physiology
Lecture 1
Signal Transduction
is the process by which cells communicate and respond to external stimuli
Only cells expressing specific receptors for a ligand can respond to that ligand
Ligand-Receptor interaction
Ligands, such as hormones or signaling molecules, bind to specific receptors on the cell membrane or inside the cell.
Receptor activation leads to a cascade of intracellular signalling events
Interaction between ligands and receptors is highly specific, with each ligand binding to its corresponding receptor with high affinity
Steps of Ligand-receptor interaction
Ligand Binding: The ligand, which can be a hormone or signaling molecule, binds to a specific receptor on the cell membrane or inside the cell.
Receptor Activation: Upon ligand binding, the receptor undergoes a conformational change, leading to its activation.
Signal Transduction: The activated receptor triggers a series of intracellular signaling events, which can involve the activation of various proteins or enzymes.
Cellular Response: The signaling cascade ultimately leads to a specific cellular response, such as changes in gene expression, cell growth, differentiation, or other physiological processes.
Types of receptors
7-Transmembrane (TM) receptors
Intracellular receptors
Tyrosine kinase receptors
7-Transmembrane (TM receptors)
Ligand binds to receptor, activating G-proteins and effectors, leading to a cellular response
Intracellular receptors
Hormone enters the cell, binds to specific receptor, translocates to the nucleus, activates gene transcription, and promotes protein synthesis
Tyrosine kinase receptors
Membrane receptors that have intrinsic tyrosine kinase activity in their intracellular loop
Activate two pathways: PI3K-Akt and Ras-MAPK pathways.
What are the steps of the PI3K-Akt pathway?
Ligand Binding and Receptor Activation:
When a growth factor binds to its receptor on the cell membrane, it induces a conformational change in the receptor, leading to receptor dimerization.
The receptor undergoes autophosphorylation, activating its tyrosine kinase activity.
Activation of PI3K:
Phosphorylated growth factor receptors recruit and activate phosphatidylinositol 3-kinase (PI3K) by binding to its SH2 domain .
PI3K, a lipid kinase, converts membrane phospholipids to 3-phosphoinositides (PIP₃), specifically phosphatidylinositol (3,4,5)-trisphosphate.
Activation of PDK and Akt:
PIP₃ binds and activates phosphoinositide-dependent kinase (PDK).
PDK phosphorylates and activates Akt, a serine/threonine kinase (45).
Cellular Response:
Activated Akt phosphorylates intracellular proteins, leading to cellular responses such as increased cell proliferation and survival.
Akt can also induce metabolic responses, such as the translocation of GLUT4 glucose transporters to the cell membrane, promoting glucose uptake
What are the steps of the Ras-MAPK pathway?
Ligand Binding and Receptor Activation:
When a ligand (such as insulin or a growth factor) binds to its receptor on the cell membrane, it induces a conformational change in the receptor, leading to receptor activation (52, 50).
The activated receptor undergoes autophosphorylation, enhancing its tyrosine kinase activity.
Activation of Ras:
Phosphorylated receptor recruits and activates insulin receptor substrate-1 (IRS-1) or other adaptor proteins.
Phosphorylated IRS-1 binds to the Grb2-SOS complex, leading to the activation of Ras.
Grb2, via its SH2 domain, binds to phosphorylated residues on IRS-1, bringing SOS (a GTP/GDP exchanger) to the plasma membrane to activate Ras by converting it to a GTP-bound form.
Activation of MAPK Cascade:
Activated Ras binds to Raf kinase and activates it.
Raf kinase phosphorylates MAP kinase kinase (MEK), leading to its activation.
Activated MEK phosphorylates MAP kinase (MAPK), also known as ERK (extracellular signal-regulated kinase), activating it.
Cellular Response:
Activated MAPK (ERK) translocates to the nucleus and phosphorylates various transcription factors, leading to changes in gene expression and cellular responses.
Role in Cell Proliferation and Survival:
The Ras-MAPK pathway is crucial for regulating cell proliferation, survival, and differentiation in response to extracellular signals, such as growth factors
Dysregulation of this pathway can contribute to diseases like cancer, where overactivation of Ras and MAPK signaling leads to uncontrolled cell growth and proliferation.
How does the P13K-Akt pathway affect cancer?
Over activation can lead to cell proliferation leading to cancer
Ligand Binding and Receptor Activation:
When a ligand (such as insulin or a growth factor) binds to its receptor on the cell membrane, it induces a conformational change in the receptor, leading to receptor activation (52, 50).
The activated receptor undergoes autophosphorylation, enhancing its tyrosine kinase activity.
Activation of Ras:
Phosphorylated receptor recruits and activates insulin receptor substrate-1 (IRS-1) or other adaptor proteins (52, 50).
Phosphorylated IRS-1 binds to the Grb2-SOS complex, leading to the activation of Ras.
Grb2, via its SH2 domain, binds to phosphorylated residues on IRS-1, bringing SOS (a GTP/GDP exchanger) to the plasma membrane to activate Ras by converting it to a GTP-bound form (52, 50).
Activation of MAPK Cascade:
Activated Ras binds to Raf kinase and activates it.
Raf kinase phosphorylates MAP kinase kinase (MEK), leading to its activation.
Activated MEK phosphorylates MAP kinase (MAPK), also known as ERK (extracellular signal-regulated kinase), activating it (52, 50).
Cellular Response:
Activated MAPK (ERK) translocates to the nucleus and phosphorylates various transcription factors, leading to changes in gene expression and cellular responses (52, 50).
Role in Cell Proliferation and Survival:
The Ras-MAPK pathway is crucial for regulating cell proliferation, survival, and differentiation in response to extracellular signals, such as growth factors (52, 50).
Dysregulation of this pathway can contribute to diseases like cancer, where overactivation of Ras and MAPK signaling leads to uncontrolled cell growth and proliferation.
Effect of Insulin of MAPK pathway
insulin influences the MAPK pathway by initiating a signaling cascade that involves the activation of IRS-1, recruitment of the Grb2-SOS complex, activation of Ras, and subsequent activation of the MAPK cascade.
This pathway plays a crucial role in mediating cellular responses to insulin, including metabolic effects such as glucose uptake and gene expression regulation.
Ligand Binding and Receptor Activation:
Insulin binds to its receptor on the cell membrane, leading to a conformational change in the receptor and subsequent autophosphorylation.
The autophosphorylation of the insulin receptor enhances its tyrosine kinase activity.
Activation of Insulin Receptor Substrate-1 (IRS-1):
Phosphorylated insulin receptor recruits and phosphorylates insulin receptor substrate-1 (IRS-1), a key adaptor protein in insulin signaling.
Phosphorylated IRS-1 serves as a docking site for downstream signaling molecules.
Recruitment of Grb2-SOS Complex:
Phosphorylated IRS-1 binds to the Grb2-SOS complex, which consists of Growth factor receptor-bound protein 2 (Grb2) and Son of Sevenless (SOS).
Grb2, through its SH2 domain, recognizes and binds to phosphorylated IRS-1, facilitating the recruitment of SOS to the plasma membrane.
Activation of Ras:
The Grb2-SOS complex activates Ras by promoting the exchange of GDP for GTP on Ras, leading to Ras activation.
Activated Ras then initiates downstream signaling events in the MAPK pathway.
Activation of MAPK Cascade:
Activated Ras binds to Raf kinase and activates it.
Raf kinase phosphorylates MAP kinase kinase (MEK), leading to MEK activation.
Activated MEK phosphorylates MAP kinase (ERK), resulting in the activation of ERK.
Cellular Response:
Activated ERK translocates to the nucleus and phosphorylates transcription factors, thereby regulating gene expression and influencing cellular responses.
Metabolic Effects:
In fat and skeletal muscle cells, insulin-activated Akt (part of the PI3K-Akt pathway) leads to GLUT4 glucose transporter translocation to the plasma membrane, increasing glucose uptake
Cellular response
The signaling cascade initiated by ligand-receptor interaction results in specific cellular responses, such as changes in gene expression, cell growth, or differentiation.
Key players in Signal Transduction
Tyrosine Kinase receptors
Calmodulin
What do Tyrosine Kinase receptors do?
Play a crucial role in cell signaling by phosphorylating tyrosine residues on target proteins
What does Calmodulin do?
Calcium-binding protein involved in various cellular processes
What is cGMP (cyclic guanosine monophosphate)
It’s a second messenger in intracellular signaling events initiated by activation of certain hormone and neurotransmitter receptors.
Mediates cellular responses through the activation of specific cGMP-dependent protein kinases (PKG).
Lecture 2
Resting Membrane potential
It is maintained by the unequal distribution of ions, with higher concentrations of Na+ outside and K+ inside the cell
Na+/K+ pump plays a crucial role in maintaining this gradient by actively pumping Na+ out and K+ in, against their concentration gradients.
What are typical resting membrane potentials?
Large nerve & skeletal muscle fibers: -90 mV
Small nerve & skeletal muscle fibers: -40 to -60 mV
Membrane potential
refers to the electrical potential difference across a cell membrane.
essential for various cellular functions, including signal transmission and maintaining cell integrity.
Generating and measuring membrane potentials
Can be generated by the movement of ions across the membrane through ion channels and pumps.
It can be measured using a voltmeter by inserting an electrode inside the cell
How do membrane potentials affect excitable cells?
A concentration difference of ions across the membrane creates an electrical potential that allows excitable cells to propagate action potentials
What is the Nernst potential?
is the equilibrium potential at which the diffusion of an ion is balanced by its electrical gradient, preventing net ion movement
Action potential
is a rapid and transient change in membrane potential that allows for the propagation of signals along neurons.
It involves a series of depolarization and repolarization phases, driven by the opening and closing of ion channels.
The action potential curve typically consists of depolarization, repolarization, and hyperpolarization phases
Myelinated axons
Myelinated axons are nerve fibers surrounded by a myelin sheath, which acts as an insulator and speeds up the conduction of action potentials.
Nodes of Ranvier are the unmyelinated gaps along the axon where action potentials are regenerated, allowing for saltatory conduction
Parts of a neuron
Cell body/soma
Dendrites: branching processes coming off of soma. Gets signals from neurons and sends them to axon
Axon: generates action potential and sends it to next cell (tail)
Action potential & dendrites
stimulus causes action potential in soma
dendrites have a high concentration of potassium leak channels When they are stimulated there is a grater excitatory/inhibitory effect
Myelin sheath
surrounds axon
Diseases of myelin sheath
In demyelinating disorders, the myelin sheath deteriorates
In multiple sclerosis, scattered progressive demyelination of axons in CNS results in loss of motor control
Schwann cell
envelops axon and rotate around it. has multiple layers of plasma membrane with sphingomyelin (insulator that decreases ion flow)
Nodes of Ranvier
Interruptions of myelin sheath every 1-3mm on axon
easy ion flow & action potential only happens at nodes
Myelinated & Unmyleinated fibers
Large fibers: unmyelinated
Small fibers: unmyelinated
Saltatory conduction
conduction of action potentials from node to node in myelinated fibers
increases velocity of nerve transmission
Facilitation
if another stimulus comes, it would be easy for neuron to develop another action potential
Synaptic fatigue
defense mechanism against exhaustion of neurotransmitters in synaptic terminals
Toxin blocking pumps
Certain toxins can block ion pumps, disrupting the maintenance of membrane potential
Includes Saxitoxin, TEA and TTX
TTX
blocks sodium channel
found in ovaries of blowfish
Saxitoxin
Sodium channel blocker found in tissues of shellfish
Can have life-threatening paralysis
Tetraethylammonium (TEA)
Blocks potassium pump
Lecture 3
Action potential phases
Resting: resting membrane potential (-)
Threshold: action potential occurs because of sodium channel opening
Depolarization: positive change in membrane potential [closer to 0] because of influx of sodium
Repolarization: returning to negative membrane potential because of efflux of potassium
Refractory period: can be absolute or relative. it limits the amount of action potentials that can happen at a time
absolute: interval of time a normal impulse can’t re-excite an already excited area of membrane
sodium channels closed and can’t be reopened until repolarized
relative: interval of time that an excitable tissue is more difficult to stimulate
before returned to resting potential, channels are inactivated and need a stronger than normal stimulus
Threshold & Action potential
EPSP (Excitatory post synaptic potential)
causes depolariation
moves toward threshold of action potential
IPSP (Inhibitory postsynaptic potential)
causes hyperpolarization
moves away from causing action potential
PM has more neurons is more - than RMP so neurotransmitter modify channels so ions can move into the cell
Summation
Spatial summation
postsynaptic potentials happen by activating many terminals on widely spaced areas of membrane
Temporal summation
successive postsynaptic potentials
Action potentials in neurons
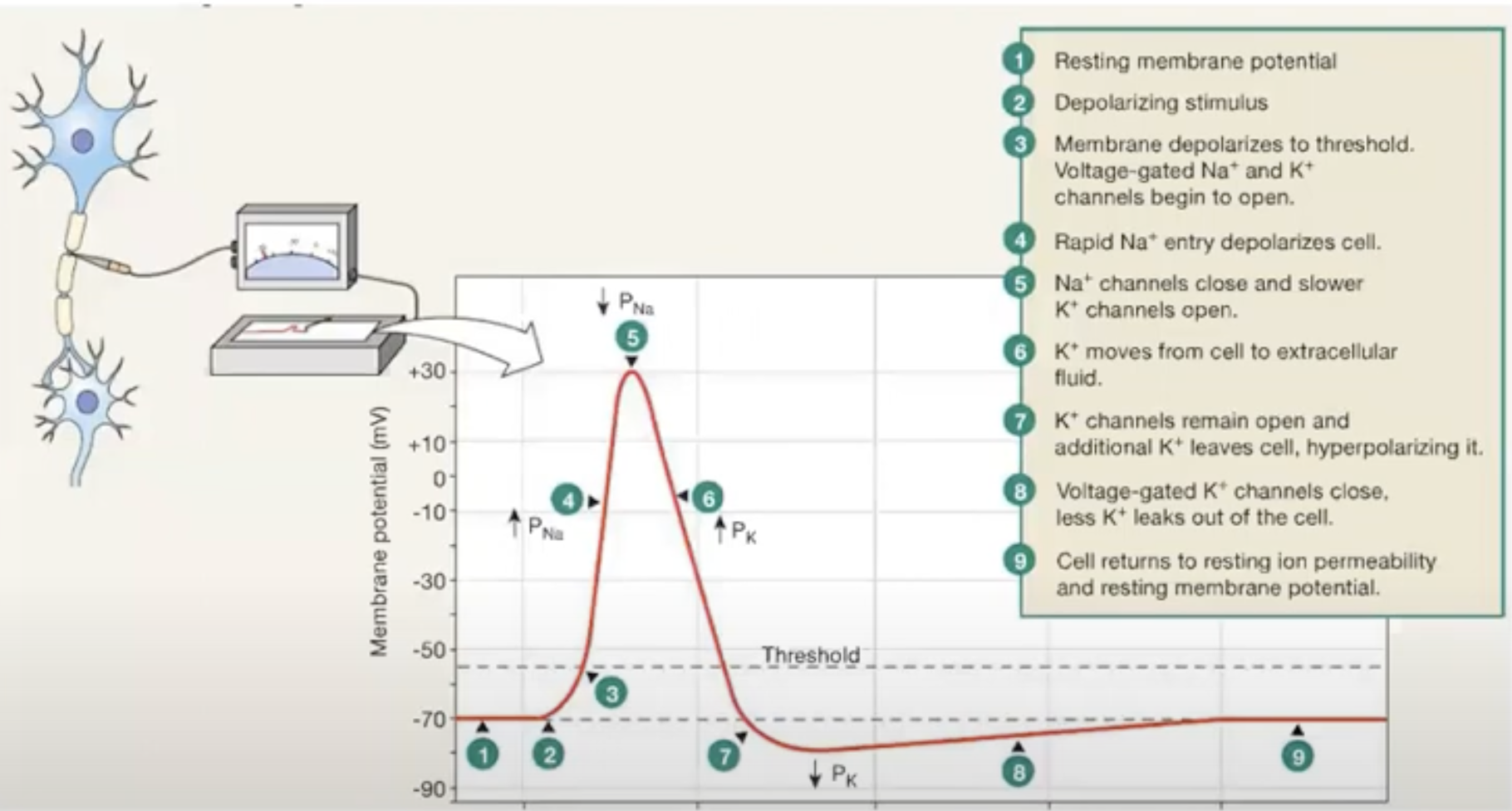
Non-active neuron cells have a RMP of -70 mV
Depolarizing stimulus makes sodium rise in cell, making MP more positively charged (bc sodium is +)
It reaches threshold membrane potential of -55 mV (when channels on axon open at a particular voltage)
Voltage gated sodium channels open at this threshold
Sodium outside of cell rushes inside of cell, causes membrane potential to increase and depolarize.
It peaks at +30 mV and sodium channels will close, and potassium channels will open and K+ will leave the cell
this makes the membrane potential to become more negative, as K+ is +
This is repolarization
*More potassium leaves the cell than more sodium that was able to enter the cell bc potassium channels are slower to close
Hyperpolarization occurs as membrane potential becomes more negative than it was at the very beginning (cause of slow K+ channels)
Potassium leakes out of cell through leak channels, making membrane potential go back to it’s RMP
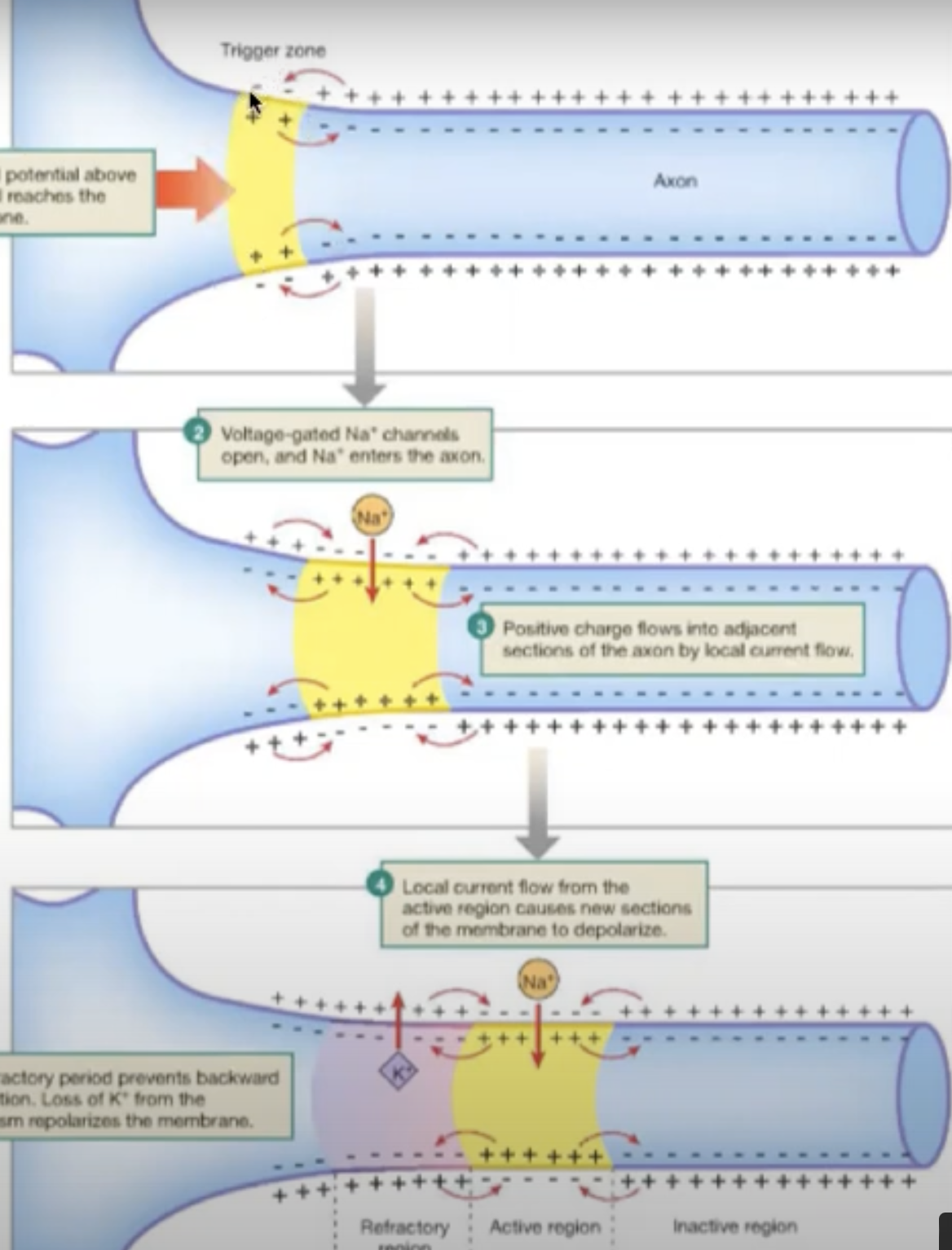
Positive current from the action potential in one end of the neuron will make adjacent sodium channels open
this current spreads to part to part of axon (moves down the axon)
channels in reverse will be hyperpolarized so you cannot make an action potential, so action potential moves in forward direction only
All or nothing principle
The an action potential has been elected on one point on the plasma membrane it will travel over the whole membrane [or not at all if conditions aren’t right]
Action Potentials in Myocardial Contractile Cells
similar to action potentials in neurons, but they have longer action potentials because of calcium entering the cell
action potential is longer because of influx of CA2+ in stage 2
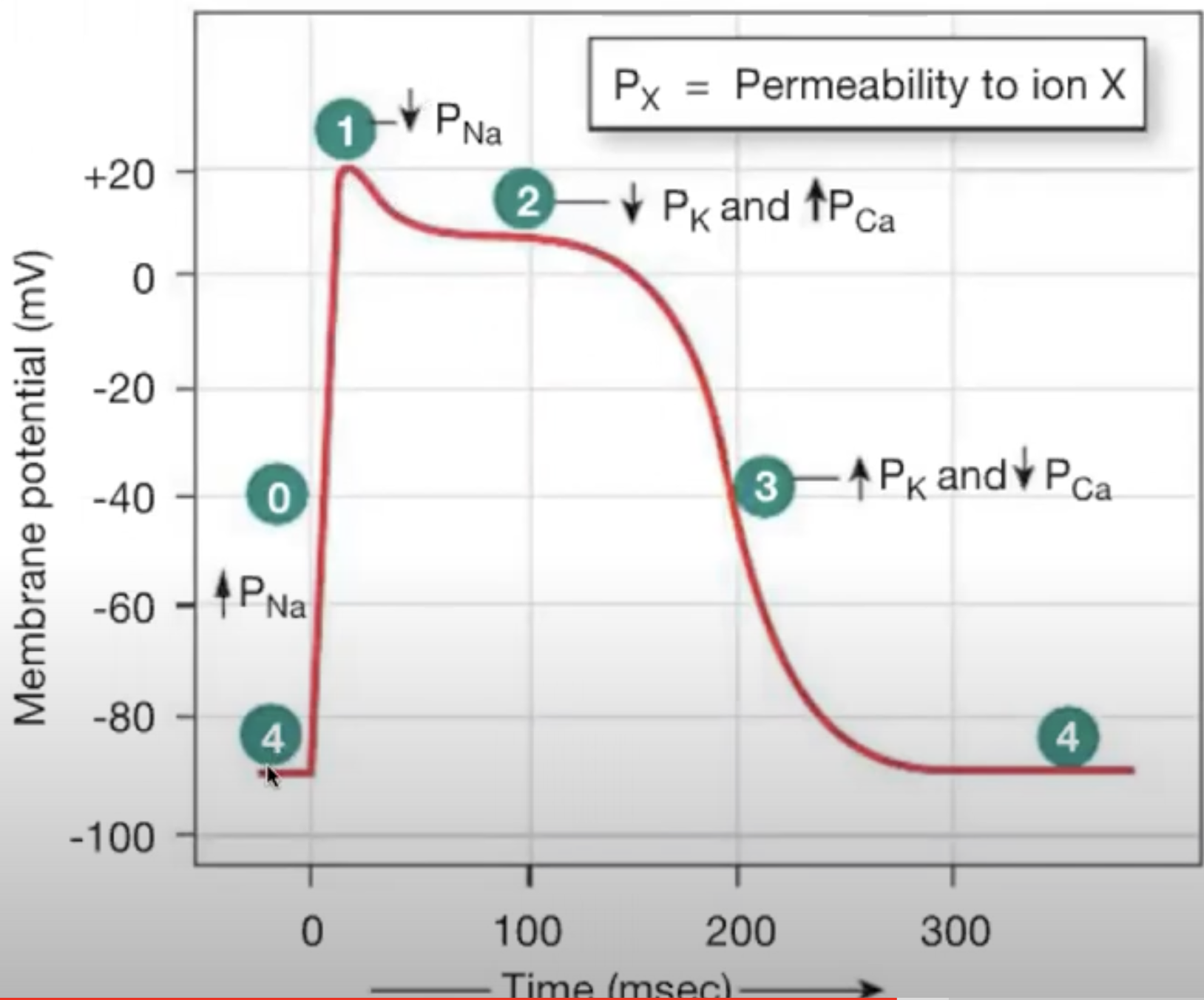 ..
..After depolarization, there is a plateau and then it repolarizes
Starts at phase 4
Myocardial contractile cells have RMP of -90mV
Depolarization occurs quickly because of open voltage-gated sodium channels
reaches +20 mV
sodium channels in cardiac cells open faster than in neurons
Sodium channels close and K+ channels open. K+ leaves the cells and leads to initial depolarization (little dip before plateau)
Calcium channels also open on cell membrane. K+ leaves cells, and CA2+ enters the cell. (Efflux of K+ and influx of CA2+ causes plateau)
Plateau takes long time to repolarize because of calcium influx [Calcium pump is slow to activate]
Calcium channels will close leading plateau to end. K+ channels are still open, causing K+ to leave the cell. this causes rapid depolarization
Refractory Period in myocardial contractile cells
Refractory period is long just like the action potential
Lecture 4
Muscle filaments
Thick filaments: myosin [protein]
Thin filaments: actin [protein]
Muscle fibre anatomy
Sarcomeres = unit of muscle
Z disk = where thin actin filaments attach to each other
I bands = only occupied by thin actin filaments
A bands= regions that have entire lengths of thick myosin filaments
H zones = only occupied by thick myosin filaments
M lines = divides each A band into half
Myosin
A motor protein
has a tail and two heads (paired)
Hinge region connects tail and head regions
makes heads swivel around attachment to actin
heads have binding site for Actin & ATP
Actin
Repeating sequence of G-CTIN units that make polymers called F-Actin
2 F-actin chains twist to make an actin filament
Troponin protein binds Calcium
Tropomyosin: a long protein that is controlled by troponin
G-Actin has a myosin binding site that’s covered by tropomyosin
Myosin can bind but it will be weak
When calcium binds to troponin, it moves tropomyosin and exposes the myosin binding sites
Myosin Power stroke [Binding of Myosin and Actin]
When calcium enters cell, calcium in cytosol increases and calcium binds to troponin
This binding will make tropomyosin to move from actin’s myosin binding site
Myosin head will binds strongly to actin and makes a power stroke (head swivels forward and pulls actin filaments together to shorten muscle fibre)
In the power stroke the actin moves toward centre of sarcomere
Energy comes from ATP
ATP for myosin-actin power stroke
Myosin will be in rigor state when it tightly binds to actin
ATP will bind to the myosin head which makes myosin let go of actin (muscle relaxes)
Myosin hydrolyzes ATP into ADP + Pi which stay bound to myosin.
This rotates myosin head into upright position so it can bind weakly to actin
Calcium enters cell and binds to troponin attached to tropomyosin
It moves tropomyosin out of the way to myosin head can bind tightly to actin
Myosin head binds tightly and swivels forward on actin filament and pulls it together [power stroke]
when myosin head power strokes (swivels forward), phosphate is released
Myosin head releases ADP and power stroke ends
nothing is bound to myosin head apart from actin
actin is only released with ATP. No ATP = muscle stays contracted
cells need to make energy to uncontract muscle
Excitation- Contracting coupling
Details how electrical signals lead to contraction of muscles
Action potentials initiate the contraction process
these potentials come spontaneously from pacemaker cells of heart
Muscle contraction [right side of photo]
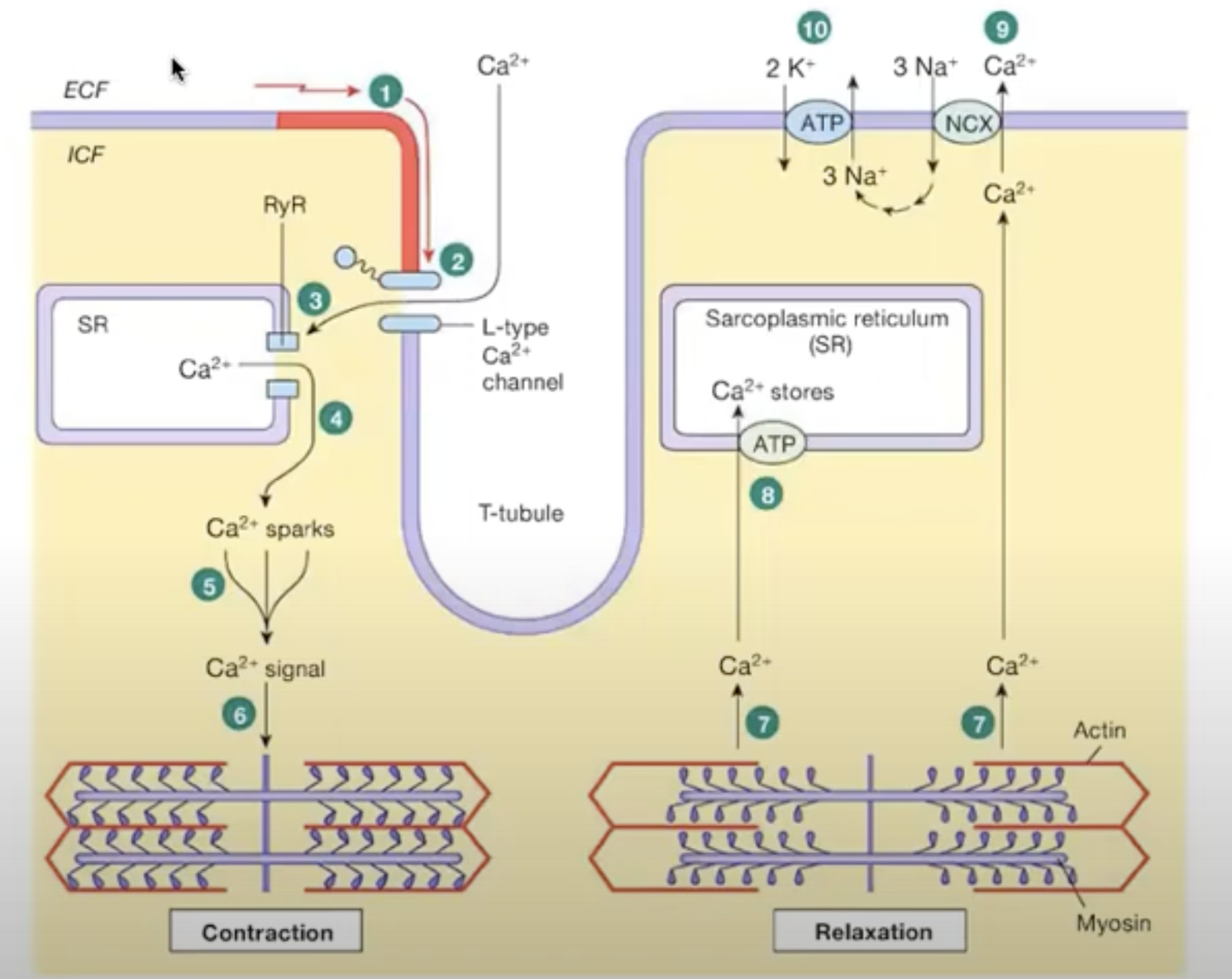
first action potential creates a voltage (electrical signal) which reaches a channel on the surface of the cardiac muscle cell. This channel is the calcium voltage channel
voltage causes calcium channel to open and calcium outside of cell enters the cell
the calcium entering the cell binds to a nyanodine receptor on the sarcoplasmic reticulum storing calcium. then the receptor opens (RYR receptor) which makes more calcium enter cell
intracellular calcium is increases
this makes many calcium sparks, which leads to a calcium signal
Calcium signal goes to contractile unit of muscle cell
This is the same calcium that binds to troponin on the actin filament
Muscle relaxation [left side of photo]
calcium unbinds from troponin and tropomyosin blocks site for myosin to bind to actin
actin filaments are pulled apart, power stroke is done
Calcium is pumped back into sarcoplasmic reticulum [calcium pump actively pumps it into cell, it’s primary active transport]
Some calcium is pumped out of the cell by a sodium calcium exchanger, [NCX antiporter]
concentration gradient for this to work is maintained by sodium potassium pump