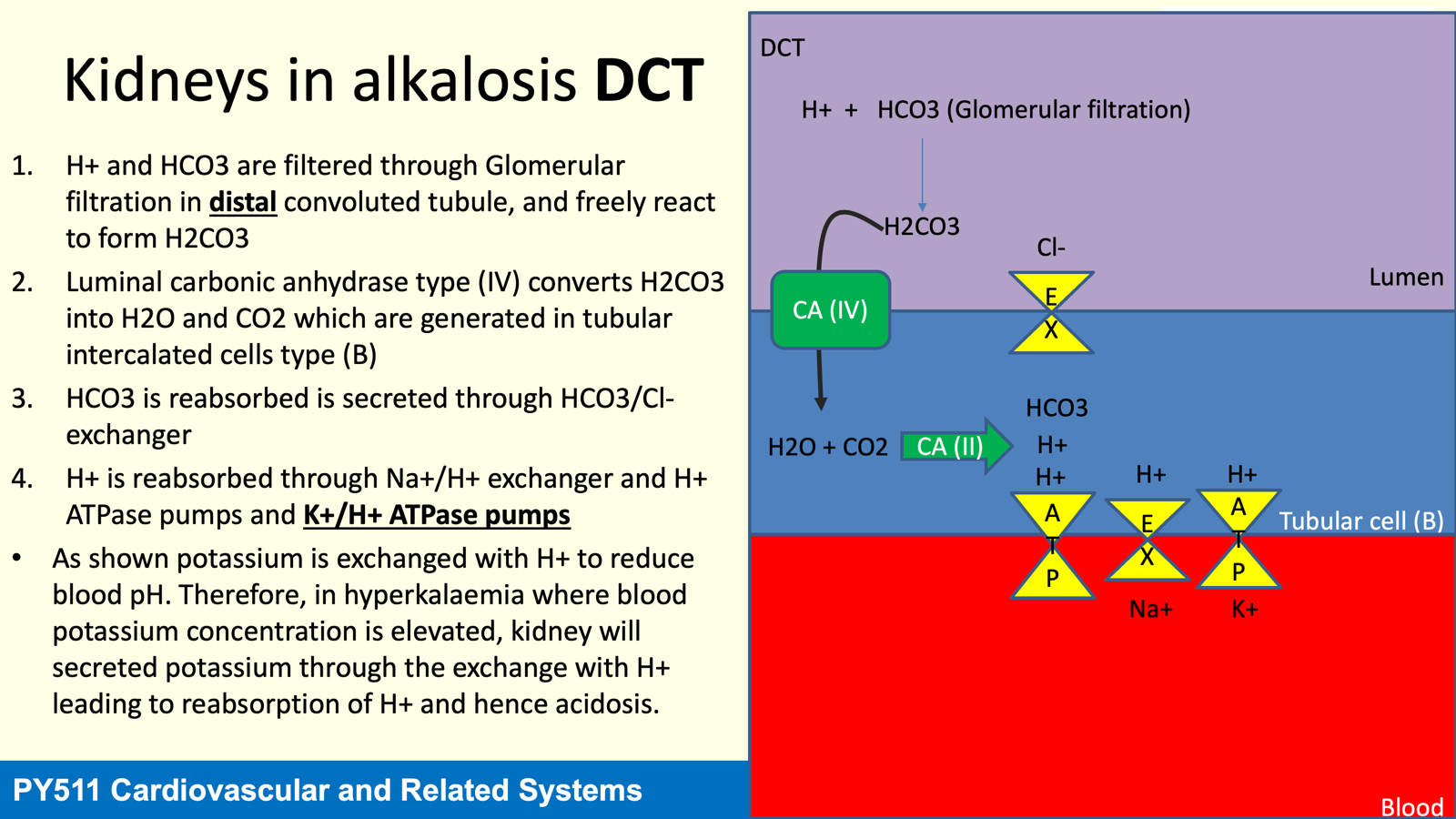
Most HCO3- in the filtrate is reabsorbed, and H+ is secreted maintaining urine pH around 6.0.
Renal tubular acidosis (RTA) can occur in renal failure due to ineffective HCO3- reabsorption.
In acidaemia (fall in ECF pH), there is increased secretion of H+ and reabsorption of HCO3- to elevate pH.
When urine pH falls below 4.5, H+ secretion is inhibited.
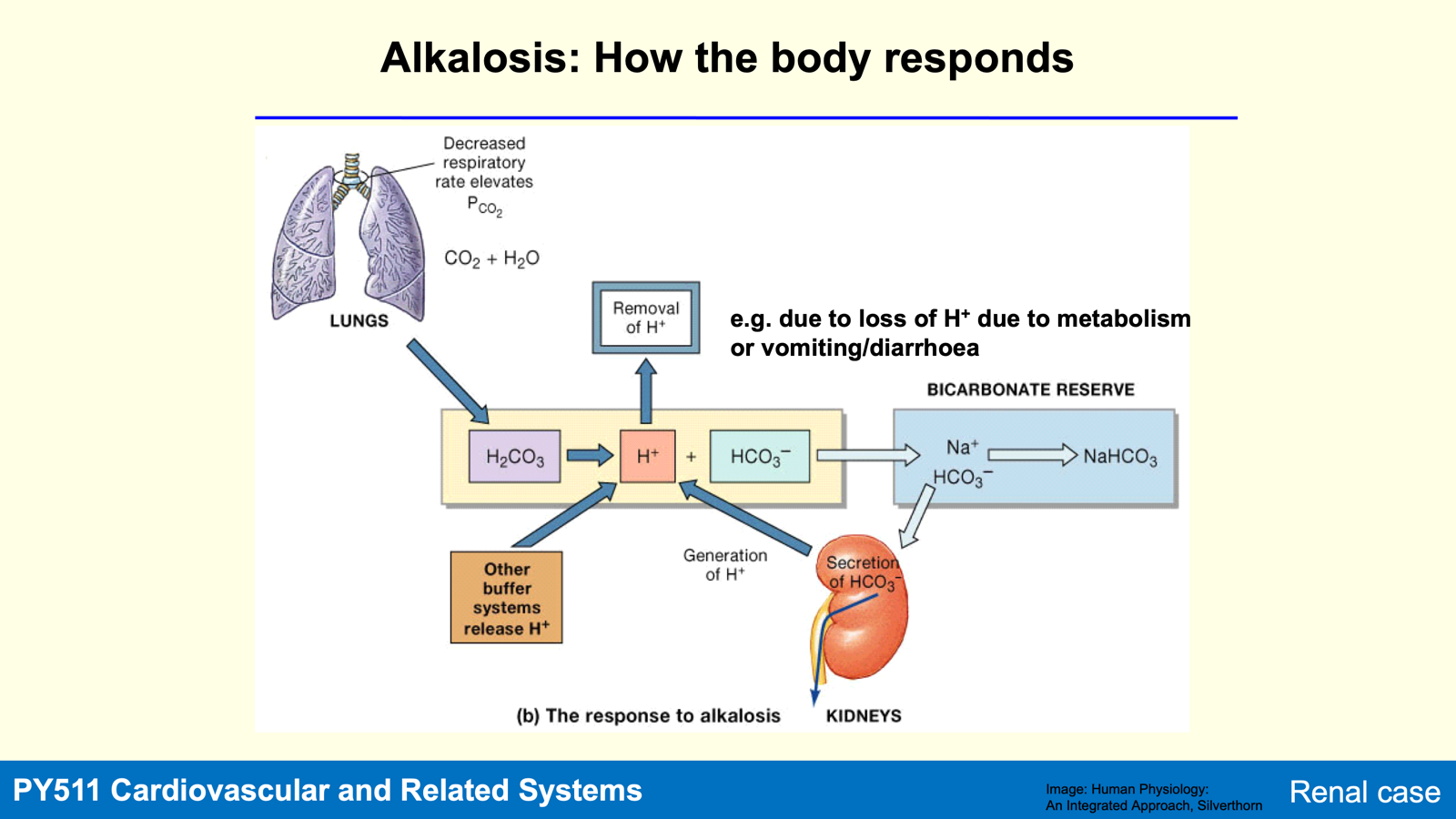
In alkalaemia (rise in ECF pH), both H+ secretion and HCO3- reabsorption decrease, decreasing extracellular pH.
Acidosis and Alkalosis Definitions
Acidosis: pH of body fluids falls below 7.35 due to:
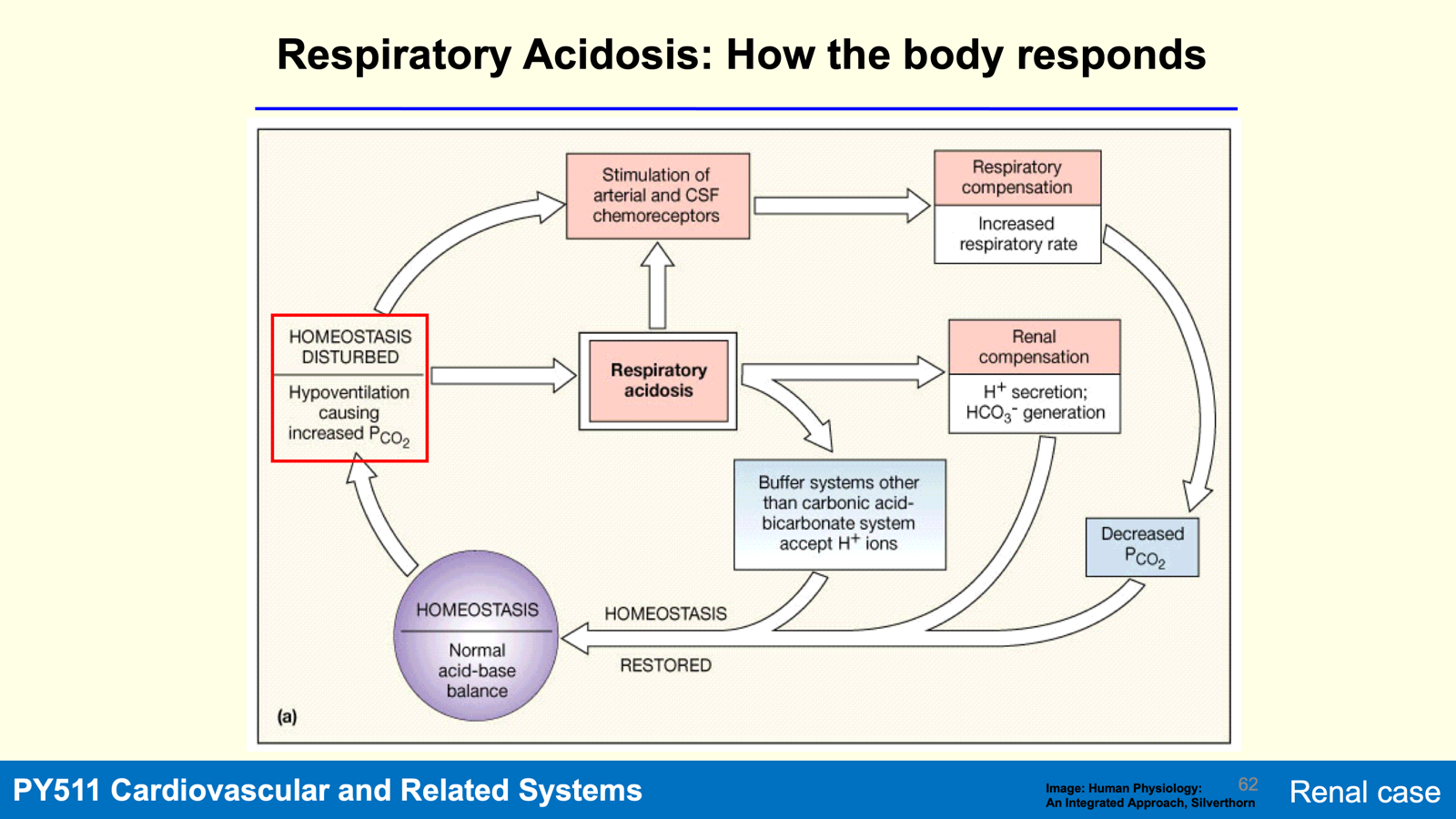
Respiratory Acidosis: Caused by inadequate ventilation (acute/chronic).
Metabolic Acidosis: Results from non-respiratory conditions decreasing pH (always chronic).
Causes for increased H+:
CO2 production from metabolism (H2CO3 formation).
Non-volatile acids like lactic acid from metabolism.
Loss of HCO3- through diarrhea or urine.
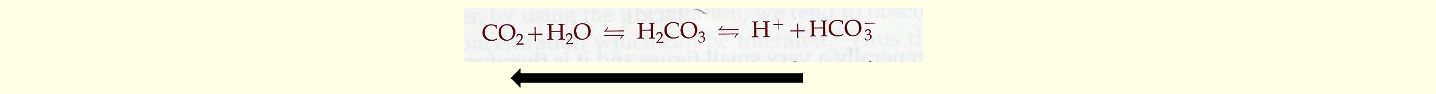
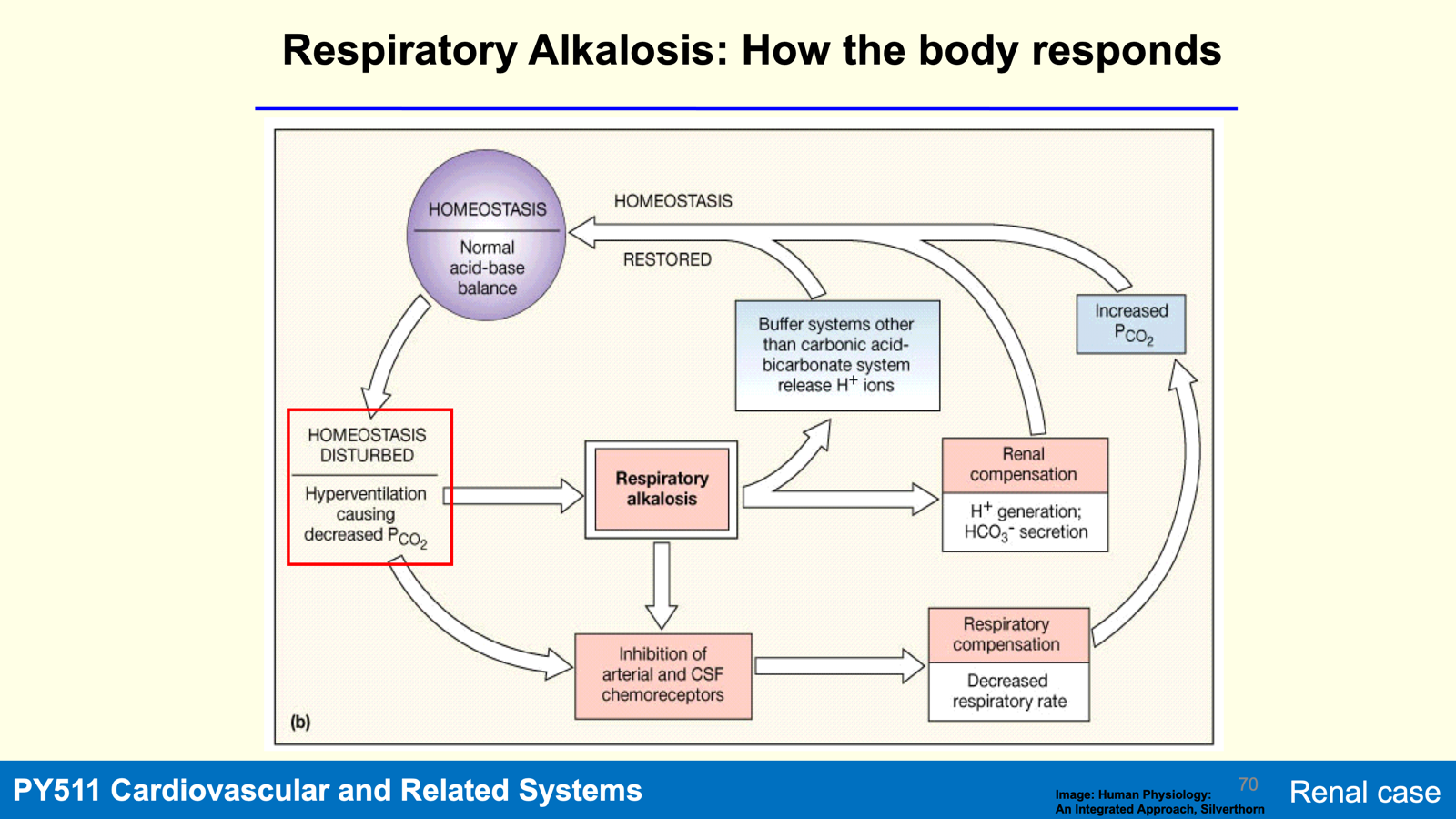
Alkalosis: pH of body fluids exceeds 7.45 due to:
Respiratory: Often from hyperventilation (acute/chronic).
Metabolic: Non-respiratory conditions causing increased pH (also chronic).
Causes for losing H+:
Using H+ during metabolism of organic anions.
Loss of H+ via vomiting.
Loss of H+ in urine or via hyperventilation.
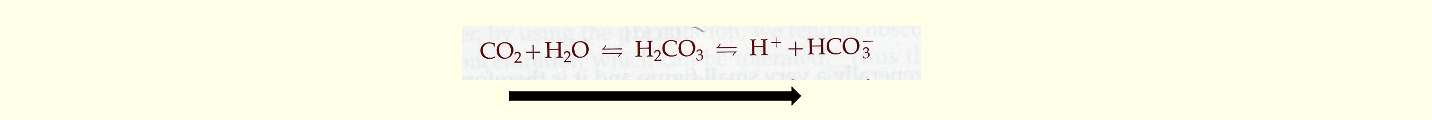
Treatments for Acidosis and Alkalosis
Respiratory Acidosis Treatment:
Restore ventilation.
Treat underlying conditions.
IV lactate solutions to be converted to HCO3- buffer.
Metabolic Acidosis Treatment:
Give IV isotonic HCO3-.
Administer IV lactate (like Hartmann’s solution) converting to HCO3-.
Alkalosis Treatments:
Respiratory alkalosis: address underlying cause, breathe into a paper bag to increase pCO2, IV chloride (Cl-) to reduce HCO3- excretion.
Metabolic alkalosis: provide electrolytes, and administer IV Cl-.
Clinical Cases and Compensatory Mechanisms
Uncontrolled diabetes: Leads to ketoacidosis with compensatory hyperventilation and H+ excretion by the kidneys.
Severe exercise: Can lead to lactic acidosis requiring similar kidney compensatory response.
Vomiting/diarrhea: Causes volume depletion and metabolic alkalosis, compensatory hypoventilation to preserve CO2 by the kidneys.
Panic attack: Hyperventilation leads to respiratory alkalosis requiring hypoventilation response and kidney adjustment.
Opioid overdose/COPD: Results in respiratory acidosis due to hypoventilation; compensatory mechanisms increase H+ secretion and bicarbonate reabsorption.
Summary of Lecture Points
Understanding of the kidney's role in regulating blood volume and acid-base balance.
Knowledge of cellular mediators in these processes and their functions.
Able to discuss modifications in H+ and HCO3- absorption relative to states of acidosis or alkalosis.
Example MCQ
Question: Which enzyme located in the kidney helps to regulate the balance between carbon dioxide (CO2) and hydrogen ions (H+) in the control of acid-base balance?
a) Adenylate cyclase
b) Carbonic anhydrase (CHECK)
c) Creatine kinase
d) Guanylate cyclase
e) Na+K+ATPase
Example SAQ
Question: Explain how prolonged vomiting alters the body’s acid-base balance and describe the kidney's compensatory response. List two treatment methods to normalize this disruption in acid-base balance from vomiting. (5 marks)