Stoichiometry
Formulae
A formula is a way to represent the elements and their proportions in a compound.
Each element is represented by a unique chemical symbol, such as “H” for hydrogen, “O” for oxygen, and “Na” for sodium.
We use numbers to indicate the number of atoms of the element. (O2, H, N2)
Molecular Formula of a Compound
The molecular formula of a compound is the exact number of type of different atoms in one molecule.
Empirical Formula of a Compound
The empirical formula of a compound is the simplest whole number ratio of the different atoms or ions in a compound. It tells you the reduced version of a chemical formula, not necessarily the exact number of atoms in one molecule.

Finding the molecular formula from the empirical formula:
To find a molecular formula from an empirical formula:
Find the molar mass of the empirical formula
Divide the compound’s actual molar mass by that empirical-formula mass
Multiply every subscript in the empirical formula by that whole number
Finding the empirical formula from the molecular formula:
Divide all the subscripts by their greatest common factor. That gives the simplest whole-number ratio of atoms
Valency
Valency is how much an atom can make bonds with other atoms.
It tells us how many electrons an atom needs to gain, lose, or share to achieve a stable electron configuration.
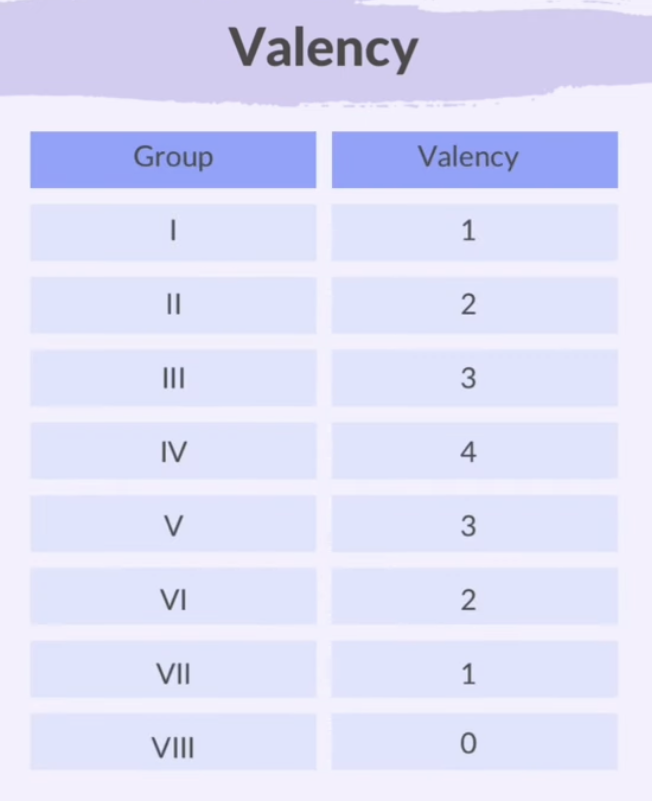
Groups I to III lose electrons.
Groups V to VII gain electrons.
Group IV can do either.
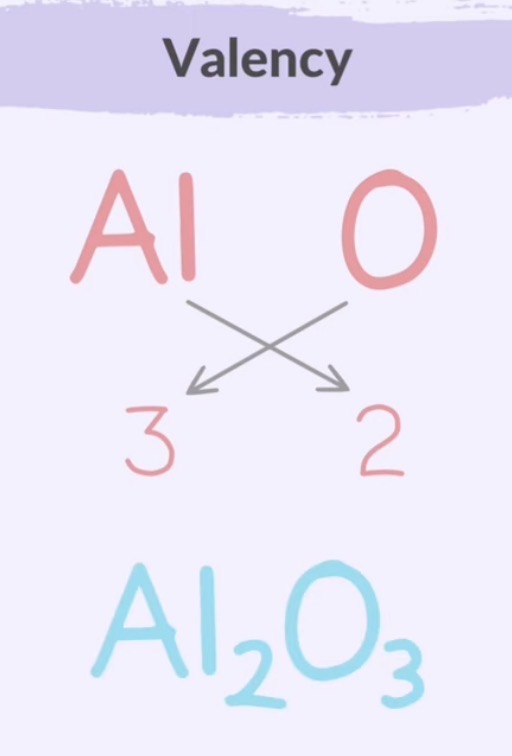
If valencies are equal, you must cancel them out.
Criss-cross method determines the correct subscripts in chemical formulas for ionic compounds by swapping valencies to balance the positive and negative charges.
Write the metal cation symbol first, and then the non-metal anion symbol. Criss cross and simplify.
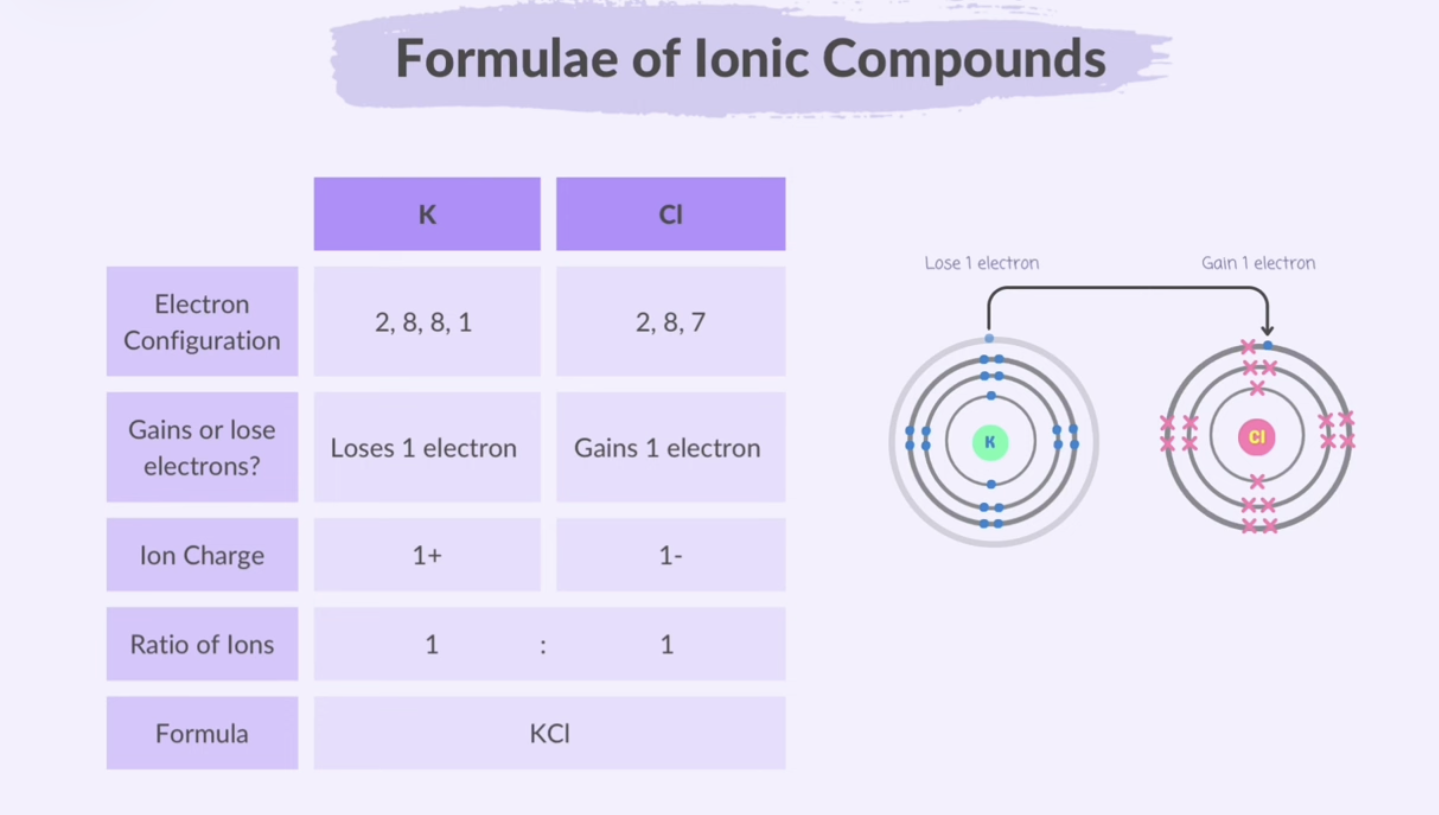
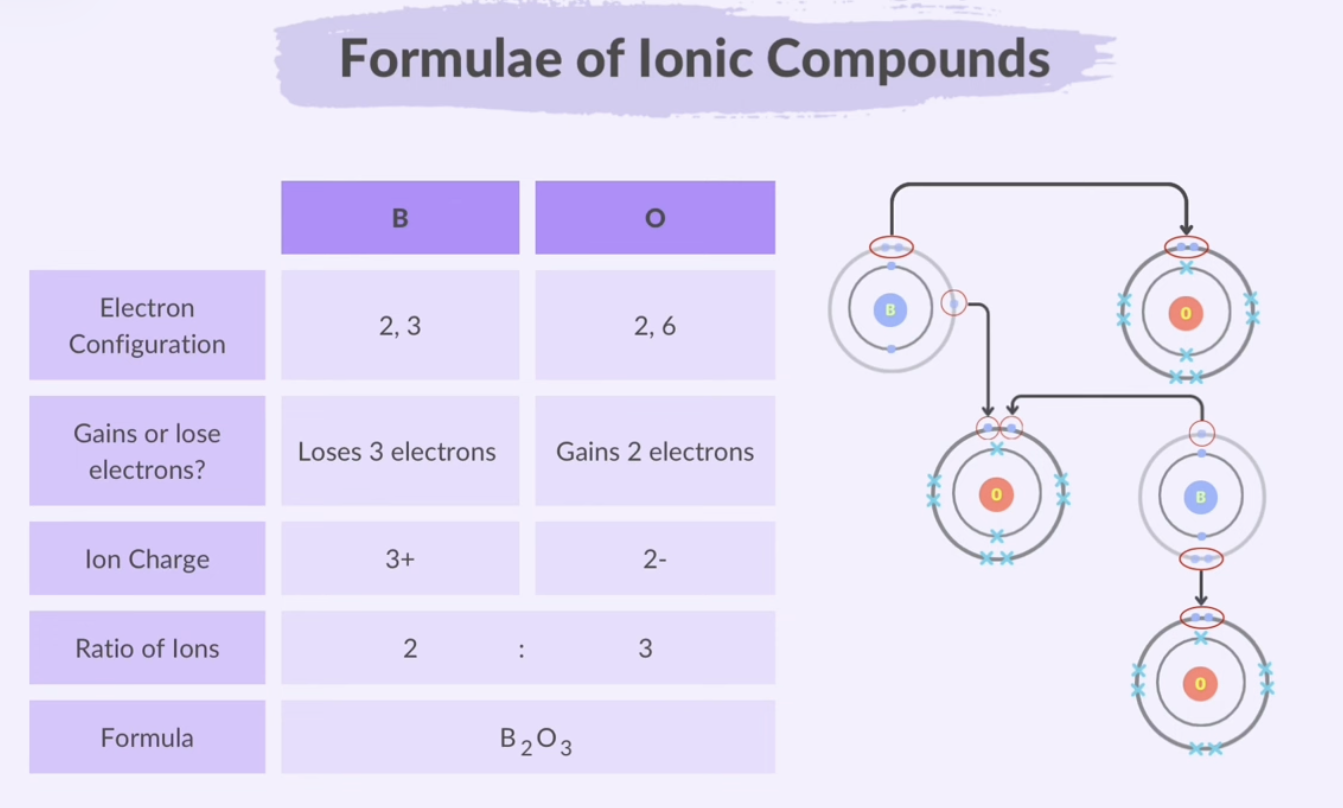
Formulae of Ionic Compounds
Ionic compound formulas are always empirical.
When atoms form ions through ionic bonding, their valency also tells you the charge of the ion.
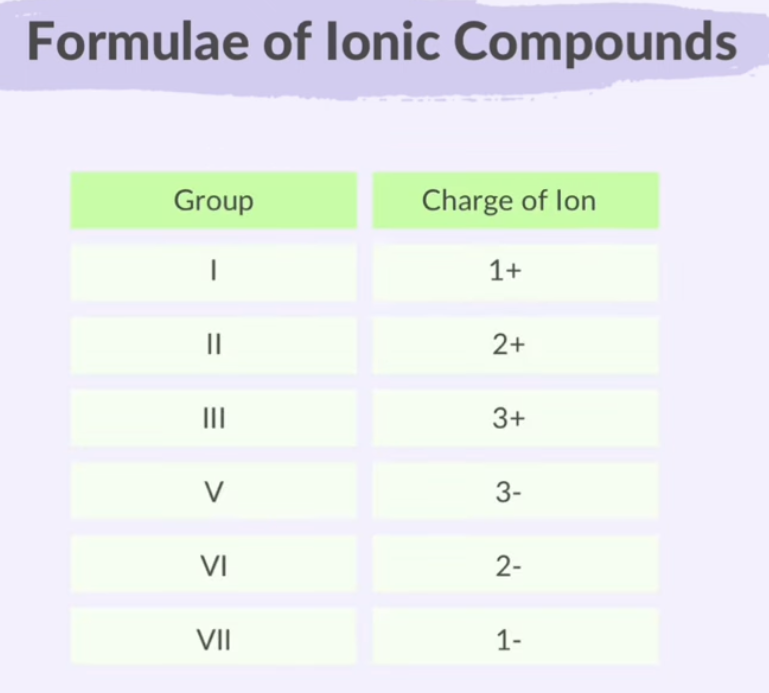

In an ionic compound, the positive and negative charges of the ions cancel each other out, resulting in a total charge of zero for the compound.