ATOMIC STRUCTURE
The structure of an atom
Neutrons have a mass number of 1, and no charge
Protons have a mass number of 1, and +1 charge - they determine the element
Electrons have a -1 charge, and a very very very very small mass
The mass number of an element is the total number of neutrons and protons
The atomic number is the total number of protons ( and therefore electrons)
Isotopes have the same number of protons and a different number of neutrons, so a different mass number
Only about half of an elements isotopes are stable - unstable isotopes decay into other elements by emitting radiation (radioactive decay)
Electrons are arranged in shells and can jump up energy levels if they have enough energy - get ‘excited’
The energy needed comes from electromagnetic radiation
Soon after, electron will fall back to the previous shell and emit energy as electromagnetic radiation
Ionisation is when an electron leaves the outer shell of an atom and becomes a + ion
Ionising radiation is radiation that is able to knock electrons of the outer shells of atoms
Nuclear radiation
Only a few of an element’s isotopes are stable
unstable isotopes undergo radioactive decay, where they emit something (neutron, proton, waves or electrons)
Alpha radiation
2 protons and 2 neutrons are emitted (He nucleus)
2+ overall charge
Relatively large, so it can’t penetrate very far into other materials
Can only travel a few cm in air and is absorbed by a single sheet of paper
Is strongly ionising (can easily knock off electrons)
Beta particles
Electrons are emitted
Charge of -1 and very little mass
They aren’t electrons emitted from shells, but instead a neutron splits into a proton and an electron, which is emitted at high speed
It is moderately ionising
Penetrates moderately far - several metres in air and is absorbed by a few mm of aluminium
Gamma rays
Waves of electromagnetic radiation
Is often emitted after alpha or beta radiation
There is no mass or charge
Is weakly ionising
Penetrates really far in air - a few km and is only absorbed by thick lead
Emission of a neutron
If an isotope has too many neutrons, it can emit one to become more stable
Half lives and radioactive decay
Unstable isotopes can emit radiation to become stable - radioactive materials
This decay process is random, so you can’t tell when an isotope is going to decay
Activity is the overall rate of decay of all isotopes in a sample
A becquerel is 1 decay per second
Half-life is the time taken for the number of nuclei in a radioactive sample to halve or the time taken for the number of decays to half, which are both correlated
For example, if the activity was 600, then 300 and then 150 every 2 hr (halving), the half life would be 2 hours
Activity can be recorded by a Geiger-muller tube and counter, which records all the decays that reach it per second (includes background radiation - very small) - known as the count rate
Radioactive contamination
Irradiation is the process by which objects are exposed to radiation, but doesn’t continue to ionise when you leave the area
It can be ionising or not (like ultraviolet)
Any radiation reaching you is irradiating you
Contamination is when radioactive particles are on or in other objects
If you are contaminated, the radioactive isotope is more likely to decay and therefore irradiates you
Ionising radiation is the most dangerous as it can enter other cells and interact with the molecules inside - can ionise DNA and causes mutations, developing cancer
Alpha is the most harmful in/on the body and then Bata, then Gamma
Out of the body, Beta and Gamma are the most dangerous as they can penetrate skin easily
Dosage depends on how far you are from the source, how long the exposure is and how radioactive it is
Precautions include wearing protective, lead lined clothing, storing in a lead lined box and using tongs to handle it.
ONLY CONTAMINATION IS HARMFUL TO OTHERS
Uses of radiation
Medical
Sterilisation of surgical instruments
Gamma rays to kill cancerous tumours - They are aimed at the tumours, so it gets the highest dose and attacks the cells
Beta radiation can be put directly in the body next to the cancer/ in it, and is more damaging, but it can’t travel as far
Potential side effects could be that healthy cells are killed or damaged
Domestic and industrial
Smoke alarms use Alpha radiation, and is absorbed by smoke when present, which breaks circuit and sets it off
Thickness monitoring with Beta radiation
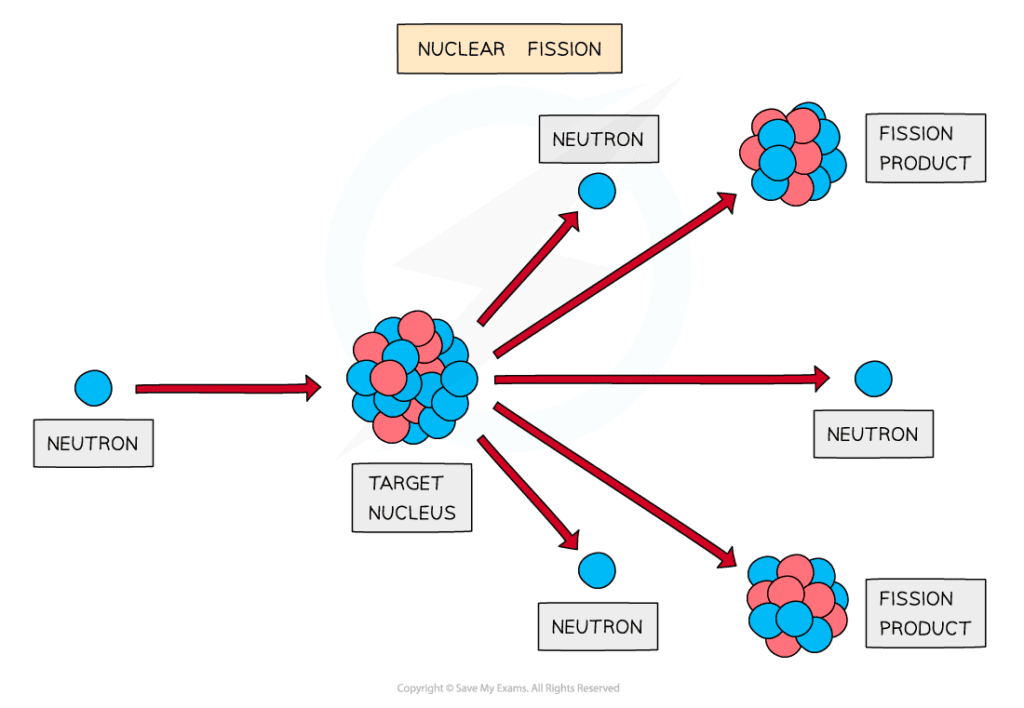
Nuclear fission
Nuclear fission is the splitting of a large, unstable nuclei into smaller nuclei while releasing energy
It can be spontaneous and split by itself, unforced - rare
Or it can happen by absorbing a neutron
This splits a nucleus
It is used in nuclear reactors
Usually uranium 235 is used as it is a large, unstable nucleus
A neutron is fired at it, causing the nucleus to split into two smaller nuclei, and also releases a few more neutrons and lots of energy
When this happens, the neutrons released fire at more uranium 235 and cause a continuous chain reaction (what happens in a nuclear reactor and a nuclear bomb - when uncontrolled)
In nuclear reactors, the rate of fission has to be carefully controlled
Done by control rods which are lowered into the reactor to absorb neutrons and stop the chain reaction
The energy released can be used to heat up water, to create steam and drives turbines to power generators and create electricity
PROS - Uranium or Plutonium fuel is relatively cheap
It creates a large and steady amount of energy
It is clean energy
CONS - Nuclear power plants are expensive to build
Nuclear waste is expensive to remove
Risks of major disaster
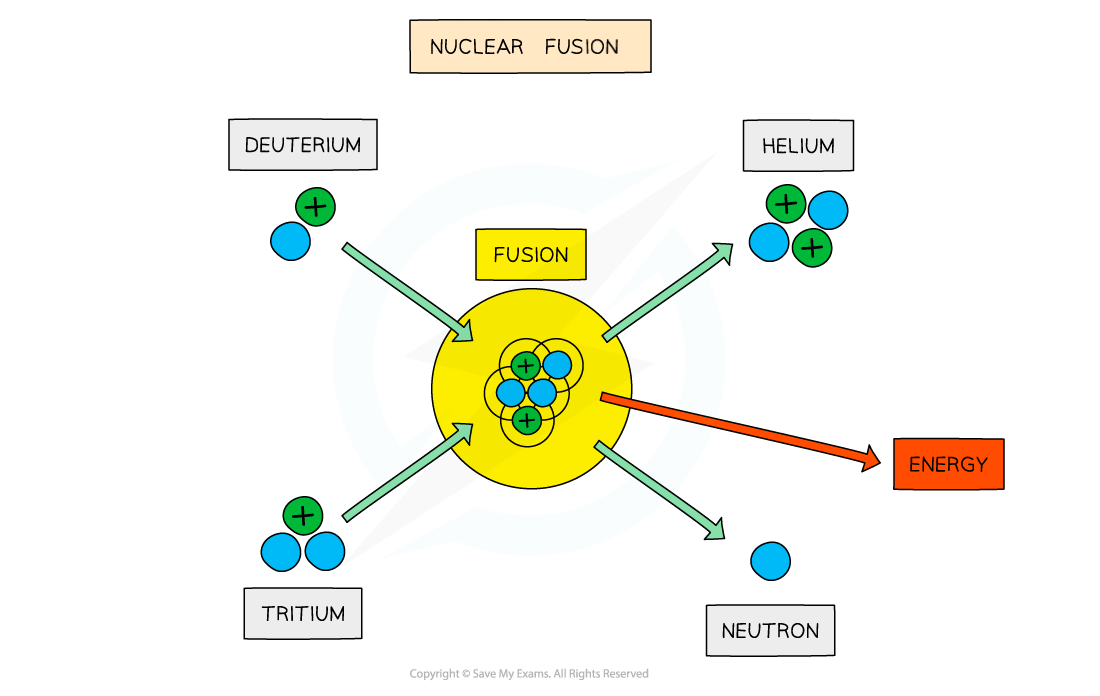
Nuclear fusion
Nuclear fusion is two nuclei fusing together to create a larger one, and releases energy
For example, 11 H + 12 H 32 H , and loads of energy is released (and a neutron)
This is the process used to fuel stars
Some of the mass from original nuclei is converted into energy
PROS - Produces no radioactive waste
Can easily make hydrogen for fuel
CONS - It can only happen at really high temperatures and pressure (10000000 degrees c), so we can’t currently do it on earth
Background radiation
Everyone experiences and is exposed to a low level of radiation everyday
From natural and man-made sources
It can be from - buildings, radon gas from the ground (50%), food and drink and artificial sources (12% - 90% of which are medical, and nuclear and weapon testing makes up less than 5%)
The level you experience can depend on where you live, your job, etc.
This most be taken into account in experiments
Generally irradiation, but some contamination in foods 🍌
A becquerel (Bq) is 1 activity per second
A sievert (Sv) is the unit to measure radiation dosage
The amount of damage that would be caused by absorption of 1J in each kg of body mass
For example, eating a banana is 0.000098 mSv and a fatal dose is 10000mSv
Nuclear equations
Alpha is a helium nucleus (2 protons and 2 neutrons)
Mass number changes by -4, and atomic number changes by -2
Beta is the loss of an electron from a neutron
+1 to the atomic number (no mass change as so small)
Gamma has no change as it is pure energy
DONE!!!