ENERGY CHANGES
Energy transfers during exothermic and endothermic reactions
Different chemicals store different amounts of energy in their bonds
CH4 + 2O2 \rightarrow CO2 + 2H20
Before this reaction, the bonds have more energy, and after energy has been released to the surroundings, through heat
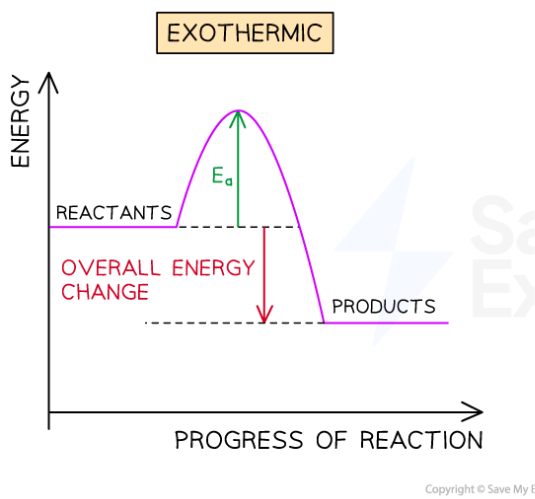
Exothermic reactions release energy to the surroundings
Energy is EXiting the reaction
The surroundings get hotter
Transfers energy to surroundings
Endothermic reactions take in energy from the surroundings
Energy is ENtering the reaction
The surroundings get cooler
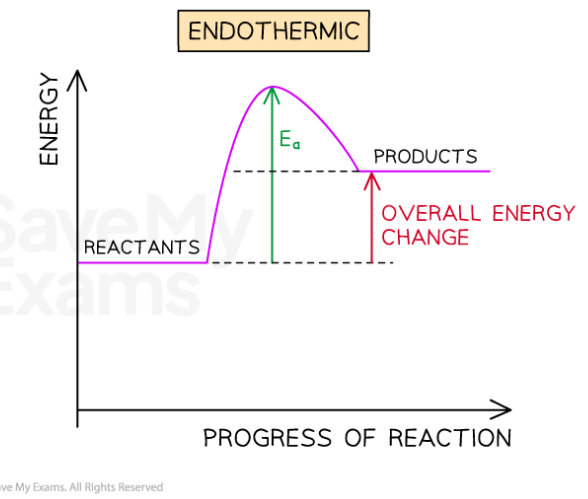
Activation energy is the minimum energy the reactant particles need in order to collide with each other enough to cause a reaction
Energy changes of reactions
Bond energy is the amount of energy required to break one mole of a particular bond
For example, a H-Cl bond requires 431 kJ/mol (energy needed to break one mole - 6.02 × 1023 - of these bonds)
Bond breaking is exothermic, and making bonds is endothermic
To calculate:
Work out how many bonds are breaking in the reactants, and calculate the total energy required by multiplying the number of specific bonds by the kJ required
Work out the same for the products
Find the difference in the total energy required for the reactants and the products 🙂
Cells and batteries
Electrochemical cells use chemical reactions to produce electricity
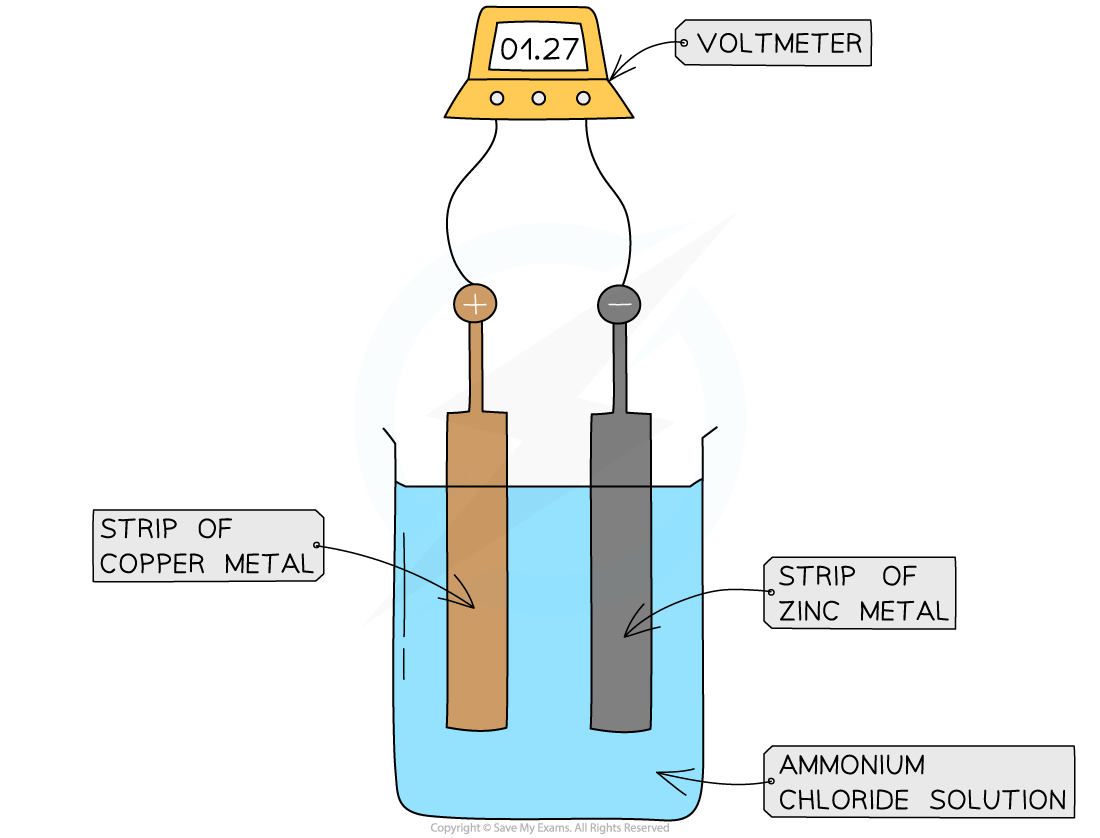
The electrodes have to be made out of two different types of metal, and they conduct
A cell can be made by connecting two different metal electrodes with wire and placing them in contact with an electrolyte solution
An electrolyte is a liquid through which charged particles can flow - creates a flow of charge, and therefore a cell
Batteries are similar, but consist of two or more cells connected in series to provide a greater voltage
Factors that affect the voltage of a cell or battery include:
The metals used - the greater the difference in their reactivities, the greater the voltage
The type and concentration of electrolyte
The conditions, such as temperature
Rechargeable batteries work as the chemical reactions inside can be reversed when an external electrical current is supplied
They are used in phones and laptops
Non-rechargeable batteries are where the reactions stop once one of the reactants has been used up
Used in smoke alarms, TV remotes, etc.
Also called Alkaline batteries
Fuel cells
Fuels cells are electrochemical cells that converts energy between chemical and electrical
We can convert the energy of oxygen and a fuel to release electrical energy we can use
The most common type of fuel cell is the hydrogen-oxygen fuel cell
It forms water and creates lots of electrical energy
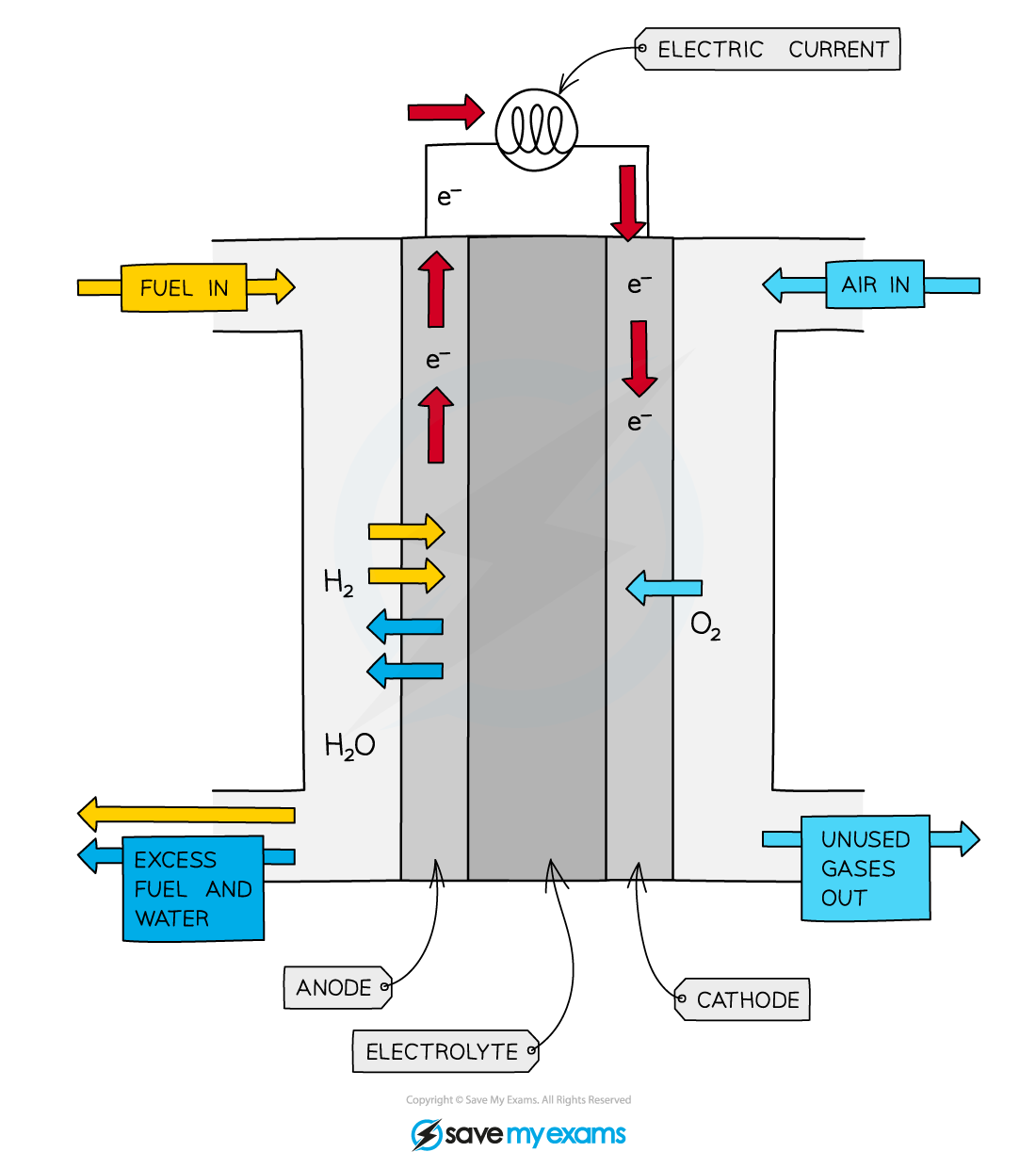
H2 enters through the left of the fuel cell, and is oxidised by the anode (-) to split into two positive hydrogen ions, and two electrons
The oxidisation of hydrogen - H2 \rightarrow 2H+ + 2e-
The electrons pass through the wire, creating a current and electrical energy, to the cathode
The hydrogen ions pass through the electrolyte to the cathode (+)
Oxygen enters from the right side
Hydrogen ions and electrons can react with oxygen to produce water
Two H2 and one O2
O2 + 4H+ + 4e- \rightarrow 2H2O
The water leaves the fuel cell via the outlet, as well as heat (non-useful energy)
The overall equation of this process is O2 + 2H2 \rightarrow 2H2O
The electrical energy comes from the flow of electrons through the wire
As the fuel enters the cell, it becomes oxidised, creating a potential difference across the cell
PROS - Only requires oxygen and hydrogen
No waste is created
They last longer than batteries
Simple process
CONS - H2 is expensive to store as it takes up lots of space
H2 is explosive in air ☹
To make hydrogen fuel, we need energy, often from fossil fuels
DONE!!!