CHEMICAL BONDING
Compounds- when elements combine in fixed ratios
Chemical bonds- what hold compounds together
3 TYPES OF CHEMICAL BONDING
Metallic- between 2 metals
Metal atom arrangement
Electron sea model- model used to describe solid metals
Valence electrons are freely flowing; they arent associated with a single atom and are instead shared within the metal cations
the metal is held together by the strong attraction between the cations and electrons. This is metallic bonding
Properties of metals (prove how metallic bonding is strong)
High melting point- the temperature where the metal goes from solid to liquid
Liquids are less compact than solids, therefore if the melting point is high, the bond must be strong too since the atoms wouldnt move apart (like how you need for it to be a liquid)
Malleability- ability of an object to be shaped into sheets without breaking
the metal is deformed, but the free flow of electrons keep the positive atoms from repelling each other
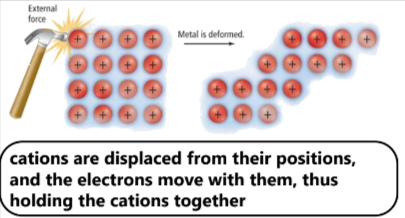
Ductility- ability of an object ot be shaped into wires or rods without breaking
has the same mechanics as malleability, the ‘sea’ of electrons keep the positive atoms from repelling each other, which means it will break
Conductivity- ability for heat/electricity to pass through
the mobile sea of electrons help in the distribution of heat and electricity, since they are constantly moving
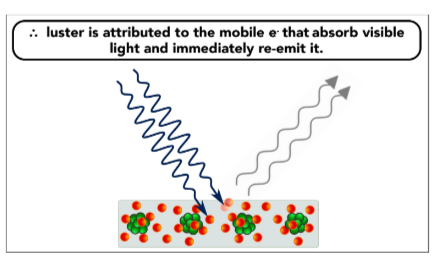
Luster- ability to reflect light, giving a shiny appearance
the mobile electrons absorb light and immediately re-emit it
Ionic- between a metal and a nonmetal
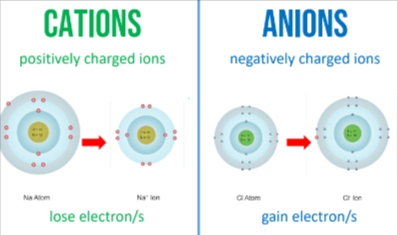
Ions- atoms or groups of atoms that are electrically charged
Cations- positively charged
Anions- negatively charged (a n=negative)
THIS MEANS: metals tend to be negatively charged while nonmetals tend to be positively charged, opposite charges attract each other
Properties of ionic compounds
Crystalline- ionic compounds create crystal-like lattice structures (imagine salt)
these crystals form because of the alternating positive and negative ions
Hard and brittle- this means that the structure is solid but it is easily crushed
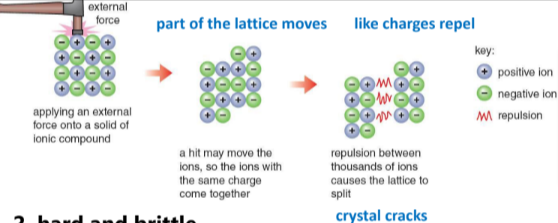
the crystal is arranged in alternating positive and negative ions, when an external force presses on it, it moves, displacing the alternating ions. when this happens, positive ions and negative ions will be next to each other, causing them to repel each other (break)
High melting and boiling points- you need high temperatures to seperate the compound
it is similar to metallic compounds in this way, the bond between metals and non-metals cant be easily seperated through temperature.
Conducts electricity- ionic compounds can conduct electricity when it is dissolved in water
this is because you partially expose the metal part of the compound, and the ions can also move around more easily. this makes it easier to conduct electricity (the same for heat)
Octet rule
atoms like having a full shell (8 valence electrons) like noble gases. to achieve this in a compound, they borrow each others’ electrons
5 and up: tend gain electrons
4 and down: tend to lose electrons
Valence electrons
Valence electrons are electrons on the outermost orbit/ring of the atom
Shielding electrons
Shielding electrons are electrons in the inner orbit/ring of the atom
Lewis structure
This is how you represent the valence electrons in an atom (represented by dots)
Ionic bonds
attraction lies in the 2 oppositely charged atoms (which is what bond the compound together)
Covalent- between 2 non-metals
HOW TO WRITE LEWIS STRUCTURE
First start off with the 4 outer circles in a clockwise motion
Once you complete those 4, you go back again
you can do this in any order
always write the metal first
EXPLANATIONS FOR WRITTEN EXAMPLES: