M3L8 Vacularisation, hypoxia, acidosis, and the Warburg effect
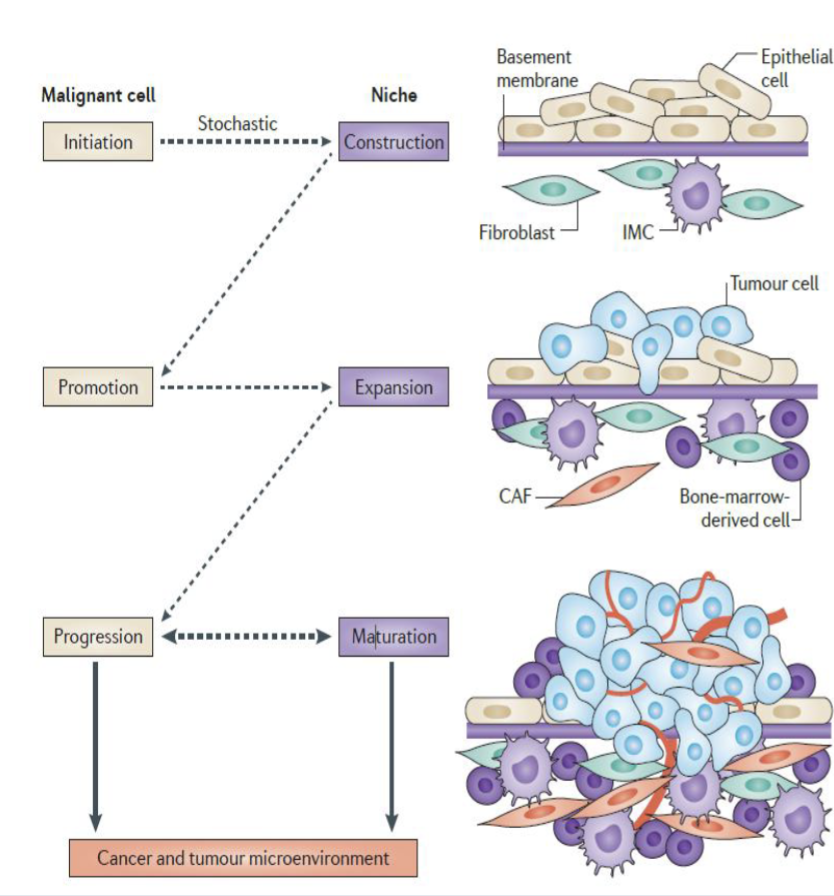
Niche construction - spontaneous interaction between activated stromal cells and normal cells that enables initiated/transformed clone survival
Niche expansion - microenvironment that generates secreted factors that remodel local tissue, concurrent with initiates clone expansion and parallels tumour promotion
Niche maturation - recruitment of bone marrow derived cells as well as resident cells (esp. fibroblasts) dries niche maturation from a nascent to an established TME
At some point a successful niche evolves and matures into a dynamic feedback system
Niche types (not mutually exclusive to each other):
Hypoxic
Acidic
Immune microenvironmnet
Innervated niche
Metabolism microenvironment
Mechanical microenvironment
Tumour growth depends on the adaptive responses exhibited by tumour cells in response to the evolving niche
Hypoxia
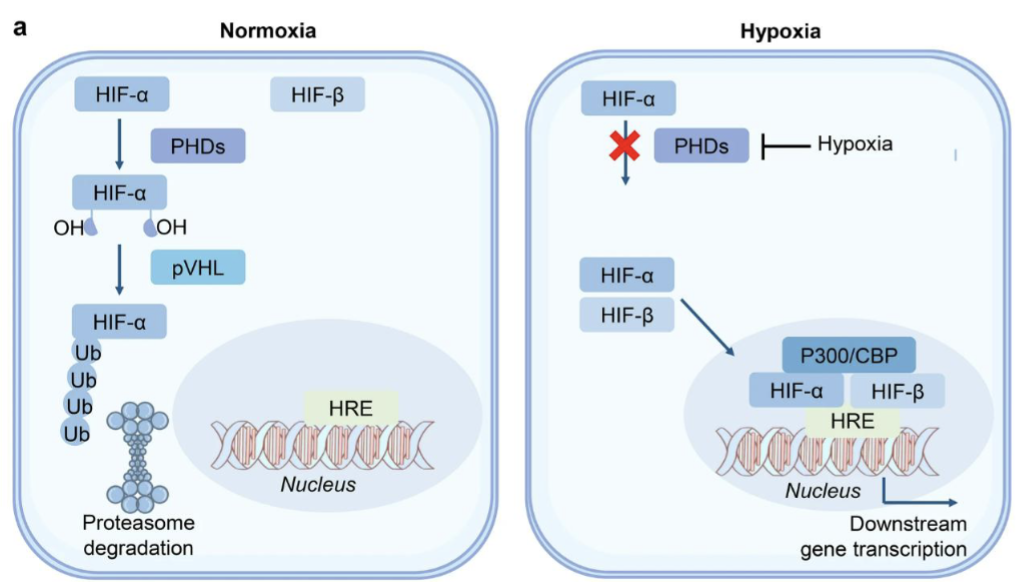
Adaptation to hypoxia is through HIFs
HIF-dependent signalling promotes adaptation and selection of cancer and stromal cells to the surrounding consitions, promoting pro-tumourigenic changes
HIF family of TFs includes HIF1, HIF2, HIF3 which all contain an oxygen sensitivity HIF-α subunit which dimerises with the constitutively expressed HIF-β subunit
Under normoxia. HIFs undergo ubiquitination mediated by PHDs (oxygen-dependent hydroxylase family) and pVHL (von Hippel-Lindau tumour suppressor protein)
Activity of PHD is prohibited under hypoxia
In the nucleus HIF-α binds to hypoxia response elements (HRE) to promote gene expression of genes with this promoter
The HIF1α gene has a DNA binding and dimerisation domain, oxygen-dependent degradation domain, and transactivation domain
PHDs and FIH can hydroxylate oxygen-dependent degradation domain and transactivation domain to mark for degradation
FIH hydroxylation of transactivation domain without PHD hydroxylation stabilise HIF1α
CAD active/NAD active (high hypoxia) vs CAD inactive/NAD (lower hypoxia) active states of HIF1α reflects differential activation of target genes in response to different levels of hypoxia
HIF-mediated hypoxic impact - secretion of signalling molecules, metabolic changes, switch to aerobic glycolysis
Hypoxia leads to dyregulation of fibroblasts that may support tumourigenesis
Fibroblasts can be transformed into CAFs, leading to ECM remodelling that supports metastases
Different levels of hypoxia can trigger varying responses - eg. cell death in severe cases, immune regulation in lower levels of hypoxia
Immediate response may be angiogenesis to promote reoxygenated
In a 3D TME, tumour cells can be exposed to fluctuating O2 response leading to cycling hypoxia (acute hypoxia/anoxia followed by reoxygenation) and differential oxygen graduents for different time periods
Fluctuations can lead to differential biology based on hypoxia severity, duration, or whether it is terminated by cell death/reoxygenation
Hypoxia/ROS can cause permanent DNA damage
Warburg effect
Warburg effect - aerobic glycolysis in cancer
Reverse Warburg effect - two-compartment model where stromal cells are induced by cancer cells to undergo aerobic glycolysis and transfer products back to cancer cells to be used for OXPHOS
Acidosis
High metabolic demand of cancer cells —> accumulation of H+ in TME due to lactic acid production from the Warburg effect
Disorganised tumour vasculature prevents efficient wash-out of H+
Genes involved in mitochondrial energy metabolism facilitate cancer cell survival under acidotic stress
OXPHOS inhibition kills cancer cells in low pH conditions
This is because without oxygen normal cells can do glycolysis
However H+ negatively feeds back to inhibit glycolysis, thus cancer cells relying on aerobic glycolysis will die in low pH TME
Dysregulated pH is an emerging hallmark of cancer (low pH outside cells, higher pH inside - reversed pH gradient)
Carbonic anhydrase IV (CAIX) is a pH sensor upregulated for this
Proton transporters pump out H+, thus upregulated
Can cause immune suppression in TME due to low pH or decrease drug uptake
Vascularisation and angiogenesis
Compared to normal vasculature, tumour vessels exhibit immature hierarchy with discontinuous endothelial lining, incomplete pericyte coverage and leakiness —> elevated interstitial pressure, narrowed lumen, impaired O2/drug delivery to tumour
Tumour endothelial cells with overexpressed VEGF receptors replace the normal ones and make the vessels more sensitive to VEGF.
Antitumor immune function is retarded due to diminished T cell extravasation
Cells lacking O2/nutrients become nectrotic, thus angiogenesis is triggered to develop tumour vasculature