CHEMICAL CHANGES
Acids and alkalis
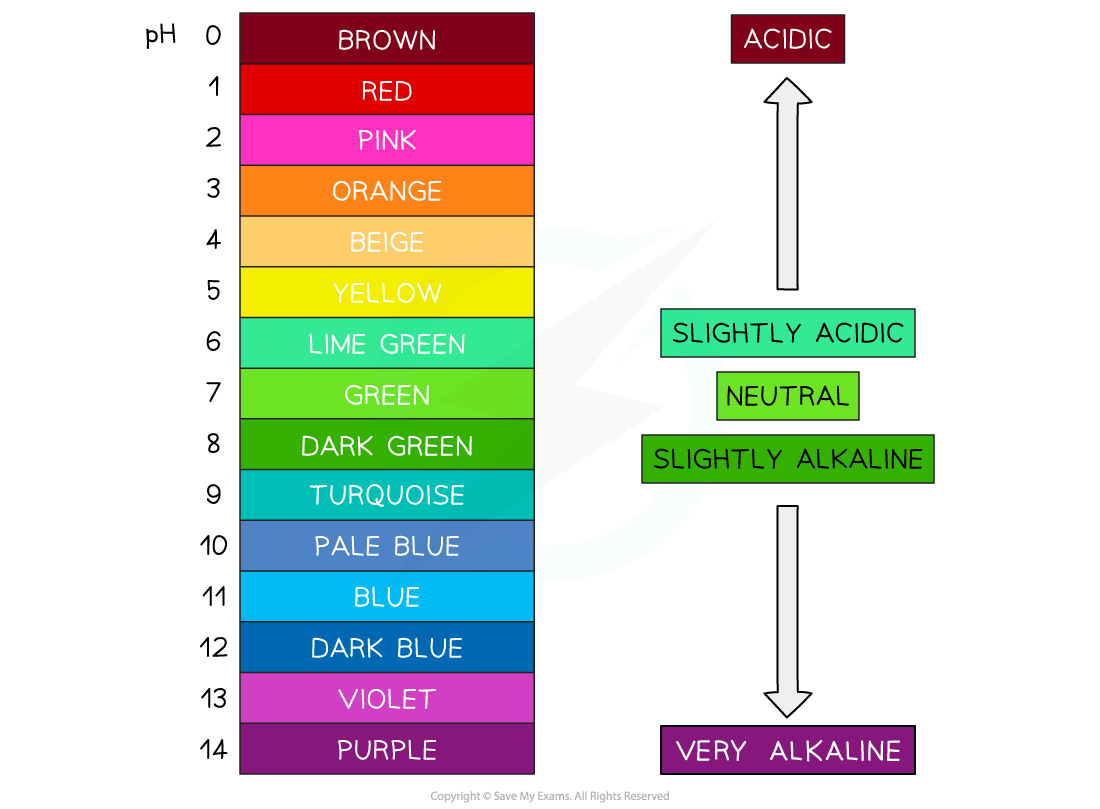
For example, stomach acid is around pH 2, and bleach is around pH 12
Indicators are chemical dyes that change to laine with the pH colour
Universal indicator is the most common, and changes to the same colours as above 🙂
A pH probe and meter give a number reading, and are more accurate and precise
Acids are any substance that forms an aqueous solution with a pH less than 7
A base is any substance with a pH greater than 7
Alkalis are any substances that dissolves in water to form a solution with a pH more than 7
Neutralisation reactions are when you react an acid and a base to form a solution CLOSER to pH 7 (pure water)
They form a salt and water
HCl + NaOH \rightarrow NaCl + H2O
Acids I need to know
Hydrochloric acid is HCl
Sulfuric acid is H2SO4
Nitric acid is HNO3
Alkalis I need to know
Sodium hydroxide is NaOH
Calcium carbonate is CaCO3
Titration practical
A titration is an experimental technique used to find an unknown concentration of an acid or alkali
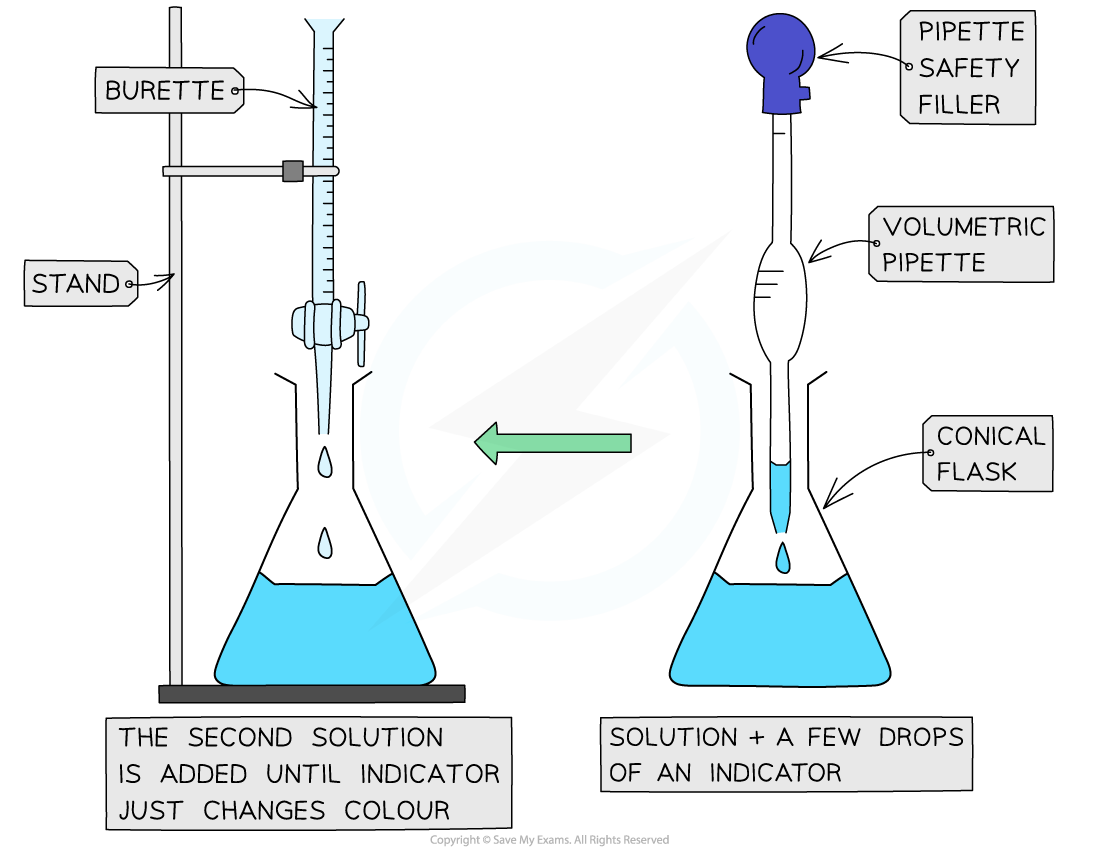
Neutralising an alkali with acid
Use a pipette to add 25cm3 of alkali to a conical flask
Add a few drops of an indicator (can’t be universal indicator) and place on a white tile
Fill a burette with acid and note the starting volume
Slowly add acid from the burette to the conical flask, swirling slowly, so that it is evenly distributed
Stop when the end point is reached - the acid has neutralised the alkali and the indicator has changed colour
Note the final volume reading and calculate the total acid added
Repeat until you get concordant results - within 0.10 cm3 of each other
Calculate a mean
Indicators that can be used:
Litmus is RED in acids, and BLUE in alkaline solutions
Phenolphthalein is COLOURLESS in acids, and PINK in alkaline solutions
Methyl orange is RED in acid, and YELLOW in alkaline solutions
Universal indicator can’t be used as it gradually changes, and you can’t clearly see where it has reached the end point
Strong and weak acids
Acids are ionised in aqueous solutions to release H+ ions
For example, HCl exists as H+ and Cl- ions
Strong acids ionise completely - all particles will dissociate
Like HCl or H2SO4
Concentration is the amount of acid per amount of volume
The amount of H ions per unit of volume
Weak acids don’t fully ionise - their reactions are reversible and stop reacting when they reach an equilibrium (net reactions)
Like CH3COOH - ethanoic acid
pH is a measure of concentration of H+ ions in a solution
The higher the concentration, the lower the pH
HCl is a strong acid and could easily be pH0 at most concentrations
If it was a weak acid, to be pH0, it would have to be very very concentrated
Neutralisation reactions
Metal oxide + acid \rightarrow salt + water
Metal oxides end in O
Metal hydroxides + acid \rightarrow salt + water
Metal hydroxides end in OH
Metal carbonates + acid \rightarrow salt + water + carbon dioxide
To make a soluble salt, we have to react an insoluble base with an acid
We gently heat an acid, and slowly and a base a bit at a time until it stopes dissolving
This shows the acid is neutralised and that the base is in excess
Then filter the excess base, and the product is dissolved soluble salt
To form crystals from this solution, we can gently heat it to evaporate any water (at less than 60 degrees)
When crystals begin to form, stop heating and leave to cool
This will cause more crystals to form, which can be filtered and dabbed dry
The reactivity series and displacement
Potassium | please |
Sodium | send |
Lithium | little |
Calcium | Charlie’s |
Magnesium | monkeys |
Aluminium | and |
CARBON | cheeky |
Zinc | zebras |
Iron | in |
Tin | thick |
Lead | large |
HYDROGEN | heavy |
Copper | cages |
Mercury | most |
Silver | securely |
Gold | guarded |
Elements below carbon can be reduced with carbon (in metal oxides)
Reactivity is how easily something forms a positive ion
Metals + acid \rightarrow Salt + hydrogen
Reaction gets less violent down the reactivity series
Causes bubbles, explosions and temperature changes
Metals + water \rightarrow Metal hydroxide + hydrogen
Only in the most reactive metals
Displacement example:
Mg + FeSO4 \rightarrow MgSO4 + Fe
Magnesium displaces iron, as it is more reactive
Separating metals from metal oxides
Oxidisation is the process of losing electrons (gaining oxygen)
2Mg + O2 \rightarrow 2MgO
Reduction is the process of gaining electrons (losing oxygen0
2MgO \rightarrow 2Mg + O2
OILRIG - Oxidisation is loss, reduction is gain
Most metals are fairly reactive
They reactive with oxygen (from the air) to form metal oxides
If they are really unreactive, they don’t react, like gold
To get pure metals, we want to reduce them to remove the oxygen
This is done by reacting metal oxides with carbon (displacement)
Oxygen will react with carbon and leave behind the pure metal
This only works with metals less reactive than carbon
It is cheap and quick
For example, 2Fe2O3 + 3C \rightarrow 4Fe + 3CO2, when mining for iron
More reactive metals are extracted via electrolysis, which uses lots of energy and is expensive
The process of electrolysis
Electrolysis involves using to break down electrolytes to form elements
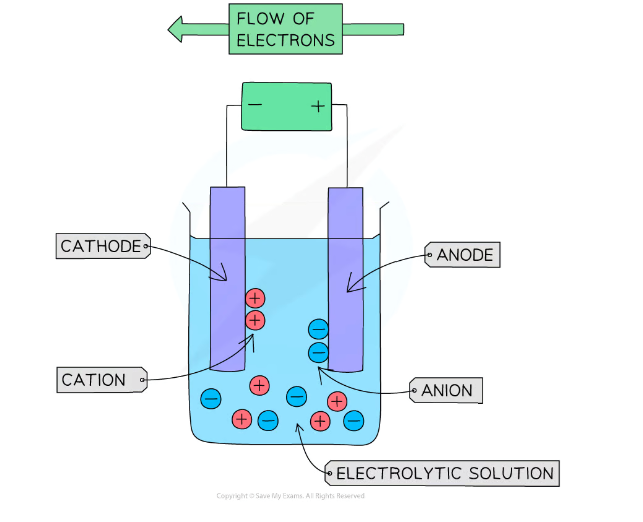
- ions are attracted to the anode (+ electrode)
They are then discharged, and become atoms
+ ions are attracted to the cathode (- electrode)
They are then discharged to become atoms, and produce either a solid or a gas
At the cathode, Pb2+ + 2e- \rightarrow Pb, reduction which forms solid lead
At the anode, 2Br- \rightarrow Br2 + 2e-, oxidisation which pairs up and forms gas (covalent bonds)
Electrons at the anode that are loss are passed through the wire to the cathode, where they are given off to the positive ions to form atoms
Electrolysis of aqueous solutions
Electrolysis is used to split compounds into their elements
Soluble compounds can be dissolved in water to become aqueous, so charged particles are free to flow
Because the compounds are dissolved in water, both the ions of the compound, and H and HO ions will be present
For example:
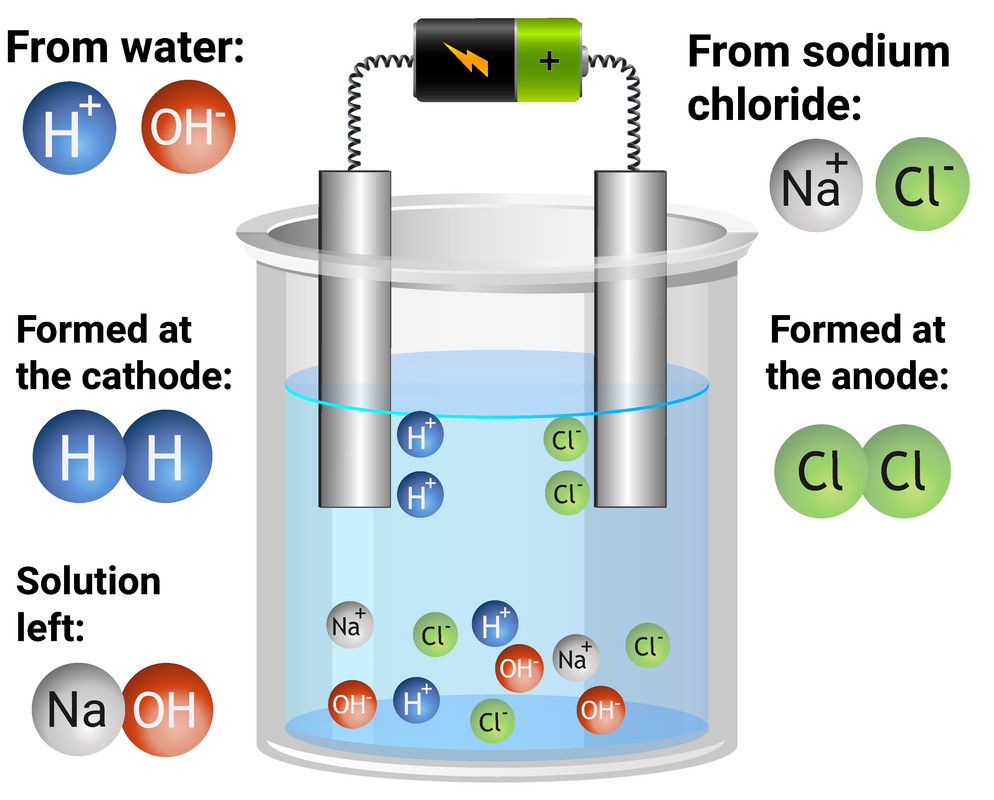
The cathode attracts positive ions - Na+ and H+
Only the less reactive will be discharged at the cathode
As hydrogen is less reactive than chlorine, hydrogen atoms will be formed, and given off as hydrogen gas
The anode attracts negative ions - OH- and Cl-
If a halide is present, it will be discharged. If not, OH- will be discharged
Halides are any of group 7 - F, Cl, Br, I and At
Chloride is a halide, so chloride is discharged, and forms chlorine gas at the anode
This leaves NaOH in the beaker as a solution
Oxidation and reduction in terms of electrons
Oxidation is loss, reduction is gain
Reduction and oxidisation reactions both take at the same time - redox reactions
This is because the electrons that are loss also need to be gained
Ionic equations are used to show displacement
They only show the particles that take part in the reaction and change
Ca + Fe2+SO42- \rightarrow Ca2+SO42- + Fe
Ca + Fe2+ \rightarrow Ca2+ + Fe
SO42- is left out as it doesn’t change
Half equations show the gain and loss of electrons for each element involved
Ca \rightarrow Ca2+ + 2e-
Fe2+ + 2e- \rightarrow Fe
The charged particles are always placed on the same side
Reactions of acids with metals
The products of reactions of acids with metals are always a salt and hydrogen
Acid + metal \rightarrow salt + hydrogen
They are redox reactions
2H+ + Mg \rightarrow Mg2+ + H2
Magnesium loses electrons - is oxidised
Hydrogen gains electrons - is reduced
Soluble salts
Soluble salts can be prepared by reacting an acid with a suitable insoluble reactant
The reactant can be a metal, metal oxide, carbonate or a metal hydroxide
The reactant depends on the salt required
For example, copper doesn’t react with dilute acids, so can’t be used, and sodium is too reactive to use
The reaction between an acid and a metal produces hydrogen
Hydrogen is flammable, so we usually use metal oxides or carbonates and an acid instead
Making a salt
Add the powdered insoluble reactant to an acid in a beaker, 1 spatula at a time
The acid can be gently heated by a Bunsen burner to increase the reaction speed (particles have more energy)
Stir between each addition and continue until the powder is in excess and no longer reacts
Filter the mixture in the beaker with filter paper and a funnel to remove excess solid - this means the filtrate now only contains the salt and water
Heat the solution in a evaporating dish over a water bath
Stop when small crystals begin to appear - the solution is now saturated and most of the water has evaporated
Leave the solution for 1-2 days at room temperature to allow the rest of the water to evaporate and leave large crystals
Dry by gently dabbing with filter paper
Electrolysis of molten ionic compounds
This is the process of separating elements in insoluble ionic compounds (splitting with electricity)
In electrolysis, the electrolyte has to be a liquid or aqueous solution that contains an ionic compound - so the ions can move freely
CuSO4 is soluble, and can be dissolve in water to create this
However, lead bromide is insoluble and has to be melted and made molten to allow ions to move freely

PbBr2 is heated to become molten
It splits into it’s two ions - Br- and Pb2+
Br- is attracted to the positive anode and is discharged as bromine gas
Br- \rightarrow Br + 2e-
Two bromines react covalently to form 2Br as a gas
Pb2+ is attracted to the negative anode and is discharged as solid lead
Pb2+ + 2e- \rightarrow Pb
This creates a layer of molten lead in the beaker
Ions are being oxidised and reduced at the electrodes
Cathode = reduction
Anode = oxidisation
Electrons that are lost when Br- become Br are transported via the wire with current to the anode, where Pb2+ gains two electrons
Extracting metals with electrolysis
Reactive metals are extracted from metal oxides by melting and making them molten compounds
The cheapest way to reduced a metal from an oxide is with carbon, but it only works with metals that are less reactive than carbon
So we use electrolysis instead, which is expensive and needs lots of energy
Electrolysis only works in ions can freely move through the solution
For example, Al2O3 \rightarrow Al + O2
Aluminium is more reactive than carbon, so electrolysis is used
Aluminium oxide is found as a solid, and found mixed with bauxite when mined
This needs to become molten 🔥
First, we need to purify Al2O3 from bauxite
Then, we mix aluminium oxide with cryolite as it lowers the melting point
Aluminium oxide has a very high melting point (it was ~ 2000 degrees C), but is lowered by cryolite
We then melt this mixture to become molten - lots of energy is required as the melting point is still high
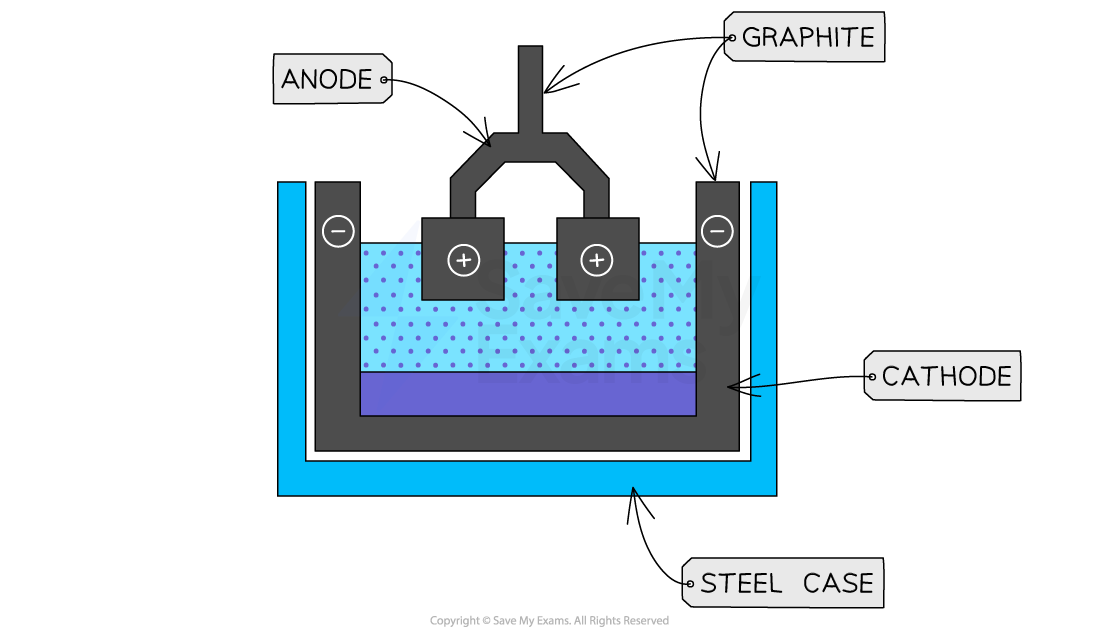
The electrodes are made of graphite, and the cathode is found around the outside of the steel case
O2- is attracted to the anode in the centre
At the anode, it is discharge and becomes oxygen gas (makes a pair and covalently bonds once atoms0
2 electrons are lost per oxygen, and they are transferred through the wire to the cathode for reduction
2O2- \rightarrow O2 + 4e-
Al3+ is attracted to the cathode around the edge
It is discharged and becomes Al
3 electrons are gained from the electrons lost at the anode
The aluminium formed pools at the bottom, and leaves via a channel at the bottom to be collected
Al3+ + 3e- \rightarrow Al
2Al2O3 (l) \rightarrow 4Al (l) + 3O2 (g)
DONE!!!