Unit 1:Matter
Isotopic Abundance - Week 2
Isotopes are atoms of the same element that have the same number of protons and electrons but different number neutrons - Will change atomic mass
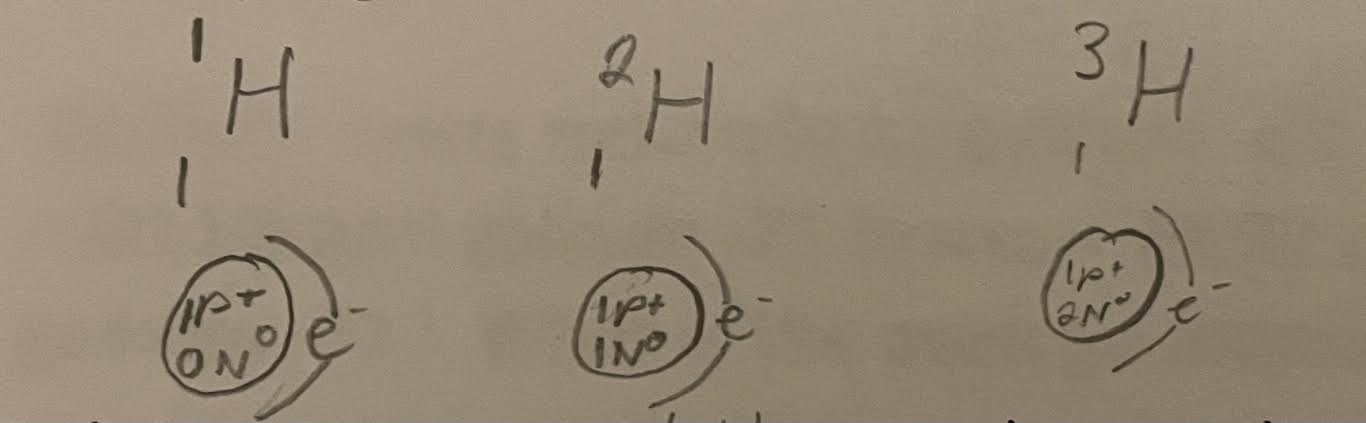
Radioisotopes are unstable because the neutrons are not properly balanced with the number of protons. This instability causes them to decay into a nuvleus that is more stable. This decay results in an atom with one more proton and one less neutron.
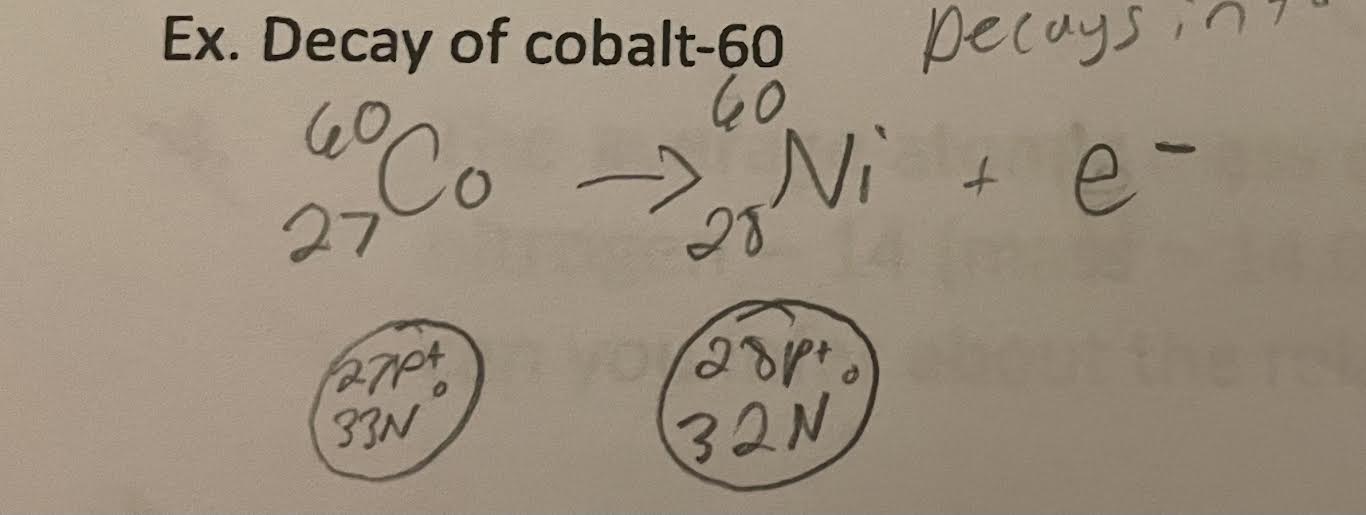
Calculating Average Atomic Mass and Isotopic Abundance
The mass of atoms is measured in atomic mass units (a.u). One atomic mass unit is defined at 1/12 of the mass of a 12C atom and is equivalent to 1.66×10^-24 g.
1 a.u - 1.66 × 10^-24 g
Many elemtns have two or more naturally occuring isotopes. The average mass is calculated using the relative abundance of each isotope.
Formula: Average Mass = (the sum of) %isotope/100 x mass of isotope
The closer the mass of the isotope is to the element the more abundant it is.
Periodic Trends - Week 3
Periodic Law
When elements are arranged by atomic number their chemical and physical properties recur periodically.
Horizontal Rows are called periods. Period number equals the number of energy levels.
Vertical columns are called groups. Group number equals the number of valence electrons.
Periodic Trends
Atomic Radius
Atomic radius decreases from left to right across a period as the postive force (number of protons) of the nucleus increases - Stronger nucleus pulls electrons closer
Atomic radius increases down a group as the number of energy levels increases.
Ionic Radius
Postively charged ions, called cations, are always smaller then the neutral atom from which they are formed.
Negetively charged ions, called anions, are always bigger than the neutral atom from which the are formed
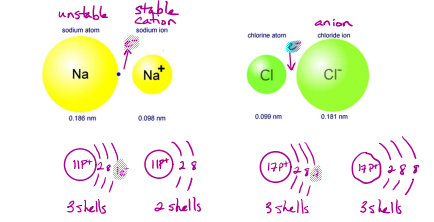
Shielding Effect - As the electrons are pulled towards the nucleus they are also trying to stay away from eachother (same charge), when another electron is added shielding happens and they spread further
Ionization Energy Trend - The amount of energy it takes to remove an electron
Ionization Energy increases from left to right across a period as the postive force of the nucleus increases, making it more difficult to remove an electron - Stronger attraction
Ionization Energy decreases down a group as the number of energy levels increases, making it easier to remove an electron - Further from nucleus means weaker attraction
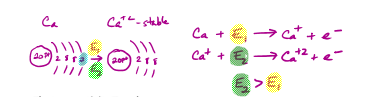
Requires more energy to get a second or more electrons because the shell gets smaller after removing one - Getting closer to the nucleus causing a stronger attraction
Electronegativity Trend - Ability to attract electrons
Electronegativity increases from left to right across a period as the postive force of the nucleus increases.
Electronegativity decreases down a group as the number of energy levels increases.
Metals have low electronegatibity because their shells are less then half full meaning they are trying to lose electrons.
Non-metals have high electronegativity because their shells are more then half full meaning they are trying to gain electrons.
Noble gases lack electronegativity because their shells are full, meaning they are stable and wont attract electrons.
Electron Affinity - The amount of energy gained or lost when an electron is added
Electron Affinity increases from left to right across a period because they are closer to stability
Electron Affinity decreases down a group because of the size increase - New electrons are further from the nucleus
Noble gases have maximum electron affinity because they are stable - Their electrons are tightly bonded to the nucleus
Classifying Compounds - Week 3
Chemical Bonds
Occur when valence electrons of 2 or more atoms interact
Compunds are more stable than individual atoms they are produced from
There are two types of bonds: Ionic (metal+non-metal) and covalent/molecular (non-metal+non-metal
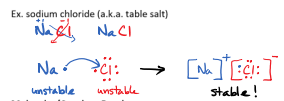
Ionic Bonds
Usually occurs between a metal and a non-metal
The metal loses electrons to the non-metals
Electrons are tranfered (not shared)
Molecular/Covalent Bonds
Usually occurs between 2 non-metals
Electrons are shared (not transfered)
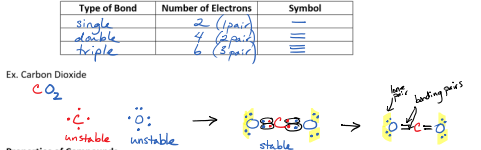
Properties of Compounds

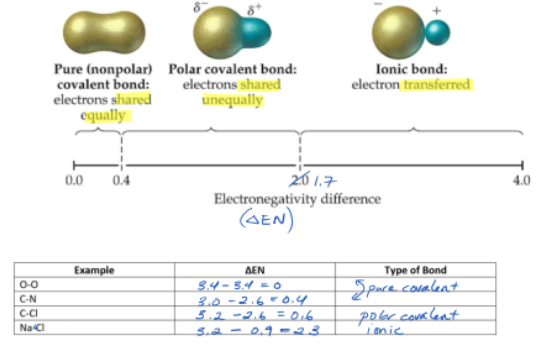
Electronegativity (EN)
Electronegativity is the relative ability of an atom to attract shared electrons. The smaller the atom, the greater the electronegativity
Electrons will spend more time around the atoms with the greater electronegativity
The greek letter s (curled end), is used to represent the slight/partial charges of atoms in a covalent bond
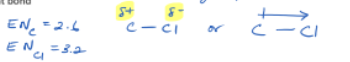
Electronegativity values can be used to determine the type of bond present in a compound
Molecular Shapes and Polarity - Week 4
The distance between electrons in a molecule must be mazimized because electrons are repelled by one another. This affects the molecules shape.
There are two types of electrons:
Electron that are shared between atoms are called bonding pairs
Electrons that are not shared are called lone pairs
Basic Molecular Shapes
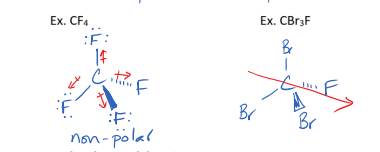
I. Linear Molecules
have no lone pairs on the central atom
have a bond angle of 180 degrees/straight line
can be polar or non-polar depending on their symmmetry
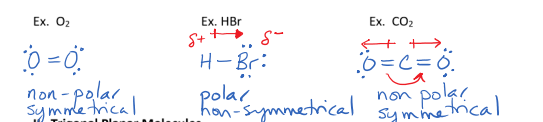
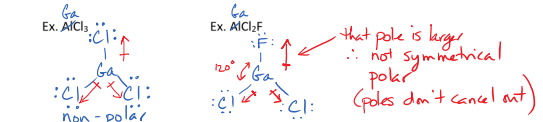
II. Trigonal Planar Molecules
have no lone pairs on central atom
3 bonds
have a bond angle of 120 degrees
can be polar or non polar depending on their symmetry
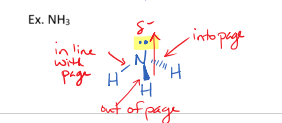
III. Trigonal Pyramidal Molecules
have one lone pair on the central atom
3 bonds
have a bond angle of less than 120 degrees
are always polar because they are non-symmetrical
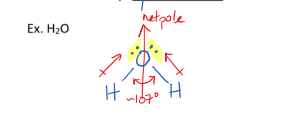
IV. Angular/Bent/V shaped Molecules
have one or two lone pairs on the central atom
have a bond angle of 109 degrees
are always polar because they are non-symmetrical
V. Tetrehedral Molecules
have zero lone pairs on the central atom
4 bonds
have a bond pair of 109 degrees (approx)
can be non-polar or polar depending on their symmetry