Exothermic & Endothermic Reactions
Energy is conserved in chemical reactions. The amount of energy in the universe at the end of a chemical reaction is the same as before the reaction takes place. If a reaction transfers energy to the surroundings the product molecules must have less energy than the reactants, by the amount transferred.
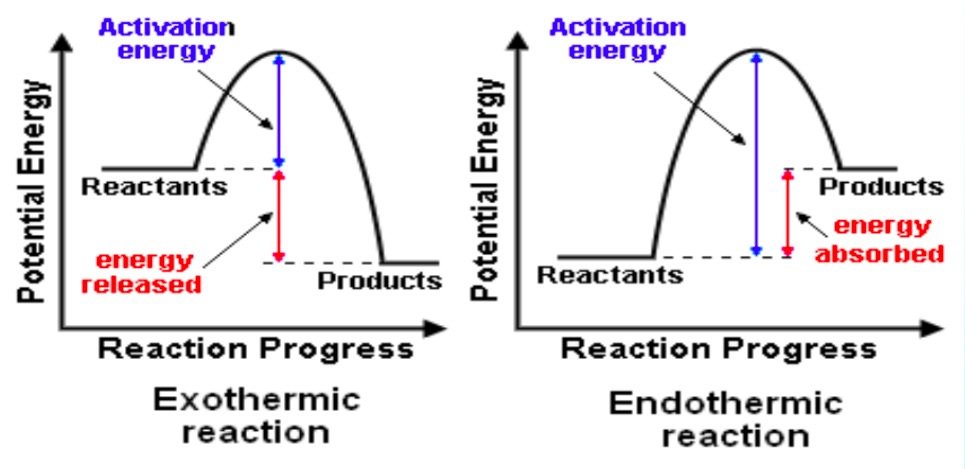
Exothermic
Energy released to surroundings → temperature increases.
Examples: combustion, neutralisation, oxidation reactions.
Reaction profile: products lower in energy than reactants.
ΔH is negative (because energy leaves the system).
Endothermic
Energy taken in from surroundings → temperature decreases.
Examples: thermal decomposition, citric acid + sodium hydrogen carbonate, photosynthesis.
Reaction profile: products higher in energy.
ΔH is positive.
Grade 9 insight:
If you release more energy (from making bonds) than you take in (breaking bonds) → EXOTHERMIC.
If you take in more energy (breaking bonds) than you release (making bonds) → ENDOTHERMIC.
Breaking bonds = endothermic
(energy must be put in)
Making bonds = exothermic
(energy released)
Reaction Profiles (Energy Diagrams)
Reactants
Products
Activation energy (Ea) → minimum energy needed to start the reaction.
Overall energy change (ΔH) → difference between reactants and products.
Endothermic → absorbs energy → solution loses heat → colder
Exothermic → releases energy → solution gains heat → hotter
Bond Energy Calculations
Energy change =
Energy required to break bonds – Energy released when bonds form
Because:
Breaking bonds = endothermic (requires energy)
Making bonds = exothermic (releases energy)
Example (Grade 9 standard):
H₂ + Cl₂ → 2HCl
Bond energies:
H–H = 436 kJ/mol
Cl–Cl = 243 kJ/mol
H–Cl = 431 kJ/mol
Calculate ΔH:
Energy in (breaking):
H–H + Cl–Cl = 436 + 243 = 679 kJEnergy out (forming):
2 × H–Cl = 2 × 431 = 862 kJΔH = 679 – 862 = –183 kJ/mol (exothermic)
What’s happening physically
Chemical reactions involve bond breaking and bond forming:
Breaking bonds = energy absorbed → endothermic
Forming bonds = energy released → exothermic
In a solution:
Energy absorbed from water → solution cools → endothermic
Energy released into water → solution warms → exothermic
💡 Key idea: the solution acts as the surroundings, so its temperature change tells you the energy flow.
Experimental setup
Use a polystyrene cup → minimizes energy loss to air
Thermometer → measures solution temperature precisely
Keep variables controlled:
Volume of reactants
Concentration of solutions
Initial temperature
Surface area of solids (if used)
Measure temperature at regular intervals → can plot temperature vs time graph for accuracy
How to tell endo vs exo
Endothermic: temperature drops → reaction absorbs energy from the solution
Exothermic: temperature rises → reaction releases energy into the solution
Grade 9 phrasing:
“The solution temperature decreases because energy is absorbed from the surroundings to break bonds in the reaction.”
Always reference energy transfer; saying “it feels colder” is not enough for full marks.
Data interpretation
Use temperature change (ΔT) to calculate approximate energy change in kJ if given volume and specific heat capacity.
Graph tip: steep drop/rise = fast reaction, gradual = slower reaction.