Enzymes
- Biological catalysts * Speed up a chemical rxn ==without being consumed== by the reaction
- Each enzyme has a ==unique 3-dimensional shape==, and this shape determines ==which rxn it catalyzes==
Enzyme Structure
- The ==substrate is the reactant== that the enzyme acts on when it ==catalyzes a reaction==
- The ==product== is the ==end chemical produced==
- The substrate ==binds to a region on the surface of the enzyme== known as the ==active site== * This forms an ==enzyme-substrate complex== * It also ==lowers the activation energy==
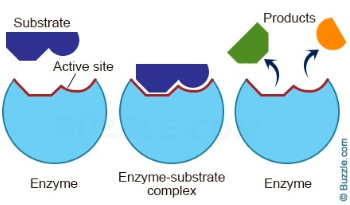
- The enzyme and substrate shape ==match== each other exactly
- Enzymes are ==specific== since ==only one enzyme acts on only one substrate==
- Once the substrate is catalyzed, the enzyme takes its original form and can be reused
Types of Reactions
- Degradation Reaction (‘breaking down’)
- Synthesis Reaction (building up)
Degradation Rxn
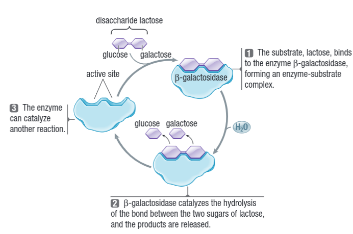
Lock and Key
- The substrate and enzymes active site are ==complementary shapes==
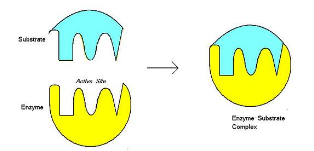
Induced Fit Hypothesis
- Enzymes are ==not rigid objects==, like locks, but they are ==flexible==
- The enzyme ==changes its shape== (conformation) prior to substrate binding so that the active site becomes ==even more precise in its ability to bind to its substrate== * This is the ==induced-fit model==
- An enzyme binds to ==one or more substrates==, forming an ==enzyme substrate complex==
- The enzyme then converts the substrate into ==one or more products==, and since enzymes stay ==unchanged== after a rxn, enzyme molecules can ==rapidly bind to other substrate molecules==, catalyzing the same rxn repeatedly
Factors Affecting Enzyme Activity
- Enzyme concentration
- Temperature
- pH
Enzyme and Substrate Concentration
Enzyme Concentration
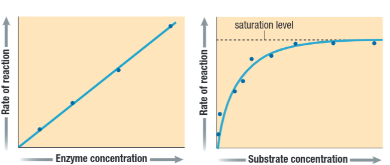
- ==Increasing enzyme [ ]== will ==speed up the reaction==, as long as there is ==substrate available to bind to==
- Once all of the substrate is bound, the rxn will ==no longer speed up==, since there will be nothing for addition enzymes to bind to
Substrate Concentration
- ==Increasing substrate [ ]== also ==increases the rate of rxn== to a certain point
- Once all of the enzymes have bound, any substrate increase will have ==no effect on the rate of rxn==, as the available enzymes will be ==saturated== and ==working at their max rate==
Temperature
- As ==temp increases==, ==enzyme activity increases==
- The temp that the enzymes work best at is called its ==optimum temperature==
- If the temp is ==too high==, the enzymes active site ==changes shape==
- When the enzymes active site has changed shape, the enzyme is said to be ==denatured== and it ==will no longer work==
- For enzymes in human cells, the optimal temp is usually around ==human body temp== (37.5)
pH
- Enzymes work best within a ==range of pH== depending on the type of enzyme
- The pH that the enzymes work best at is called ==optimum pH== * This is usually around a ==pH of 7==
- If pH is too ==high==, enzyme active site ==changes shape== (denatured)
Enzyme Activators
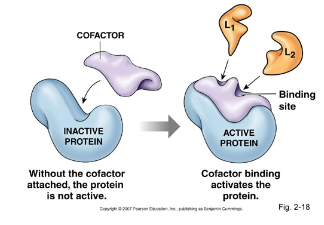
- Enzyme activators are molecules that ==bind to an enzyme== and ==turn them on== in a chemical rxn cofactors
- Types of enzyme activators include: * Cofactors * Coenzymes
Cofactors
- ==Inorganic==, ==non-protein group== that ==binds to an enzyme== and is ==essential for catalytic activity==
- Most often ==metals== (iron, copper, zinc, and manganese) are cofactors
Coenzymes
- ==Organic==, ==non-protein molecules== that ==act like cofactors==
- They act as ==electron transport carriers== during ==biochemical pathways==
- Both cofactors and coenzymes can ==bind to either the substrate or the active site== of the enzyme
Enzyme Inhibitors
- Enzymes need to be ==regulated== to ensure that levels of the product don’t rise to ==undesired levels==, and this is accomplished by ==enzyme inhibition==
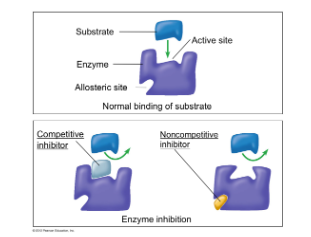
- Enzyme inhibitors are ==molecules that binds to an enzyme== and ==turn them off in a chemical rxn==
- Types of inhibitors include: * Competitive inhibitors * Non-competitive inhibitors
- Reversible and irreversible inhibitors are ==chemicals which bind to an enzyme== to ==suppress its activity== * ==Irreversible inhibitors== almost ==permanently bind to an enzyme== * ==Reversible inhibitors== are ==chemicals that transient bind to an enzyme==, either to an ==active site (competitive inhibitor)== or to ==another site on the enzyme (non-competitive inhibitors)==
- ==Competitive== inhibitors are so ==similar== to an ==enzyme’s substrate== that they can ==bind to the active site== and ==block the normal substrate==
- ==Non-competitive== inhibitors ==bind to the enzyme== at an ==allosteric site (not the active site)== and cause a ==conformational change in the enzyme==, preventing the normal substrate from binding
Allosteric Control of Enzyme Activity
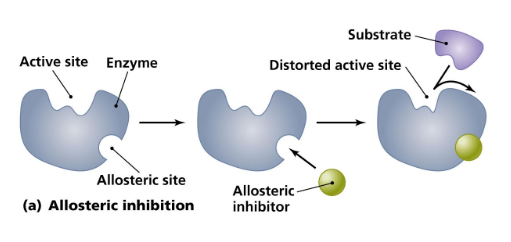
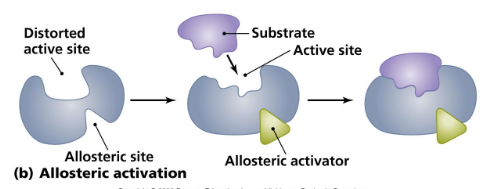
- The allosteric site is a binding site on an enzyme that ==binds regulatory molecules== * Either ==activate== or ==inhibit==, or turn off, ==enzyme activity==
- These molecules ==bind the allosteric site== and ==change the conformation, or shape, of the enzyme==
- ==Allosteric regulation== is the regulation of one site of a protein ==binding== to another site on the same protein
Allosteric Regulation
- In allosteric inhibition, the active site ==changes shape== when an inhibitor ==binds to an allosteric site==
- In ==allosteric activation==, the activators ==may bind to allosterically controlled enzymes== to st==abilize its shape== and keep all active sites available
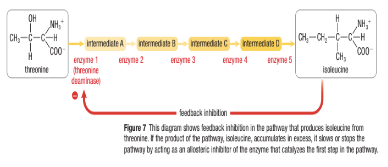
Feedback Inhibition
- Occurs when there is a ==sequence of chemical rxns== that are ==forming a common product in the end==
- When the product ==travels back== and ==inhibits an earlier enzyme== in the rxn (competitively or non-competitively) the pathway is ==interrupted== and no more product is made
Uses of Enzymes
