HW #34
Bronsted-Lowry acids/bases
The Brønsted–Lowry Theory: defines acids and bases as either proton donors or proton acceptors
According to the theory:
a Brønsted–Lowry acid donates a proton (is a proton donor)
a Brønsted–Lowry base accepts a proton (is a proton acceptor)
A lot of reactions in life involve the transfer of protons from substances called acids to substances called bases
EX: When hydrogen chloride gas dissolves in water, it donates a proton (H+) to the water molecule, which accepts the proton.
The reaction between hydrogen chloride gas and water:
The hydrogen chloride gas acts as a Brønsted–Lowry acid and the water molecule as a Brønsted–Lowry base
The solution formed is named hydrochloric acid and is an example of a Brønsted–Lowry acid
Three common types of acid:
binary acids
oxyacids
organic acids
Binary acids are formed when a halide reacts with hydrogen (HF, HBr, HCl, etc.)
Oxyacids acids are derived from polyatomic ions (HNO3(aq), H2SO4(aq), H2CO3(aq), etc)
Organic acids contain a carboxyl group (Ethanoic acid: CH3COOH(aq) & Benzoic Acid: C6H5COOH(s))
Acids can be classified as monoprotic, diprotic, or triprotic depending on the number of hydrogen ions or protons that they can donate in an acid-base reaction
Name of base Formula
Calcium oxide CaO(s)
Copper(II) oxide CuO(s)
Sodium hydroxide NaOH(aq)
Lithium hydroxide LiOH(aq)
Calcium hydroxide Ca(OH)2(aq)
Barium hydroxide Ba(OH)2(aq)
Carbonates and metal hydrogen carbonates are also examples of weak bases
Ammonia, amines and amides are also examples of weak bases
Hydrochloric acid and nitric acid are monoprotic acids
Sulfuric acid is an example of a diprotic acid
Phosphoric acid is an example of a triprotic acid
Conjugate Acids & Bases
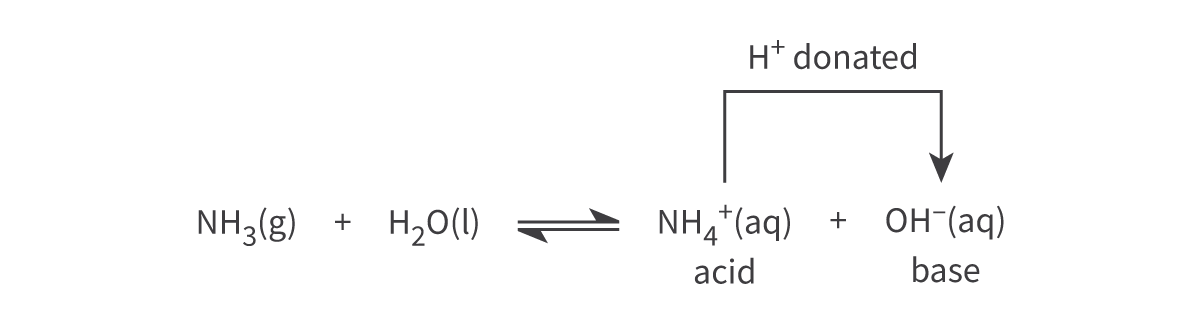
The reaction between ammonia and water to produce the ammonium ion and the hydroxide ion is:
NH3(g) + H2O(l) ⇌ NH4+(aq) + OH−(aq)
In the forward reaction, water acts as a Brønsted–Lowry acid and ammonia as a Brønsted–Lowry base
it’s a reversible reaction (as indicated by the two-way arrow) —> forward reaction occurs at the same rate as the reversible reaction & the reaction is at equilibrium
In the reverse reaction: NH4+ ion donates a proton to the OH− ion —> the NH4+ ion is acting as a B–L acid and the OH− ion is acting as a B–L base
Conjugate (Acid-Base) Pair: differ by one proton (EX: NH3 & NH4+) (EX: H2O & OH-)
Amphiprotic: species that can act as either B-L acid or base (EX: Water)
water in the reaction with ammonia behaves as an acid and donates the proton
water in the reaction with HCl gas behaves as a base and accepts the proton
HSO4- is also an example