Chemistry Unit 4
atoms form bonds to change their number of valence electrons
the type of bond shows how valence electrons are changed
valence electrons are used to connect atoms
compounds are what we interact with daily
bonds that make up a compound give it special properties
metals form different bonds to metals than non metals (vise versa is true)
polyatomic ions are examples of ionic and covalent bonding
these charged particles are created from more than one element sharing electrons through covalent bonding but functions as an ion in ionic bonding/compounds
PAI’s are made from combining nonmetals (generally)
most PAIs contain oxygen atoms in addition to another non metal (oxyanions)
most are negatively charged - more electrons than protons
changing the amount of oxygen atoms creates a new PAI with a new name but the same charge

Nick the Camel Craves a Clam, a Aspirin, a Brew, for Supper in Phoenix with Morgan
Polyatomic Ions:
Chlorine (Cl)
Perchlorate: ClO4(−)
Chlorate: ClO3(−)
Chlorite: ClO2(−)
Hypochlorite: ClO(−)
Bromine (Br)
Perbromate: BrO4(−)
Bromate: BrO3(−)
Bromite: BrO2(−)
Hypobromite: BrO(−)
Iodine (I)
Periodate: IO4(−)
Iodate: IO3(−)
Iodite: IO2(−)
Hypoiodite: IO(−)
Nitrogen (N)
Nitrate: NO3(−)
Nitrite: NO2(−)
Carbon (C)
Carbonate: CO3(2-)
Sulfur (S)
Sulfate: SO4(2−)
Sulfite: SO3(2−)
Selenium (Se)
Selenate: SeO4(2−)
Selenite: SeO3(2−)
Phosphorus (P)
Phosphate: PO4(3−)
Phosphite: PO3(3−)
Arsenic (As)
Arsenate: AsO4(3−)
Arsenite: AsO3(3−)
Chromium (Cr)
Dichromate: Cr2O7(2−)
Chromate: CrO4(2−)
Manganese (Mn)
Permanganate: MnO4(−)
Organic Anions
Acetate: C2H3O2(−)
Formate: CHO2(−)
Oxalate: C2O42(−)
Special Ions
Cyanate: NCO(−)
Thiocyanate: SCN(−)
Thiosulfate: S2O32(−)
Peroxymonosulfate: SO5(2-)
Peroxydisulfate: S2O5(2-)
a lasting attraction between atoms, ions, and molecules allows the formation of chemical compounds
only valence electrons are involved in bonding
the bond may be caused from the coulombic force of attraction between oppositely charges ions through ionic bonds or the sharing of electrons
the strength of compounds can vary:
strong bonds need a large amount of input before breaking apart
lattice energy for ionic compounds
bond energy for covalent compounds
weak bonds need a small amount of input before breaking apart
atoms form electron bonds to create more stable electron shells
the type of bonds maximizes the stability of atoms inside it
an ionic bond is made through electron transfer
one atoms donates an electron to another, one becomes stable by usually filling their valence electron shell and the other empties their outer shell
usually between a metal and a nonmetal atoms
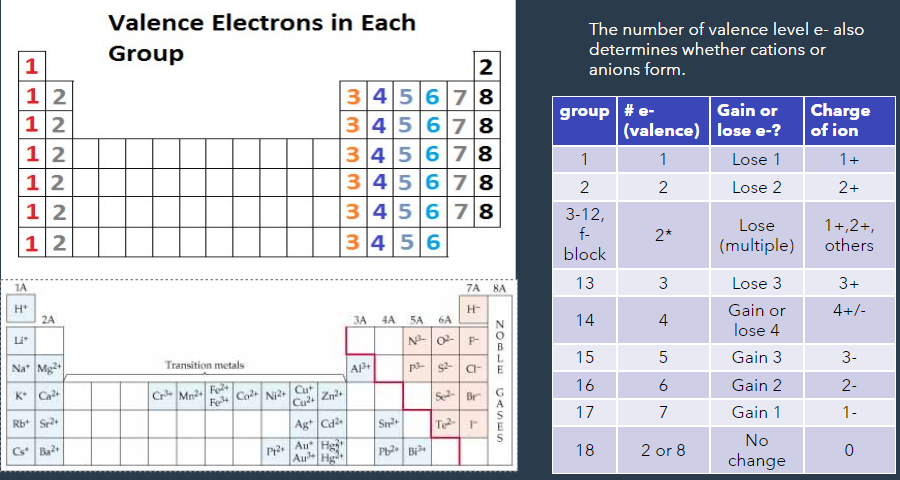
cations - positive ion
number of protons > number of electrons
created by loosing electrons
metals make cations
anions - negative ions
number of protons < number of electrons
- created by gaining electrons
nonmetals make anions
ions stay together due to electrostate attraction
ionic bonds NEVER for molecules they form formula units
ionic bonds form easily between alkali metals and halogens
all ionic compounds are electrically neutral
a covalent bond is made from sharing electrons
between 2+ nonmetals atoms and results in high stability
between nonmetal atoms
H and nonmetal = binary acit
H and PAI = ternart/oxy acitd
two nonmetals = binary molecule
called “molecules” or “covalent compounds”
can exist independently, and as discrete particles
have a low melting point and boiling point
can be found as gases, liquids, or solids at room temparature
⭐covalent compounds names and formulas are literal interpretations of each other⭐
a metallic bond is made when metal atoms jointly make a sea of delocalized electrons, allowing individual atoms to give up their outer shells, throughout a metal sample
⭐ increased stability means holding each bonding particle exists at a lower energy state ⭐
noble gases are the most stable/non reactive elements that exist
most have 8 valence electrons, that take up all their s & p orbitals
octet of electrons consists of FULL s & p orbitals
⭐ Octet Rule Defined atoms gain, lose, or share electrons to achieve the electron configuration of the nearest noble gas ⭐
the octet rule refers to the tendency of atoms to prefer having eight electrons in their valence shell.
atoms with fewer than eight electrons tend to react and form more stable compounds.
a complete octet is highly stable because all orbitals are full.
reactions that increase stability release energy (heat or light), while reactions that decrease stability absorb energy (cooling).
the octet rule:
only involves s and p electrons, excluding d and f electrons.
applies to main group elements (not transition or inner-transition metals).
an octet corresponds to electron configurations ending in s2p6s^2p^6s2p6.
pseudo-octets:
occur in transition metals and f-block metals.
these elements may have 18 electrons in the outer shell instead of 8, due to filled d orbitals.
a pseudo-noble gas configuration appears as s2p6d10 instead of s2p6
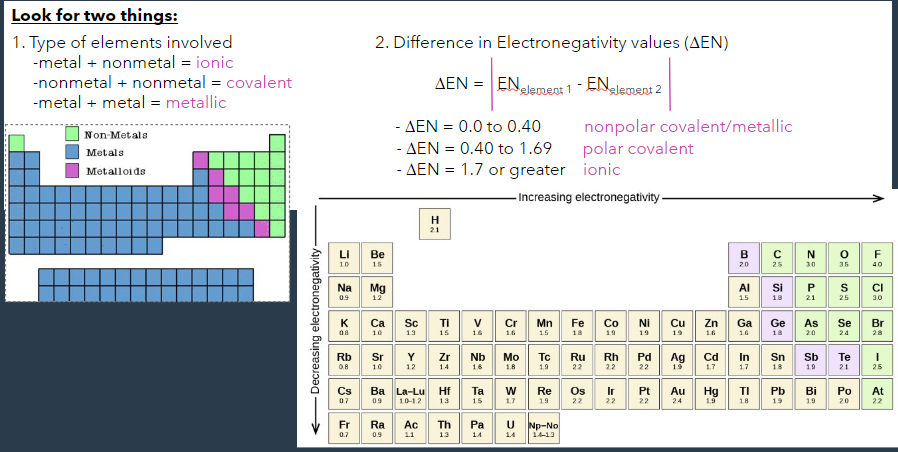
identification of bonds:
look for the type of elements involved:
- metal + nonmetal = ionic
- nonmetal + nonmetal = covalent
- metal + metal = metallicfind the difference in the electronegativity values
- change in EN = 0.0 to 0.4 → nonpolar covalent/metallic
- change in EN = 0.4 to 1.69 → polar covalent
- change in EN = 1.7+ → ionic
binary compounds - made from two monatomic ions
monatomic means ions from two elements are used
does not contain PAIs
two capital letters in the formula
no parentheses in the formula
name sounds like it is readily identifiable
write the names of the least EN element with a prefix to indicated the # of atoms of the element that are present
weite the name of the most EN element second with a prefix to indicate the number of atoms of the element that are present
suffix -ide is added to the end of the second element
ternary compounds - made from at least one PAI
usually the PAI is the anion
the only positive PAI is ammonium
has more than two capitalized letters in the formula
might have parentheses in the formula
some part of the name doesnt sound like an element name
the second word ends in -ate or -ite
ions form due to:
ionization energy (↑ across the groups, and ↓ across the periods)
|electron affinity| (↑ across the groups, and ↓ across the periods)
electronegativity (↑ across the groups, and ↓ across the periods)
when IE and EN values are the highest, the EA value is the most negative and the element will GAIN electrons (non metals)
when IE and EN are the highest, the EA value is the more positive and the element will LOSE electrons (metals)
ionic compounds contain both cations and anions in a ratio that makes no net electrical charge
sometimes more than one ion of each type is needed to create a neutral compound
positive and negative charges must cancel out for a compound to be neutral
the number and type of ions used to create a compound can be written more condensed in a chemical formula
metals/cations/lesser EN elements are written first in the formula
nonmetals/anions/more EN elements are written last in the formula
a chemical formula presents information abouyt the identity and the number of atoms that mkae up a particular chemical compount, molecule, or formula unit
chemical formulas are created using chemical element symbols, numbers, and sometimes parenthese, dashes, brackets, commas, and plus and minus signs (for charges)
⭐ for ALL chemical formulas the less EN element is written first and the most EN element is written last ⭐
if only one atom of a specific type is present, there is no subscript
if there are 2+ of a specific type of atom present, the subscript is written after the symbol for that atom
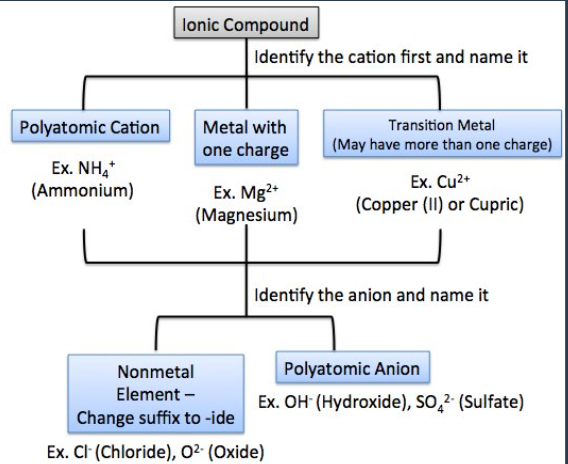
Steps for Naming Ionic Compounds (with formula):
Identify the cation by its name.
If necessary, add a Roman numeral in parentheses (for cations that form multiple positive ions).
Identify the anion by its name.
Replace the ending of the element name with "-ide" or use the appropriate PAI (polyatomic ion) suffix.
Steps to Create the Formula for Ionic Compounds (with name):
Write the symbol of the cation with its positive charge on the left.
Write the symbol of the anion with its negative charge on the right.
Add subscripts for each ion to balance the charges (ensure all positive and negative charges cancel).
Rewrite the chemical formula so that:
All superscript charges disappear.
The subscripts reflect the lowest ratio possible.
For compounds with polyatomic ions, use parentheses if more than one polyatomic ion is needed to balance the charge.
Formulas for Ionic Compounds:
identify the cation and write down the symbol and charge
identify the anion and write down its symbol and charge
combing the 2 ions to form an electrically neutral compound (proton charge and electron charge = 0)
use the cross and drop methods
all ionic compounds are written in empirical formulas, meaning a chemical compound in the simplest whole number ratio of atoms present in a compound

polyatomic ions are ions of more than one atom, they are covalently bonded together and become one single charge unit
cations are written first in the name, and the anion is written second in the name
when the formula unit contains 2+ of the same polyatomic ion, the ion is written in parentheses and the quantity is written as a subscript outside of the parentheses
⭐ parentheses and a subscript are NOT used unless more than one of a PAI is present in the formula unit ⭐
transition metals use roman numerals to show the magnitude of the positive charge on the cation