Skeletal Muscle I
Introduction to Skeletal Muscle
Mass: Constitutes a significant portion of body mass, approximately 40% in men and 30% in women.
Function: Beyond movement, active skeletal muscle releases substances called myokines and can release succinate.
Neuromuscular Junction (NMJ) and Initial Excitation
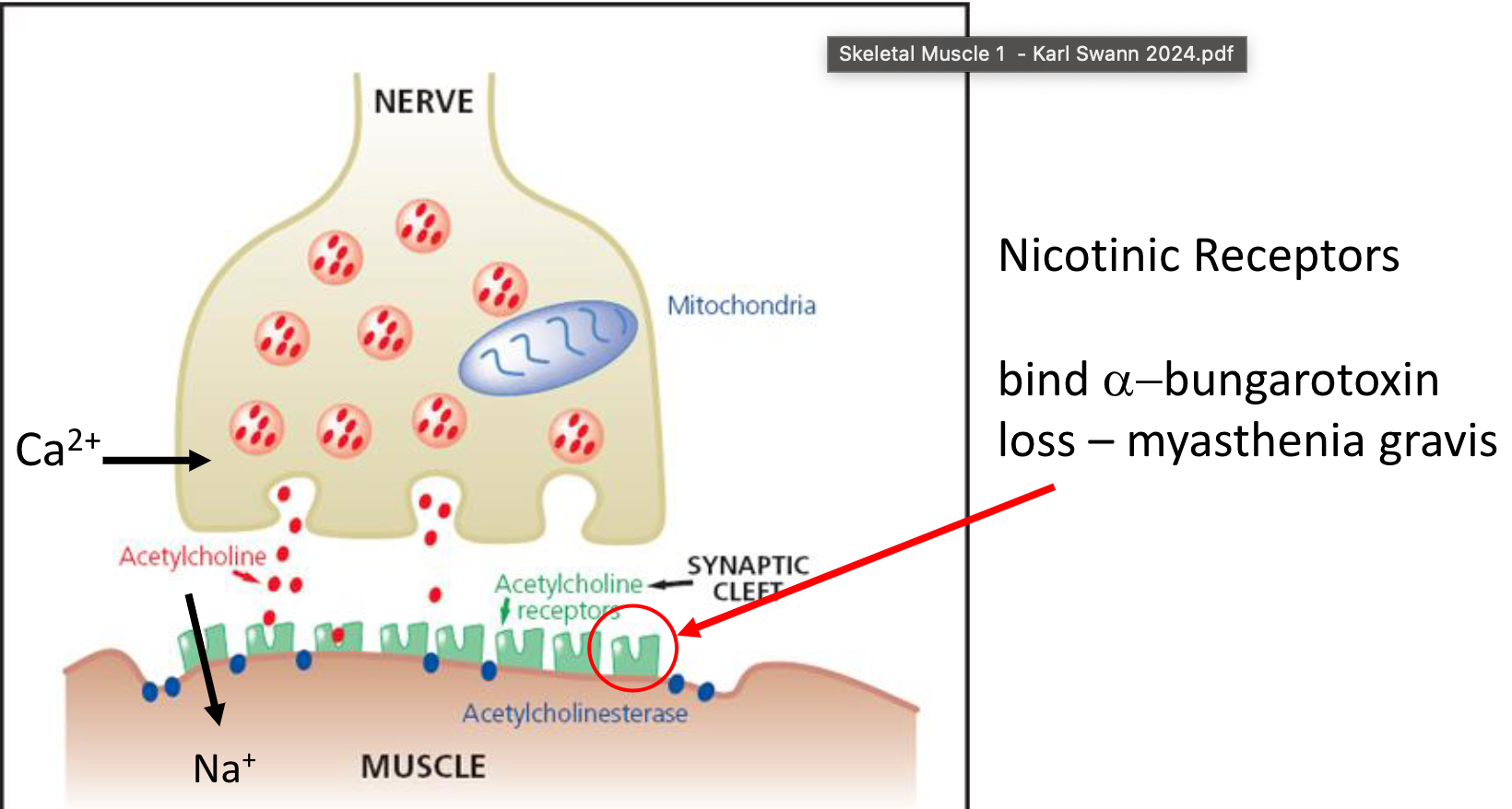
Structure Recap: The NMJ is the synapse where an efferent motor neuron's axon terminal (terminal button) connects with a muscle fiber.
Neurotransmission:
Arrival of a nerve impulse triggers calcium (Ca2+) influx into the axon terminal.
This causes synaptic vesicles containing acetylcholine (ACh) to fuse with the presynaptic membrane, releasing ACh into the synaptic cleft.
ACh diffuses across the cleft and binds to nicotinic ACh receptors (nAChRs) on the muscle fiber membrane (sarcolemma).
Note: α-bungarotoxin is a potent snake toxin that has a high affinity for the receptors and causes paralysis
Myasthenia Gravis is an autoimmune disorder that causes antibodies to bind irreversibly to recceptors causing muscle weakness and potentially paralysis.
Binding opens the nAChR channels, allowing sodium (Na+) ions to enter the muscle cell, causing depolarization.
Acetylcholinesterase in the synaptic cleft quickly breaks down ACh to terminate the signal.
Excitation-Contraction (EC) Coupling in Muscle Fiber
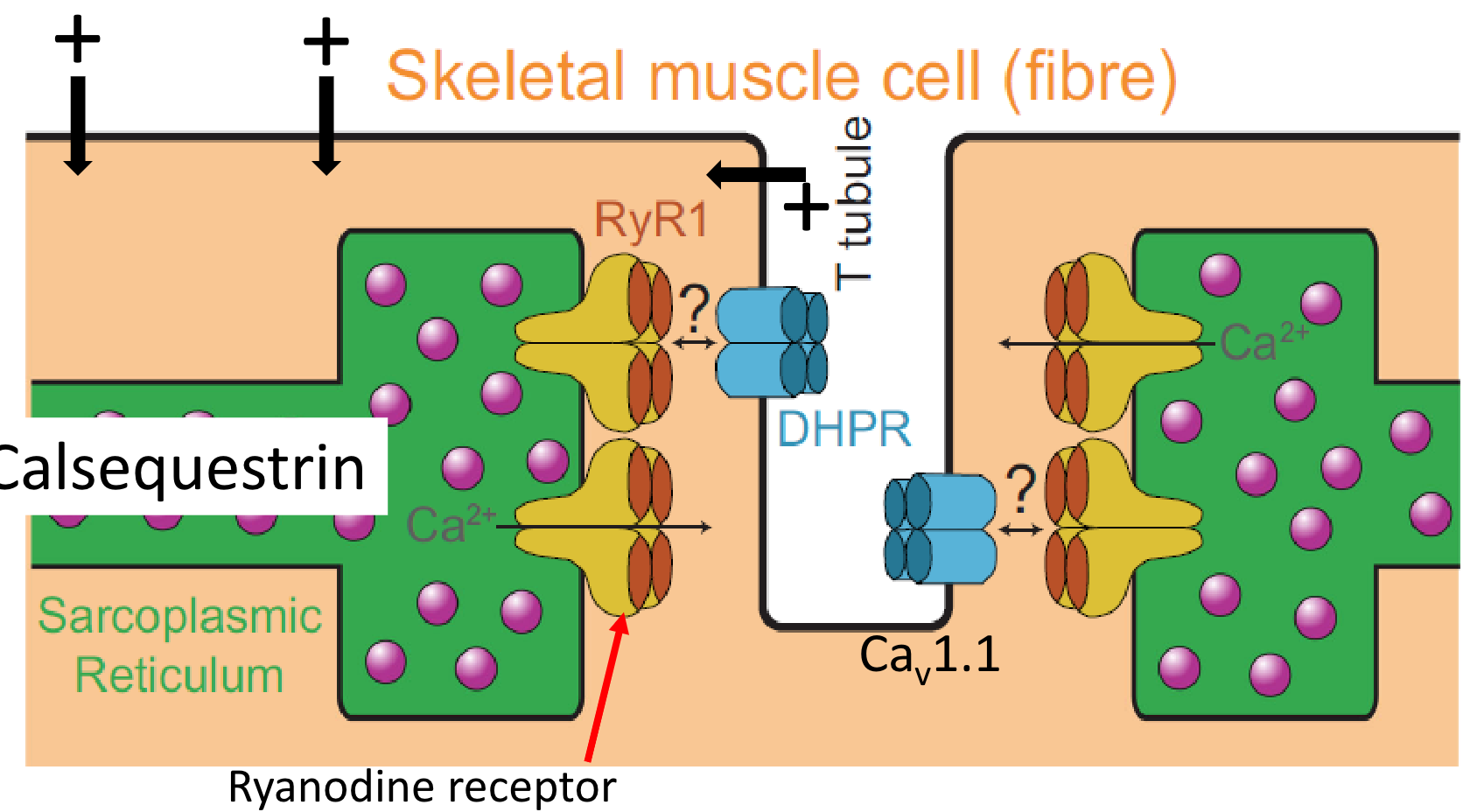
Sarcolemma and T-tubules: The depolarization initiated at the NMJ spreads across the sarcolemma and down transverse tubules (T-tubules), which are invaginations of the sarcolemma penetrating deep into the muscle fiber.
DHPR and RyR1: Within the T-tubule membrane are L-type calcium channels known as dihydropyridine receptors (DHPRs, specifically Cav1.1 in skeletal muscle). These are voltage sensors physically linked to ryanodine receptors (RyR1) located on the membrane of the sarcoplasmic reticulum (SR), the intracellular calcium store.
Calcium Release: Depolarization of the T-tubule causes a conformational change in the DHPR, which directly pulls open the RyR1 channel. This mechanical coupling allows Ca2+ stored within the SR (bound to calsequestrin) to flood into the sarcoplasm (the muscle cell cytoplasm). Note: This direct mechanical coupling is characteristic of skeletal muscle and differs from the Ca2+-induced Ca2+ release mechanism predominant in cardiac muscle.
The Contractile Apparatus: Sarcomeres
Structure: Skeletal muscle appears striated due to the highly organized arrangement of contractile proteins within sarcomeres.
Sarcomere: The basic contractile unit, spanning between two Z-lines (or Z-disks).
Filaments: Contains thick filaments (primarily myosin) and thin filaments (primarily actin, along with troponin and tropomyosin).
Bands and Zones:
A band: Region containing thick filaments (appears dark).
I band: Region containing only thin filaments, spanning across the Z-line (appears light).
H zone: Central region of the A band containing only thick filaments.
M line: Center of the sarcomere/H zone, contains proteins that anchor thick filaments.
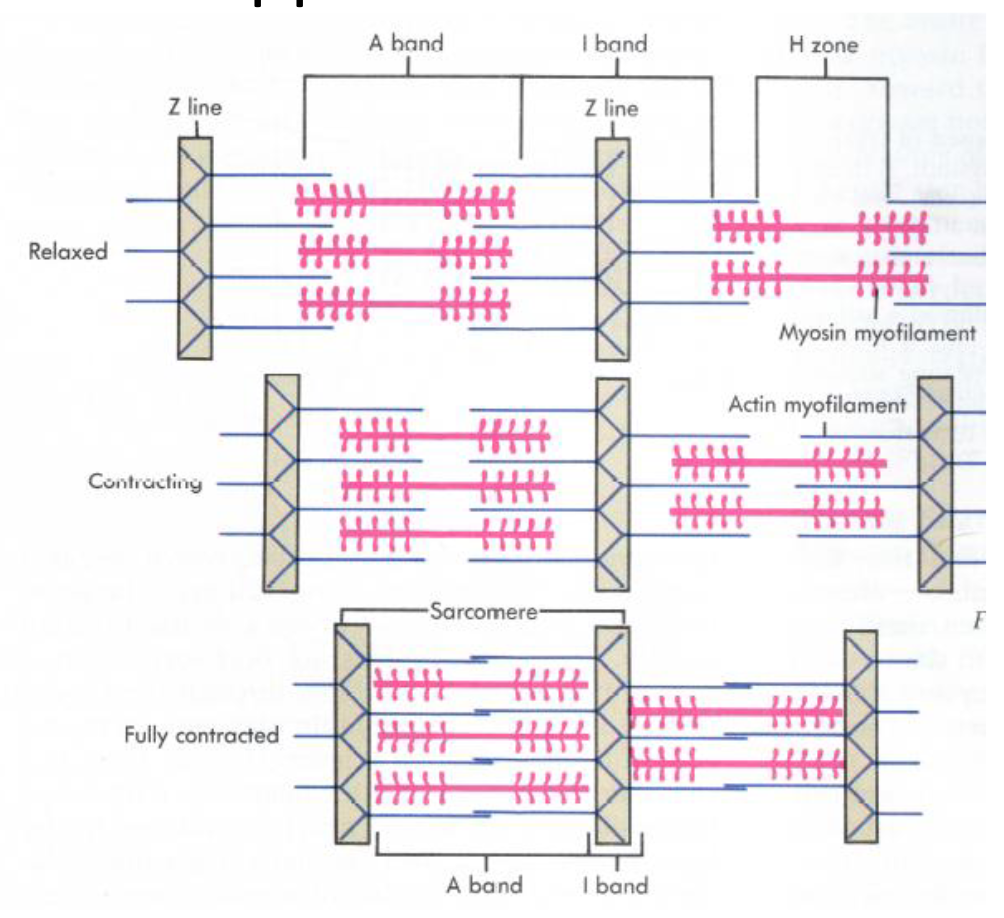
Sliding Filament Theory: During contraction, thin filaments slide past the thick filaments, pulling the Z-lines closer together. This shortens the sarcomere, I band, and H zone, while the A band length remains constant.
Molecular Mechanism of Contraction: The Cross-Bridge Cycle
Ca2+ stimulated Myosin contraction.
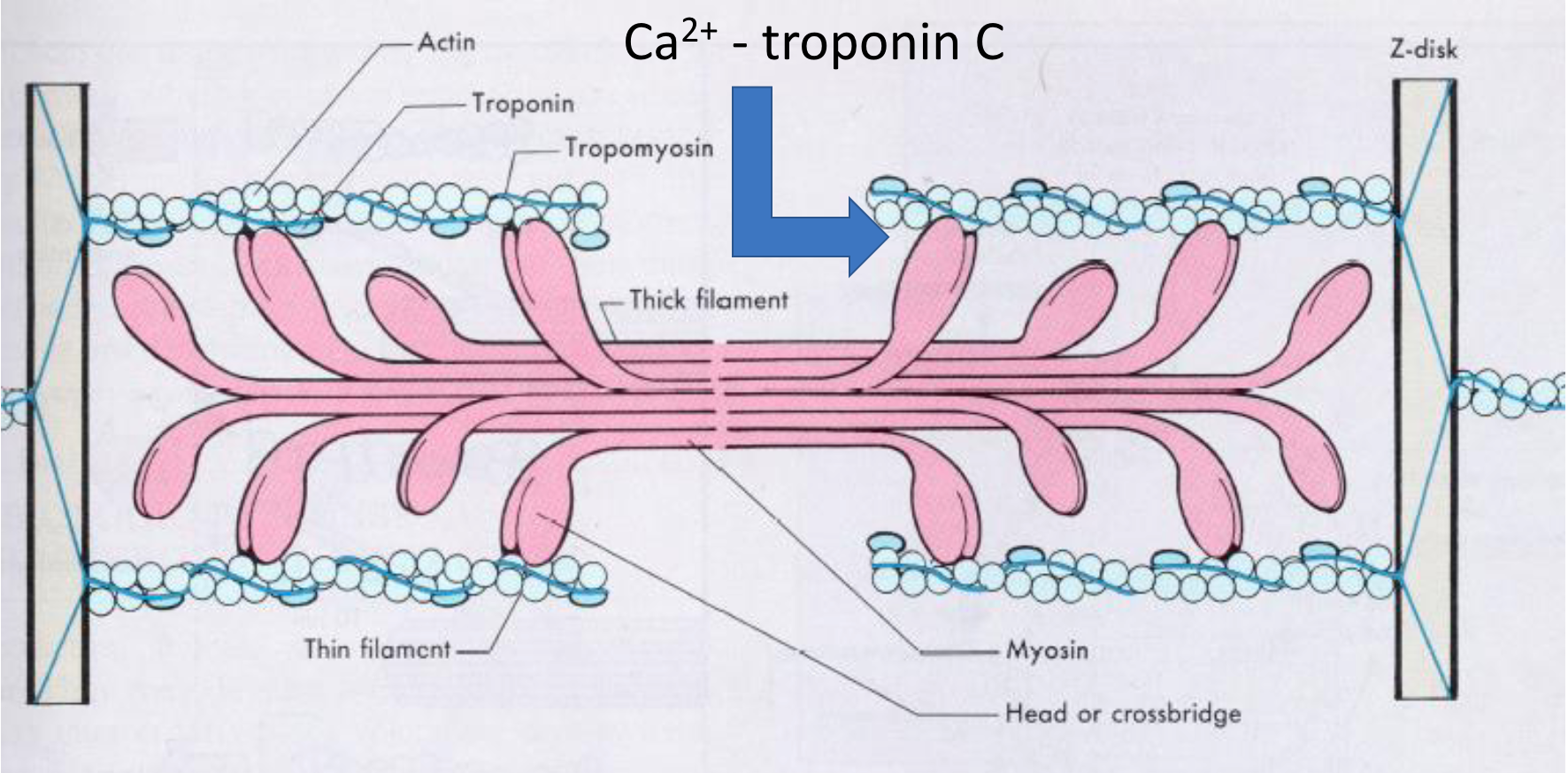
Resting State: In a relaxed skeletal muscle fiber, the protein tropomyosin lies along the actin thin filament, physically covering the sites where myosin heads need to bind. The troponin complex (specifically Troponin I) helps hold tropomyosin in this blocking position.
Calcium Binding: When the muscle cell is stimulated, calcium ions (Ca2+) are released from the sarcoplasmic reticulum. These Ca2+ ions bind to a specific subunit of the troponin complex called Troponin C (TnC).
Conformational Change: The binding of Ca2+ to TnC causes TnC to change its shape (a conformational change).
Tropomyosin Shift: This change in TnC's shape pulls on other parts of the troponin complex (specifically Troponin I), causing the entire troponin-tropomyosin complex to shift its position slightly on the actin filament.
Binding Sites Exposed: This movement physically rolls tropomyosin away from the myosin-binding sites on actin.
Contraction Initiation: With the binding sites now uncovered, the energized myosin heads can attach to actin, forming cross-bridges and initiating the power stroke that leads to muscle contraction.
Cross-Bridge Cycle (Stimulated by Ca2+ and requiring ATP):
Binding: Energized myosin heads (containing ADP and inorganic phosphate, Pi) bind to the exposed sites on actin.
Power Stroke: Binding triggers the release of Pi, causing the myosin head to pivot, pulling the thin filament towards the M-line. ADP is released during this step.
Detachment: A new ATP molecule binds to the myosin head, causing it to detach from actin.
Re-energizing: The myosin head hydrolyzes the ATP into ADP and Pi, using the released energy to return to its cocked, high-energy position, ready for another cycle.
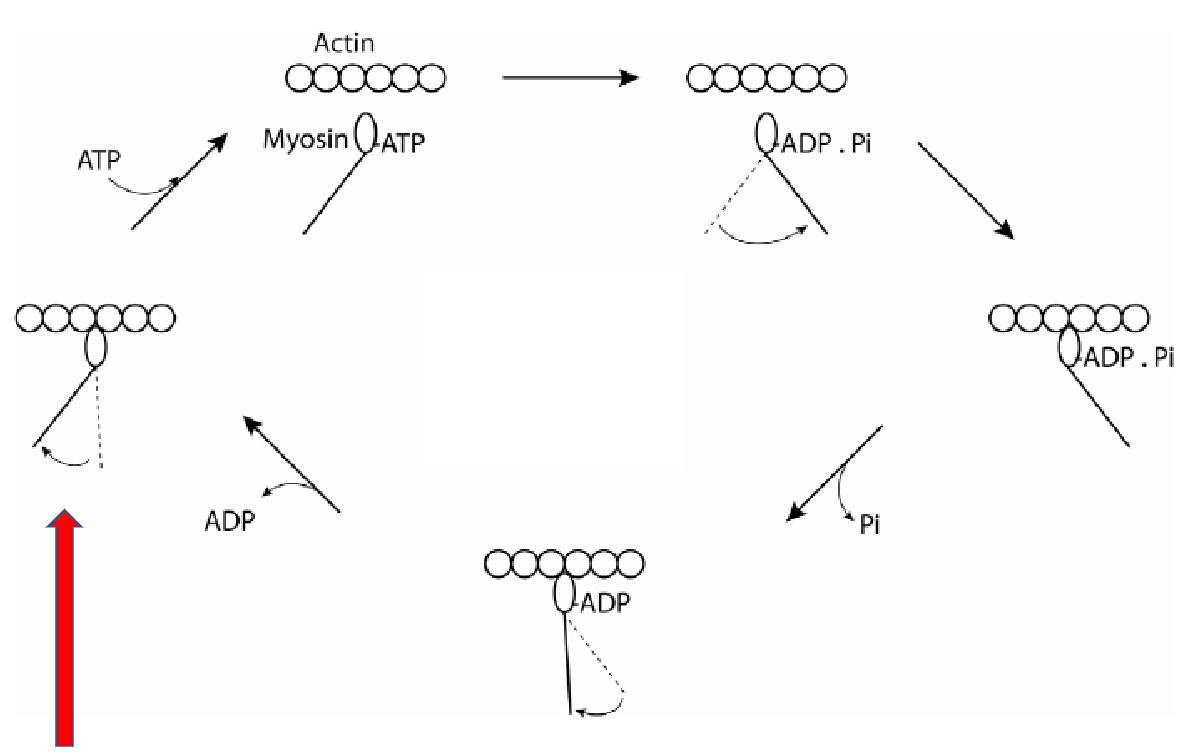
Rigor Mortis: Occurs after death when ATP production ceases. Without ATP, myosin heads cannot detach from actin, and high intracellular Ca2+ keeps binding sites exposed, resulting in muscle stiffness.
Temporal Relationship of Events
An action potential in the muscle fiber (lasting a few milliseconds) triggers a rapid but transient increase in myoplasmic (sarcoplasmic) Ca2+ concentration.
This Ca2+ binds to troponin, initiating the cross-bridge cycle.
The resulting force (tension) develops more slowly and lasts longer than the initial electrical and calcium signals.
ATP: The Energy Currency
Structure: Adenosine triphosphate (ATP) is the direct energy source for muscle contraction.
Energy Release: The hydrolysis of ATP to ADP and Pi releases energy. This reaction is kept far from equilibrium in the cell (~ ), providing a large driving force.
ATP Usage in Muscle: ATP is consumed by:
Myosin ATPase (~60%): Powers the cross-bridge cycle (detachment and re-energizing).
Ca2+ ATPase (SERCA) (~30%): Pumps Ca2+ back into the SR to allow muscle relaxation.
Na-K ATPase (~10%): Maintains ion gradients across the sarcolemma, essential for excitability/depolarisation.
Efficiency: Muscle contraction is roughly 25% efficient in converting ATP energy into mechanical work.
Muscle Energetics and ATP Homeostasis
ATP Turnover: Muscle ATP concentration is relatively high (~8 mM in muscle and ~1-2mM) and would be depleted quickly during exercise if not replenished. Resting turnover is ~1 min (typical of every cell in body), increasing 10-100 fold during exercise Note: higher concentration of ATP doesnt give more energy, as ATP is 10 orders of magnitude away from equilibrium (log relationship), it speeds up kinetic relationship.
Mechanisms for ATP Regeneration:
Phosphocreatine (PCr) System: PCr (~30 mM in muscle) acts as a rapid buffer. Creatine phosphokinase transfers the phosphate group from PCr to ADP, quickly regenerating ATP. This is the primary source for very short, intense bursts. >
Adenylate Kinase (Myokinase) Reaction: Combines two ADP molecules to form ATP and AMP. 2ADP>ATP+AMP
Glycolysis: Anaerobic breakdown of glucose (from blood or stored glycogen) to pyruvate, producing a small amount of ATP quickly. Pyruvate can be converted to lactate under anaerobic conditions.
Oxidative Phosphorylation: Aerobic metabolism in mitochondria. Uses oxygen to break down glucose, fatty acids, and amino acids, producing large amounts of ATP but more slowly than anaerobic pathways.
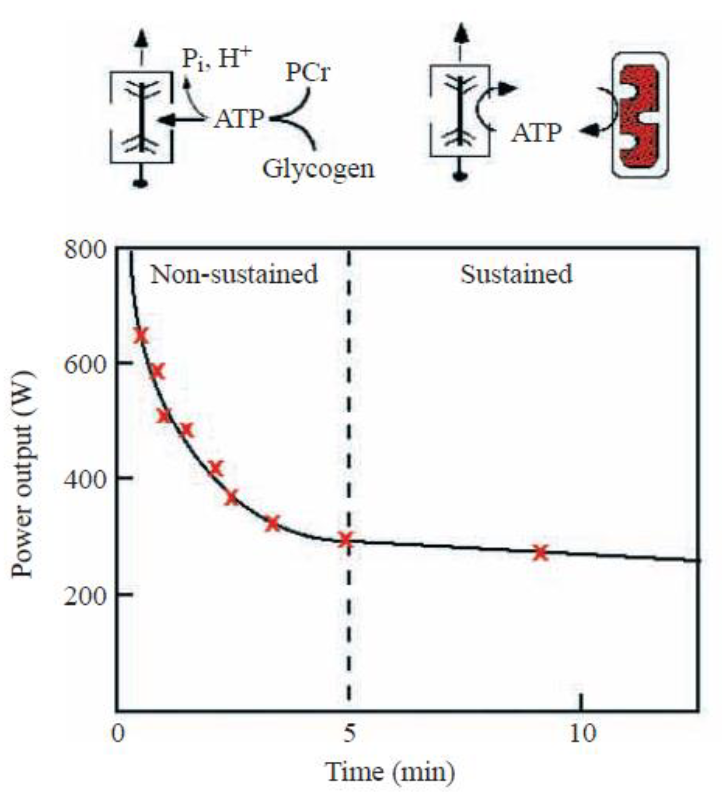
Energy Sources During Exercise:
Non-sustained (first few minutes): Primarily relies on stored ATP, PCr regeneration, and anaerobic glycolysis using stored glycogen. Power output is high but declines rapidly.
Sustained (longer duration): Relies increasingly on oxidative phosphorylation using oxygen and fuel substrates (glucose, fatty acids) delivered by blood flow, as well as muscle glycogen. Power output is lower but can be maintained longer. Myoglobin helps buffer oxygen within the muscle cell.
Muscle Fiber Types
Muscles contain a mix of fiber types, differing in their contractile and metabolic properties due to different protein isoforms (e.g., myosin heavy chains, SERCA, troponin). Note: can be found using staining.

Main Types:
Type I (Slow-twitch, Slow Oxidative):
Small motor units.
Rich in mitochondria and myoglobin (red appearance).
Relies on aerobic metabolism.
Contracts slowly, produces less force, but highly resistant to fatigue.
Used for posture and endurance activities.
Type IIA (Fast-twitch Oxidative-Glycolytic):
Intermediate size motor units.
Rich in mitochondria, myoglobin, and glycogen.
Combines fast contraction speed with significant oxidative capacity and glycolytic capacity.
Relatively resistant to fatigue.
Type IIX/D (Fast-twitch Glycolytic - Human equivalent of IIB):
Large motor units.
Rich in glycogen, fewer mitochondria/myoglobin (pale appearance).
Relies primarily on anaerobic glycolysis.
Contracts rapidly and forcefully but fatigues quickly.
Used for powerful, short-duration movements.
Type IIB (Fast-twitch Glycolytic - in rodents, not typically humans):
Similar properties to IIX but often faster/more powerful.

Studying Muscle Fibers
Methods:
Intact Single Fiber: Allows study of natural EC coupling but intracellular environment is less accessible. Microinjection can be used to introduce substances.
Skinned Fiber: The sarcolemma is removed or made permeable (using detergents like saponin, glycerol treatment, or mechanical means). This allows direct control and manipulation of the intracellular environment (e.g., setting Ca2+ concentration using buffers like EGTA) to study the contractile machinery directly.
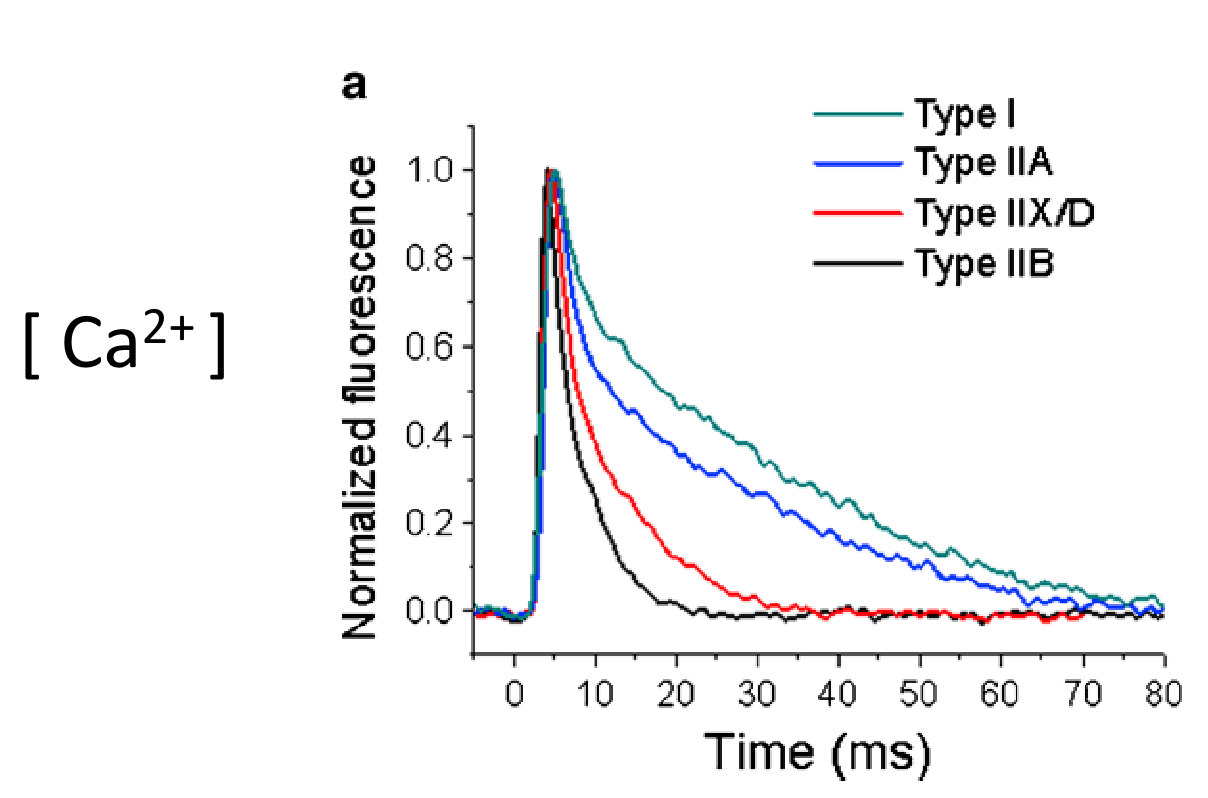
Fiber Type Differences in Ca2+ Handling:
Faster fiber types (IIA, IIX, IIB) generally show faster and larger Ca2+ transients (the temporary rise in intracellular Ca2+ following stimulation) compared to Type I fibers.
During tetanic stimulation (repeated high-frequency stimulation causing fused contraction), Ca2+ levels remain elevated, but the specific pattern and peak levels can vary between fiber types.
Fiber Type Differences in Ca2+ Sensitivity:
Skinned fiber experiments show that slow-twitch (Type I) fibers are generally more sensitive to Ca2+. They achieve a given relative force at a lower Ca2+ concentration compared to fast-twitch fibers.