Untitled Flashcards Set
An Arrhenius acid (HA) is a compound that dissolves in water and ionizes into protons (H+) and an anion (A-). The following is a generic ionization in water (hydrolysis) of monoprotic acids, which have one stoichiometric equivalent of protons to acid:
HA(aq)→H+(aq)+A−(aq)HA(aq)→H+(aq)+A−(aq)
Equation 14.1
Common monoprotic acids include nitric acid (HNO3) and the haloacids HF, HCl, HBr, and HI. Polyprotic acids have more than one stoichiometric equivalent of protons. These include the triprotic phosphoric acid (H3PO4), the diprotic sulfuric acid (H2SO4) and carbonic acid (H2CO3). Triprotic acids like phosphoric acid can donate three protons while diprotic acids can donate two protons. The hydrolysis of polyprotic acids is treated differently than that of monoprotic acids, as will be discussed in detail in Section 14.11.
Arrhenius bases dissolve in water and dissociate into a cation and hydroxide (OH-).
Group I metal hydroxides dissociate in water according to:
MOH(aq)→M+(aq)+OH−(aq)MOH(aq)→M+(aq)+OH−(aq)
Equation 14.2
Most Group II metal hydroxides also dissociate completely:
M(OH)2(aq)→M2+(aq)+2 OH−(aq)M(OH)2(aq)→M2+(aq)+2OH−(aq)
The Arrhenius interpretation of acids and bases is limited as the role of water is not considered. Because acid and base ionizations are actually reversible in water, we have to consider these systems as aqueous equilibria. Hence, an alternate interpretation is warranted.
14.3 Brønsted–Lowry Acids and Bases
14.3.1 Definitions
Brønsted-Lowry acids are defined as proton (H+) donors, whereas Brønsted-Lowry bases are defined as proton (H+) acceptors. Since acid/base reactions often occur in water, we will focus our discussion on Brønsted-Lowry acid/base behavior in aqueous systems. A Brønsted–Lowry acid ionizes to donate a proton to water (Equation 14.3, Figure 14.3):
HA(aq)+H2O(l)⇌A−(aq)+H3O+(aq)HA(aq)+H2O(l)⇌A−(aq)+H3O+(aq)
Equation 14.3
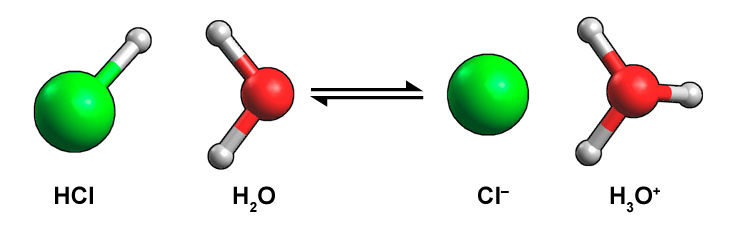
Figure 14.3 Ball and stick model of the ionization of HCl and H2O to donate a proton to water. Brønsted-Lowry acids are referred to as "proton donors". Green spheres represent Cl atoms, red represent oxygen atoms, and grey represent hydrogen atoms.
A molecule of hydrogen chloride reacts with a molecule of water to form a chlorine ion and a hydronium ion. It’s a reversible reaction. A large green sphere attached to a small white sphere depicts hydrogen chloride. A large red sphere attached to two small white spheres depicts a water molecule. A large green sphere depicts a chlorine ion. A large red sphere attached to three small white spheres depicts a hydronium ion.
When water accepts the acid’s proton, H2O becomes the hydronium cation H3O+. Any time you notice hydronium, a Brønsted–Lowry acid is present.
Brønsted–Lowry bases accept a proton from water. A typical example is ammonia, NH3 (Figure 14.4):
NH3(aq)+H2O(l)⇌NH4+(aq)+OH−(aq)NH3(aq)+H2O(l)⇌NH4+(aq)+OH−(aq)
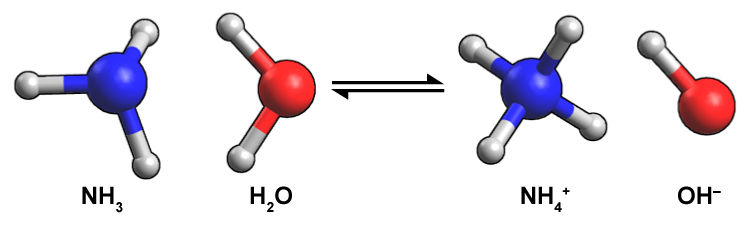
Figure 14.4 Ammonia (NH3) accepts a proton from water to become NH4+. After donating a proton, H2O becomes OH-, which makes the solution basic. Blue spheres represent nitrogen atoms, red represent oxygen atoms, and grey represent hydrogen atoms.
A molecule of ammonia reacts with a water molecule to form an ammonium ion and a hydroxide ion. This reaction is reversible. A large blue sphere attached to three small white spheres depicts ammonia molecule. A large red sphere attached to two small white spheres depicts a water molecule. A large blue sphere attached to four small white spheres represents ammonium ion. A large red sphere attached to a small white sphere represents a hydroxide ion.
Notice that this time water is the proton donor, forming hydroxide, OH-. Any time you find hydroxide, a Brønsted–Lowry base is present.
In general, the equilibrium of a weak base with water can be written as:
B(aq)+H2O(l)⇌BH+(aq)+OH−(aq)B(aq)+H2O(l)⇌BH+(aq)+OH−(aq)
Equation 14.4
The Brønsted–Lowry interpretation of acids and bases highlights the important role of water. Notice that in the HCl example, water accepted a proton. When NH3 was in solution, water donated a proton.
14.3.2 The Amphiprotic Nature of Water
Water is an example of an amphiprotic molecule, which can function as both a Brønsted–Lowry acid and as a Brønsted–Lowry base. When a Brønsted–Lowry acid ionizes in water, hydronium forms.
An interesting situation arises when we study the reaction of water with itself. This process, known as the autoionization of water, has one water molecule functioning as the acid and the second functioning as the base:
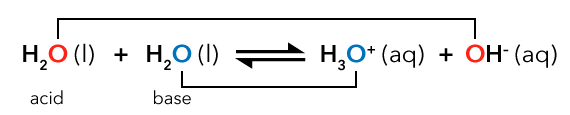
Equation 14.5
An acid, H 2 O (liquid) and a base H 2 O (liquid) are in equilibrium with H 3 O positive (aqueous) and O H negative (aqueous). The oxygen atoms in the acid H 2 O and O H negative are in red. The oxygen atoms in H 2 O base and H 3 O positive are in blue.
In Equation 14.5 above, to keep track of oxygen atoms, the O of the acid has been highlighted red, and the O of the base blue. The oxygen of the acid has lost a proton, becoming OH-, while the oxygen of the base has gained a proton, becoming H3O+.
Figure 14.5 shows this process as a ball and stick model; keep in mind that the molecular geometry of the H3O+ ion is trigonal pyramidal. Notice the equilibrium arrow, suggesting that this process is reversible. Let’s write the expression for the equilibrium constant.
At 25°C, the equilibrium constant is equal to 1.0 × 10−14 and is known as Kw, the autoionization or ion-product constant of pure water:
Kw=[H3O+]eq[OH−]eq=1.0×10−14 at 25∘CKw=[H3O+]eq[OH−]eq=1.0×10−14 at 25∘C
The small value of Kw suggests that the concentrations of hydronium and hydroxide ions in pure water are relatively small compared with the concentration of water itself at 25°C being 55.5 M (consult Chapter 11: Solutions). Physically, there is no such thing as 100% ion-free water. In the lab, deionized water is used for aqueous reactions. This type of water has had any traces of salts removed, especially Group I, Group II, and Group VII ions. However, hydronium and hydroxide are always present, just not in any appreciable amounts. In fact, when a conductivity test (refer to Chapter 7: Reactions in Aqueous Solutions) is run on tap water, a current is generated, but not enough to illuminate a light bulb.
This Kw equation (Equation 14.6) sheds light on what is meant by an acidic, basic, or neutral solution. For acidic solutions, [H3O+]eq > [OH-]eq. For basic solutions, [H3O+]eq < [OH-]eq. Finally, for neutral solutions, [H3O+]eq = [OH-]eq = 1.0 × 10−7 M.
Like amphiprotic compounds, which can act as either acids or bases, amphoteric compounds are capable of reacting with both acids and bases. In this section, we’ll focus solely on amphoteric oxides. For example, consider lead (II) oxide, PbO, reacting with hydrochloric acid:
PbO(s)+2HCl(aq)→PbCl2( s)+H2O(l)PbO(s)+2HCl(aq)→PbCl2(s)+H2O(l)
Lead(II) oxide reacts with hydrochloric acid to form a yellow precipitate of lead(II) chloride. This is a double-displacement reaction (Chapter 7). PbO also reacts with an aqueous base:
PbO(s)+KOH(aq)→KHPbO2(aq)PbO(s)+KOH(aq)→KHPbO2(aq)
This reaction with aqueous KOH forms the potassium salt of plumbite. As shown in Figure 14.6, Elements that form amphoteric oxides include
Main Group elements located on the periodic table close to and including some semimetals.
Many transition elements, including all Period 4 transition metals.
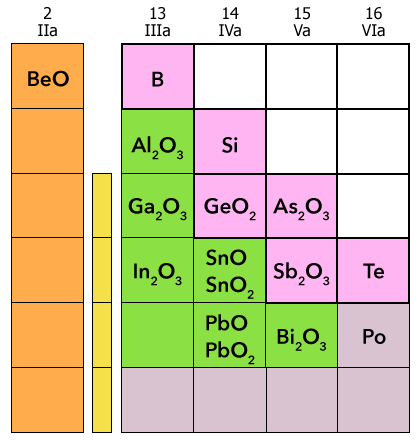
Figure 14.6 Main Group elements close to and including some semimetals that form amphoteric oxides.
A part of the periodic table lists main group elements and their oxides. Group 2 family 2a lists Be O, orange square. Group 13 family 3 a lists B, pink square; Al 2 O 3, green square; Ga 2 O 3, green square; In 2 O 3, green square. Group 14 family 4 a lists Si, pink square; Ge O 2, pink square; Sn O, Sn O 2, green square; Pb O, and Pb O 2 Green square. Group 15 family 5a lists As 2 O 3, pink square; Sb 2 O 3, pink square; and Bi 2 O 3, pale purple square. Group 16 family 6a lists Te pink square and Po pale purple square.
The terms amphoteric and amphiprotic can be confusing—both types of molecules are reactive with both acids and bases! Amphoteric is the more general term, whereas amphiprotic is specific to the transfer of protons. For example, H2O is considered to be both amphiprotic and amphoteric. However, PbO is considered to be only amphoteric. It is not amphiprotic because it does not contain protons to donate.
We can, therefore, conclude that all amphiprotic molecules are also amphoteric, but the converse is not necessarily true. Not all amphoteric molecules are amphiprotic.
In addition to being able to accept a proton to form H2S, HS- contains a proton to donate and is also amphiprotic. We’ll discuss amphiprotic salts later in this chapter.
Problem Solving Strategies: Tips for identifying acids and bases
An essential skill is to identify if a molecule is an acid or base from its chemical formula.
Acids usually have a hydrogen atom attached to an electronegative element (O, S, F, Cl, Br, I).
Commonly encountered bases are amines and metal hydroxides.
Water is an amphiprotic compound - it can donate or accept a proton.
Pure water autoionizes into OH- and H3O+ to a small extent (Kw = 1.0×10-14)
Both the Arrhenius and Brønsted–Lowry interpretations of an acid and a base are very specific: they both focus on the transfer of a proton in water.
However, it can be convenient to interpret acids and bases very generally: whether they donate or accept electrons. A Lewis acid is any atom or molecule or ion that accepts a pair of electrons, whereas a Lewis base donates a pair of electrons in a chemical reaction. For example, consider the hydrolysis of HBr:
HBr(aq)+H2O(l)→Br−(aq)+H3O+(aq)HBr(aq)+H2O(l)→Br−(aq)+H3O+(aq)
Let’s examine the Lewis structure of all reactants and products (Figure 14.7) so that we can monitor the transfer of electrons:
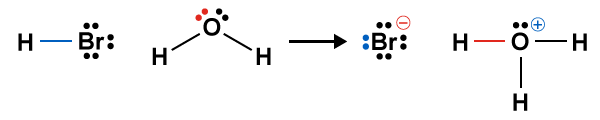
Figure 14.7 Electron transfer during hydrolysis of hydrobromic acid (HBr). The bromine atom in HBr gains a lone pair while the oxygen atom of water donates a lone pair to form a covalent bond with a proton.
Expanded structures for the hydrolysis of hydrobromic acid. In the reaction HBr and water react to produce a bromide ion, Br minus, and a hydronium ion, H3O plus.
The terms Lewis acid and Lewis base always refer to either of the two reactants, and not to the products. Let’s start off by examining HBr before and after the reaction. It appears that bromine, which started off with three lone pairs, has gained an extra lone pair to become bromide. This makes HBr the Lewis acid. The oxygen atom in water started off with two lone pairs, but ends up with only one lone pair in hydronium. The lone pair in the oxygen atom went on to form a σ bond with the hydrogen. This makes water the Lewis base. In fact, the following statement can be said of Brønsted–Lowry acids and bases: all Brønsted–Lowry acids are also Lewis acids, and all Brønsted–Lowry bases are also Lewis bases.
However, as we are about to explore, the converse statement is not necessarily true. Consider the following reaction in Figure 14.8:

Figure 14.8 Electron transfer during reaction of a carbocation (a short-lived, highly reactive carbon atom with six valence electrons) with bromide. Br- donates a lone electron pair to carbon to form a new covalent bond.
Expanded structures for the reaction of a carbocation with bromide. CH3 1 plus reacts with bromide 1 minus to produce methyl bromide or CH3Br.
The carbon in the cation, CH3+, has six electrons around it. After the reaction is complete, the carbon has gained two more electrons. This makes the reactant CH3+ a Lewis acid. Bromide, with four lone pairs, ends up in the product side with just three lone pairs: it has lost a lone pair. This makes the reactant bromide a Lewis base. Notice, however, that this reaction does not involve a transfer of a proton. Therefore, the Lewis acid CH3+ is not a Brønsted–Lowry acid and the Lewis base Br- is not a Brønsted–Lowry base in this reaction.
Common characteristics of Lewis acids:
Tend to be cations
May have less than an octet of electrons
Tend to be metals
Include all Brønsted–Lowry acids
Common characteristics of Lewis bases:
Tend to be anions
Tend to have nonbonding electrons
May have pi bonds
Tend to be nonmetals
May have an expanded octet
Include all Brønsted–Lowry bases
Lewis acids and bases are only introduced in this chapter as their main use will be in organic chemistry.
Question 14.8
Review