Chemistry Unit 2
atom – the smallest unit of any element that still maintains the identity of that element.
Postulates of Dalton’s Atomic Theory:
- Elements are composed of tiny particles called atoms. Atoms are indivisible. (We now know about protons, neutrons, and electrons. So, this part of his theory has been revised in modern atomic theory.)
- Atoms of the same element are identical in all properties (We now know about isotopes, so this part of his theory also had to be revised in modern atomic theory.), whereas atoms of a different element have different properties.
- Atoms can combine in whole number ratios to form compounds.
- Atoms are combined, separated, or rearranged in chemical reactions. In a chemical reaction, atoms of one element will remain atoms of that element. They will not change into atoms of another element.
Structure of the Nuclear Atom
Thomson - credited with the discovery of negatively charged subatomic particles called electrons using cathode rays - calculated the charge-to-mass ratio of electrons - used experimental data to determine that electrons must be present in all elements
Millikan - calculated the charge of an electron by ionizing and suspending oil droplets - used the charge of the electron and the charge-to-mass ratio of the electrons (determined by Thomson) to calculate the mass of the electron
Goldstein - credited with the discovery of positively charged subatomic protons in canal rays
Chadwick - credited with the discovery of neutral subatomic neutrons
Rutherford - discovered the positively charged nucleus of the atom by bombarding thin gold foil with alpha particles
Much of the mass of an atom is concentrated in a very small, dense nucleus. Most of the volume of an atom is the space outside of the nucleus where the electrons are located.
Distinguishing Among Atoms
atomic number (Z) = proton number = the identity of the element
mass number (A) = number of protons and neutrons in the nucleus
neutron number = mass number (A) – atomic number (Z)
isotopes – atoms of the same element with different numbers of neutrons
1 atomic mass unit (amu) = 1/12 the mass of a carbon-12 atom = the mass of about 1 proton = the mass of about 1 neutron
atomic mass (atomic weight) – a weighted average of an element that reflects the average mass of each naturally occurring isotope of that element and the relative abundance of each isotope
For example: Oxygen has three naturally occurring, stable isotopes. The first isotope has a mass of 15.995 amu and a relative abundance of 99.759%. The second isotope has a mass of 16.995 amu and a relative abundance of 0.037%. The third isotope has a mass of 17.999 amu and a relative abundance of 0.204%. The atomic mass would then be calculated as follows: (15.995)(0.99759) = 15.956452 (16.995)(0.00037) = 0.0062881 (17.999)(0.00204) = + 0.0367179 15.999458 or about 15.9994 amu
Mass spectra: A mass spectrum for gallium, would look like the figure below. Gallium has two naturally occurring stable isotopes. One isotope has a mass of 68.925581 amu and a natural abundance of 60.108 %. The other isotope has a mass of 70.924705 amu and a natural abundance of 39.892 %.
Models of the Atom
Bohr
– studied emission spectra and discovered that hydrogen always produced a distinct pattern, emitting light at only certain wavelengths
– reasoned that the electrons had measurable energy levels in fixed positions away from the nucleus
– hypothesized that electrons might move in circular orbits around the nucleus – reasoned that a specific amount of energy (quantum) would be required in order for an electron to move between energy levels
Schrodinger
– used mathematics and probability to describe how electrons must behave in an atom (this lead to quantum mechanics)
– realized that the electrons were not in orbiting the nucleus in circular paths; electron clouds of different shapes were better representations of electron positions
– defined atomic orbitals (s, p, d, f) that correspond to these shapes, areas of space where an electron may be found
– only two electrons may be found in any sublevel of an energy level:
- the first energy level only has one s orbital with up to two electrons
- the second energy level has one s orbital and three p orbitals for a total of four orbitals at this level each with up to two electrons
- the third energy level has one s orbital, three p orbitals, and five d orbitals for a total of nine orbitals each with up to two electrons
- the fourth energy level has one s orbital, three p orbitals, five d orbitals, and seven f orbitals for a total of sixteen orbitals each with up to two electrons
Electron Arrangement in Atoms Electron configurations:
1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 6s2 4f14 5d10 6p6 7s2 . . .
Aufbau principle – electrons in their ground state configuration will occupy lower energy orbitals (closer to the nucleus) first before occupying energy levels further from the nucleus
Pauli exclusion principle – only a maximum of two electrons can occupy any given orbital, each electron will have an opposite spin
Hund’s rule – electrons all occupying a given energy sublevel will have the same spin but occupy different orbitals at that sublevel before occupying another orbital at that sublevel that already has an electron
Quantum Mechanical Model
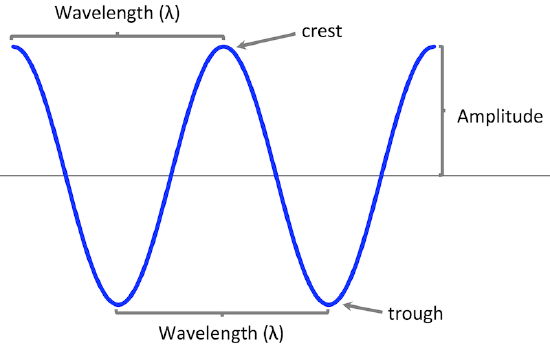
amplitude – the height of a wave from its midline to either its crest or its trough
wavelength () – the distance from the crest of one wave to the crest of the next wave, or also the distance from the trough of one wave to the trough of the next wave
frequency () – the number of complete waves to pass by a given point per unit of time (usually hertz, Hz, or per second, /s)
the speed of light (c) is 2.998 x 108 m/s
Planck’s constant (h) is 6.626 x 10–34 J s
electromagnetic radiation – all electromagnetic waves including gamma rays, X-rays, ultraviolet light, visible light, infrared waves, microwaves, and radio waves
atomic emission spectra – patterns formed when light is separated, either by a prism or by a diffraction grating, revealing the different wavelengths of light of which it is composed
ground state – the lowest energy levels occupied by electrons when not in an excited state
Einstein – demonstrated wave-particle duality for light, indicated that light can behave like particles (Photons are the names given to the discrete quanta of light that behave like particles.)
the photoelectric effect – electrons are ejected from the surface of a metal when light shines on the metal
de Broglie – predicted wave-particle duality for electrons as confirmed by the famous double slit experiment
Heisenberg – experimentally determined that the act of measuring the location of an electron alters its velocity and vice versa so that it is impossible to know both the velocity and the position of the electron (This is called the Heisenberg uncertainty principle.)
wave-particle duality – both light and electrons can behave like waves and like particles