Lecture 10 - Lipogenesis and Pentose Phosphate Pathway
Learning Outcomes:
Recall the overall scheme for the synthesis of fatty acids from glucose
Understand the reaction catalysed by and the regulation of acetyl CoA Carboxylase (ACC)
Describe the reaction sequence of fatty acyl synthase (FAS)
Apply knowledge of the regulators of acetyl CoA carboxylase to different physiological situations
Describe the process of esterification
Ingrate the pathways and processes that need to come together to support and supply lipogenesis
Summarise the effect of insulin on the activity of pyruvate dehydrogenase
Evaluate the probability of the different fates of acetyl CoA in lipogenic tissue
Explain the role of citrate in controlling the major anabolic and catabolic pathways
Outline the mechanisms for the return of oxaloacetate to the mitochondria after citrate cleavage
Describe the purpose of the pentose phosphate pathway and how it fits into lipogenesis
Illustrate the flow of substrates and key enzyme control points for lipogenesis
Lipogenesis:
Storing dietary glucose as fat
Build fatty acids (lipogenesis)
Mainly getting acetyl-coa from glucose
Everything in the pathway is driven by insulin
Join fatty acids to make triglycerides (3 fatty acids + glycerol) 3-phosphate (Esterification)
Need reductive power to synthesise fatty acids
NADPH – formed in the pentose phosphate pathway
Doing this in the tissues that respond to insulin – white adipose tissue or liver
Liver is a good place to synthesise fatty acids but not to store them – fatty liver
White adipose tissue (WAT) is a site of fatty acid synthesis
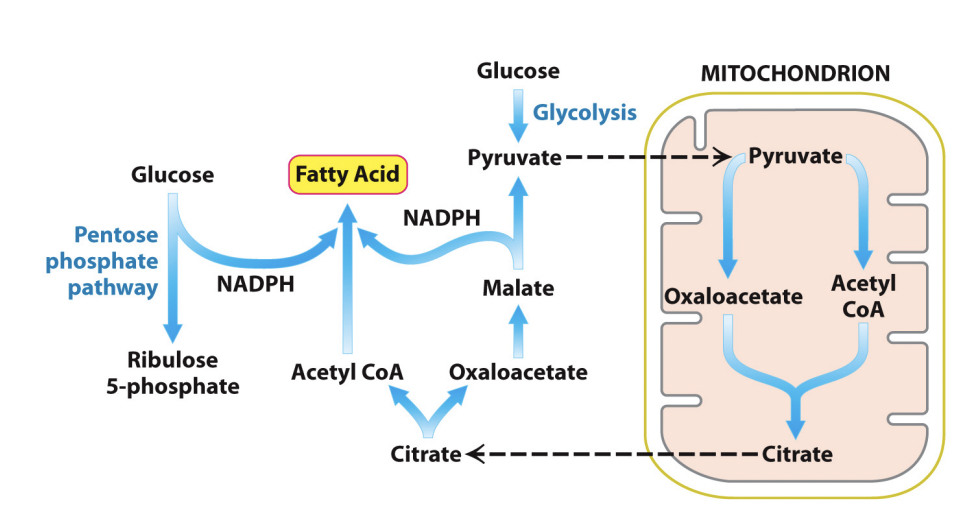
Lipogenesis occurs in the cytoplasm
Acetyl-coa go as far as citrate in the Krebs cycle to let it leave the mitochondria to the cytoplasm
Turn the citrate back into actyl-coa once it is in the cytoplasm to be used in lipogenesis
Need lots of reducing power to convert acetyl-coa to fatty acids
Reducing power comes from pentose phosphate pathway
If in the liver, need to make the decision to store as glycogen or fatty acids
WAT can only store as fatty acids
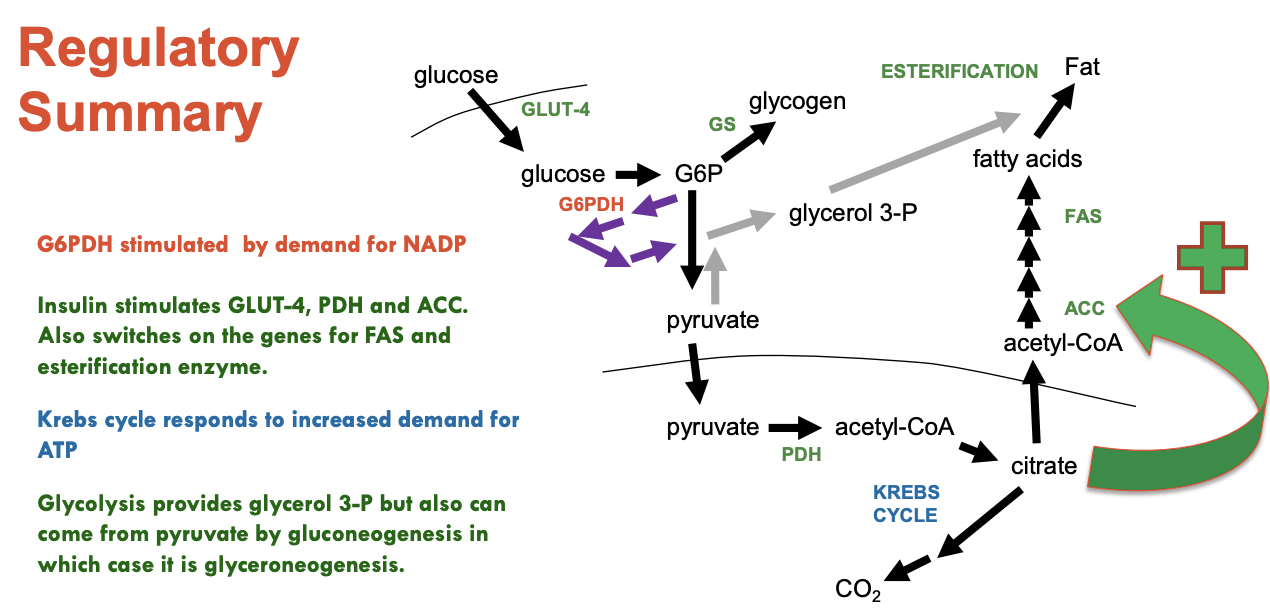
Acetyl-CoA Carboxylase (ACC)
Most regulated step in the process
Adds CO2 to acetyl-coa to make 3C chain, malonyl CoA
Requires biotin (vitamin), cofactors (Mg2+) and Used ATP
Takes the CO2 from biocarbonate (HCO3-) in the cell
Malonyl-CoA = activated Actyl-CoA
Ready for building new fatty acids
ACC Control:
Cycle of phosphorylation and dephosphoryaltion
Stimulated by insulin
Dephosphorylation by phosphates to make actice
Phosphorylation by kinase to make inactive
Insulin is actively telling the cells to turn acetyl-coa into malonyl-CoA ready for fatty acid synthesis
Activated allosterically by citrate
Lots of citrate going into the cytoplasm, this enzyme is activated
Inhibited allosterically, switched off, by its’ product – fatty acetyl-CoA
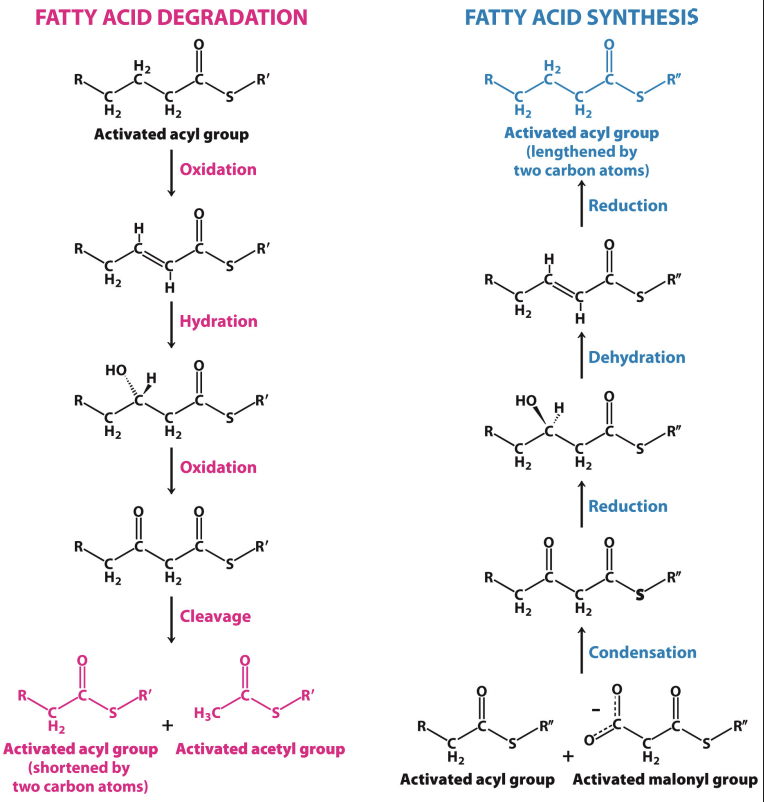
Lipogenesis = Reduction → dehydration → reduction (chain growing 2C at a time)
Beta oxidation = oxidation → hydration → oxidation (chain cutting 2C at a time)
Fatty Acyl Synthase Points:
Covalently binds to its substrates
Keep adding 2C via acetate
Do this by adding malonyl-CoA onto the FAS
Malonyl is decarboxylated (CO2 is removed) and leaves a negative charge on the malynol group
Negative charge attacks carbonyl carbon and attaches to the other chain
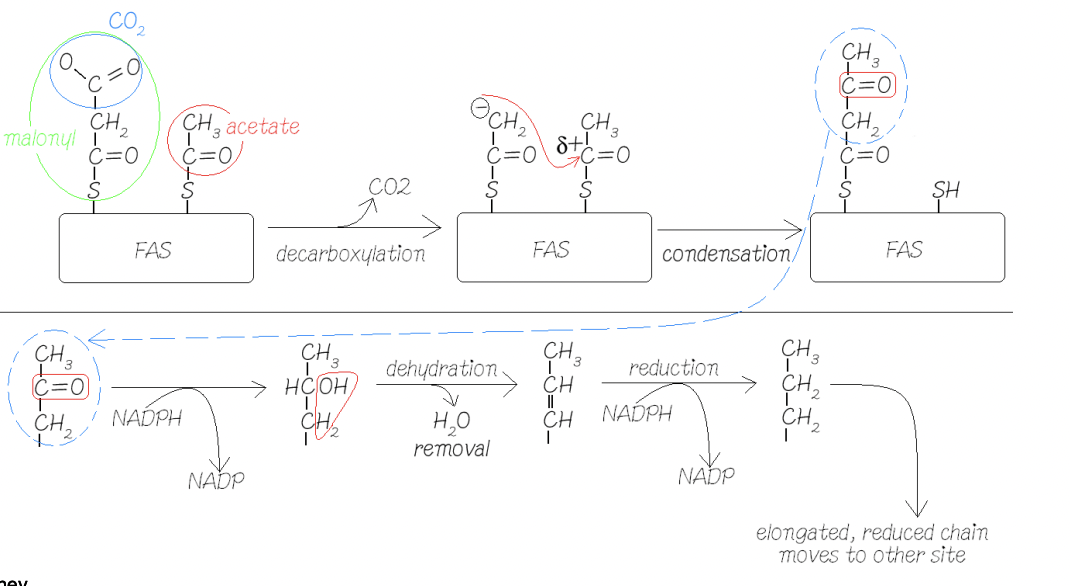
The chain produced is attached back to where the acetate was attached at the beginning, process repeats
FAS has left side for incoming malonyl CoA and right side for growing chain
Massive Multifunctional Problem:
Everything is happening on the one FAS enzyme
Input and Output
Each round of 2C addition requires:
2 molecules of NADPH
BUT NO ATP (synthase)
Only ATP used was used when ACC turned acetyl-CoA into Malonyl-CoA
Carboxylation of acetyl-CoA does ot result in fixing CO2 as it was lost immediately when attatched with FAS (Fatty Acyl Synthase)
FAs released as FA-CoA when chain gets 14 to 18 carbons long
Desaturation of CH2-CH2 to -CH=CH= is done after release from FAS
We lack enzymes that introduce doubles bonds beyond carbon 9, long unsaturatesd omega-3/7 FA are essential in the diet
Esterification
Join fatty acids to glycerol
Glycerol and 3 fatty acids forms ester bonds (condensation reaction)
Glycerol needs to be glycerol-3P
Fatty acids need to be fatty acyl CoAs
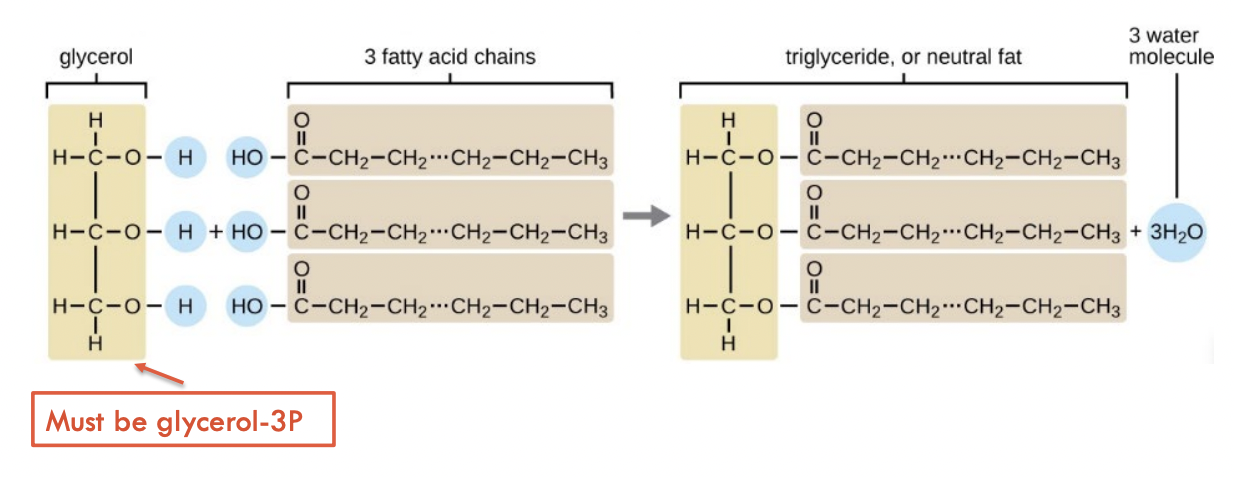
Turn FA into fat
3 fatty acid chains are added one at a time (mono-, di-, tri-)
Only the liver has the enzyme to convert glycerol into glycerol 3-phosphate (glycerol kinase)
If in fat tissue, go halfway down glycolysis and take glyceraldehyde 3-phosphate – reduce it to glycerol 3P
Glycolysis provides glycerol backbone and acetyl-coa building blocks for new FA
Esterification enzymes like to use FA-CoA not just fatty acids
Esterification enzymes and FAS are upregulated by insulin at a genetic level
FAS expression is down regulated when lots of fat is around – as in a Western diet
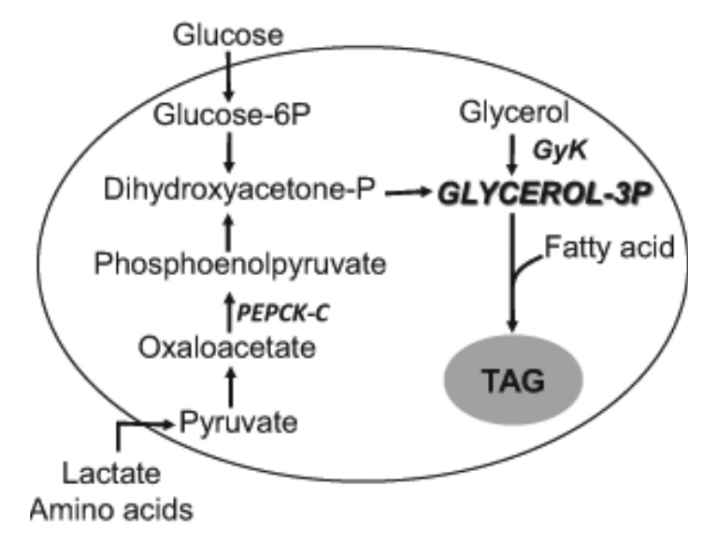
Glycerol 3P Generation:
Glucose → Glucose 6P → Dihydroxyacetone-P
Dihydroxyacetone-P (3C) can be exchanged with glyceraldehyde 3P and reduced to glycerol 3P
LIVER ONLY – Use glycerol kinase to add a phosphate to glycerol to make glycerol 3P
Can also start with lactate or amino acid skeleton as a substrate and work through gluconeogenisis until it gets to dihydroxyacetone phosphate – highly unlikely
Termed Glycerolneogenesis
Servicing Lipogenesis: Creating NADPH for Lipogenesis
Pentose Phosphate Pathway
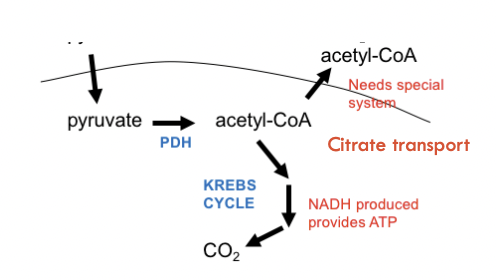
PDH is activated by insulin and lots of acetyl CoA is formed
Some may enter Krebs cycle to make NADH and make ATP (necessary for malonyl CoA)
Some acetyl CoA is let out into the cytoplasm to go in the Krebs Cycle oxaloacetate to create citrate
Allows CoA to come back, allows PDH to continue
Transport or oxidise the citrate formed
If lots of cycles happening and it is not needed in the Krebs cycle, citrate will go back out of the mitochondrion into the cytoplasm and assist with fatty acid synthesis
Fate of citrate depends on the energy charge within the cell
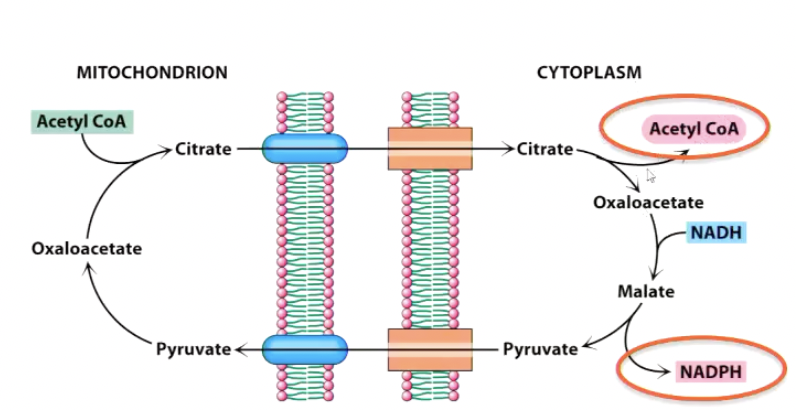
ATP-Citrate Lyase:
Opposite of citrate formation
Break 6C citrate into 4C oxaloacetate and 2C acetyl-CoA
Requires ATP
This is how the acetyl-CoA gets in the cytoplasm to do this
ACL is inhibited by hydroxy-citrate (OHCit)
Found in brindle berry
Sold as a fat synthesis inhibitor – doesn’t actually work
Oxaloacetate needs to be transported back into the mitochondria or we will run out of Kreb cycles
Replaced by reducing oxaloacetate with NADH to give malate and to pyruvate, which produced NADPH (necessary for recucing FA into triglycerides). Pyruvate can go back in and be converted to oxaloacetate by pyruvate carboxylase
Malonyl CoA Inhibits Beta-Oxidation
Malonyl CoA inhibits CAT-I
CAT1 transports FA into the mitochondrian where they could undergo beta oxidation, which is now inhibited
Therefore, FA oxidation is inhibited by malonyl CoA
Insulin stimulates malonyl CoA production by Acetyl-CoA Carboxylase (ACC)
Therefore, Insulin inhibits (beta) fatty acid oxidation by malonyl production
The Reductant: NADPH
NADPH is a form of NADH involved in anabolic reactions
Same chemical properties, just bound to different enzymes
Pentose Phosphate Pathway produced NADPH proportional to demand for lipogenesis
Pentose Phosphate Pathway:
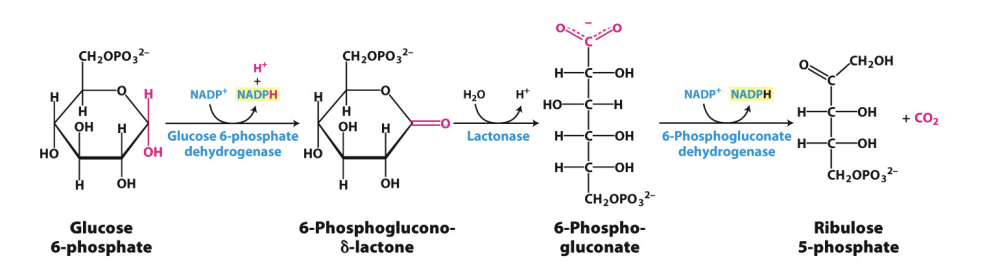
Take glucose 6-phosphate from glycolysis
Glucose-6-phosphate dehydrogenase acts on glucose 6-phosphate and produced an NADPH
Goes through other reactions and another enzyme produced an additional NADPH
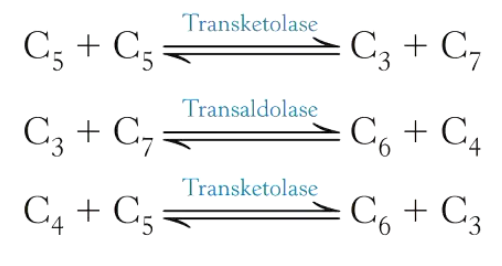
Leaves a 5C sugar – ribulose 5-phosphate
Rest of the cycle is about rearranging the amount of carbon different sugars have to produce a 6C and 3C sugar to be put back into other pathways
Glucose 6-phosphate dehydrogenase is regulated by demand for NADPH
We use NADPH during lipogenesis, which produced NADP+ which is used in the pentose phosphate pathway to produce NADPH
NADPH is a vital anti-oxidant in cells
In RBC a deficiency in G6PDH enzyme causes anemia – produced clumps of haemoglobin