Ch. 11 Oxygen in Lakes
Why study oxygen?
Essential for the metabolism of most organisms
all higher organisms require oxygen
Critical role in nutrient cycling and energy flow
oxygen is the most crucial nutrient
Solubility of Oxygen in Water
Solubility of oxygen in water depends on:
1) Temperature
warm water holds less oxygen than cold water (same with all gases → gas law)
2) Pressure → increase in pressure creates greater solubility of oxygen
allowing more gas to be dissolved in the water
3) Salinity → increase in salinity creates a decrease in solubility of oxygen
Photosynthesis and Respiration
Photosynthesis: CO2 + H2O → C6H12O6 + O2
Photosynthesis creates oxygen, which allowed oxygen in our atmosphere, which allowed for higher organisms
Respiration: C6H12O6 + O2 → CO2 + H2O
Respiration consumes oxygen
Most oxygen consumption comes from the decomposition of organisms
Sinks and sources of Oxygen in Lakes
Sources:
Diffusion
oxygen from the atmosphere is diffusing into the lake
Photosynthesis
aquatic macrophytes create oxygen in the lake using photosynthesis
Water inflow
from rivers that contain high oxygen
Sinks:
Respiration (BOD)
taking in oxygen and turning into CO2
Decomposition (BOD)
Photoxidation
Water inflow
from rivers that are contaminated and have less oxygen
Chemical oxygen demand (COD)
certain chemicals take in oxygen
BOD = biological oxygen demand
COD = chemical oxygen demand
Anoxia: absence of dissolved oxygen
zero oxygen
Hypoxia: low dissolved oxygen conditions
some organisms are dead before they get to zero oxygen
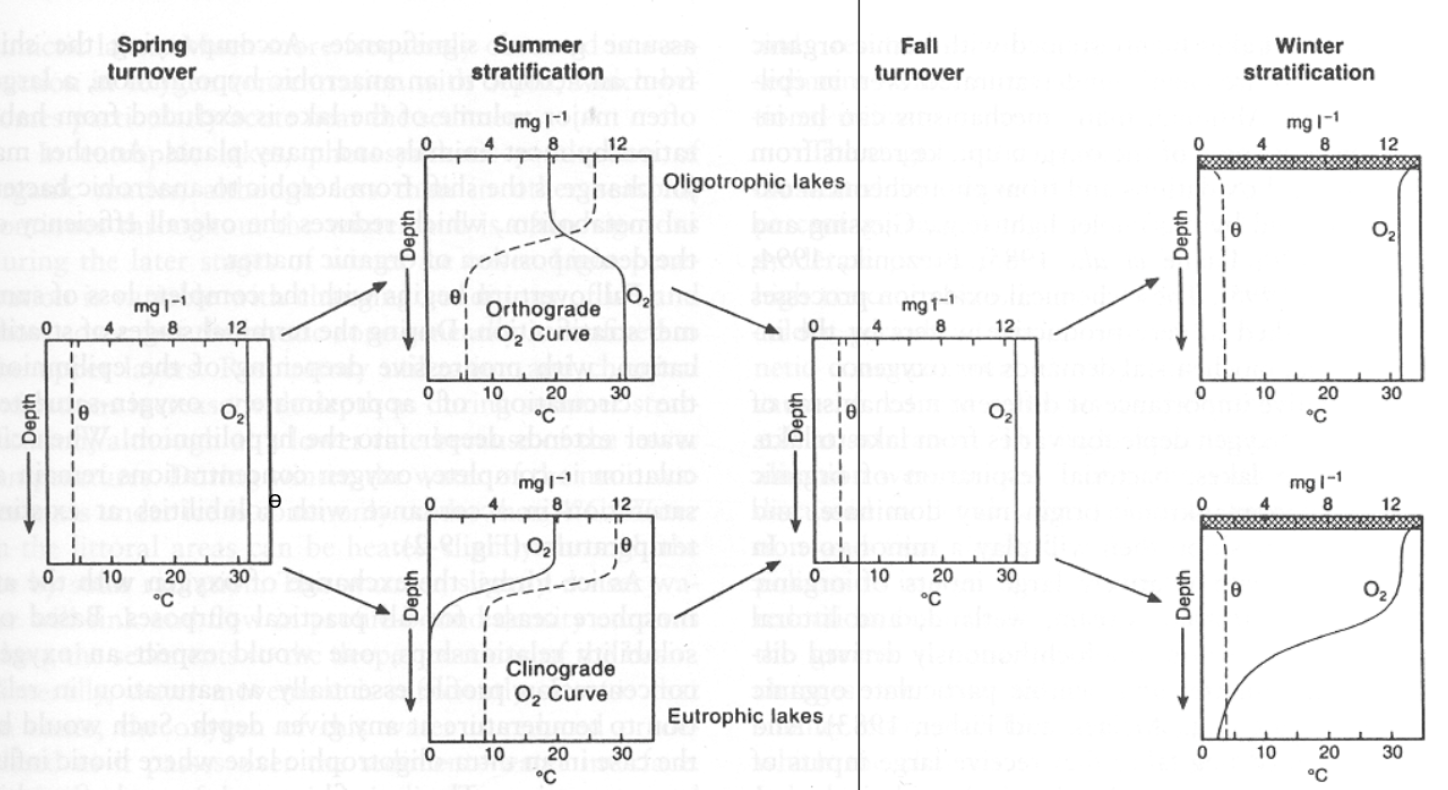
Lake Trophic State Classification
Ogliotrophic = nutrient poor, low algae production
Mesotrophic = medium level of nutrients, medium algae production
Eutrophic = high level of nutrients, high algae production
high algae production can lead to less light in deep water → less photosynthesis in deep water → less oxygen because of more decomposition and more respiration
Significance of Anaerobic Layer
Obligate requirement of many organisms for oxygen (e.g. kills fish)
Important implications for nutrient cycling
Ways to Express Dissolved Oxygen (DO)
2 ways we typically express dissolved oxygen (DO) data:
1) Concentration data (mg/L or ppm)
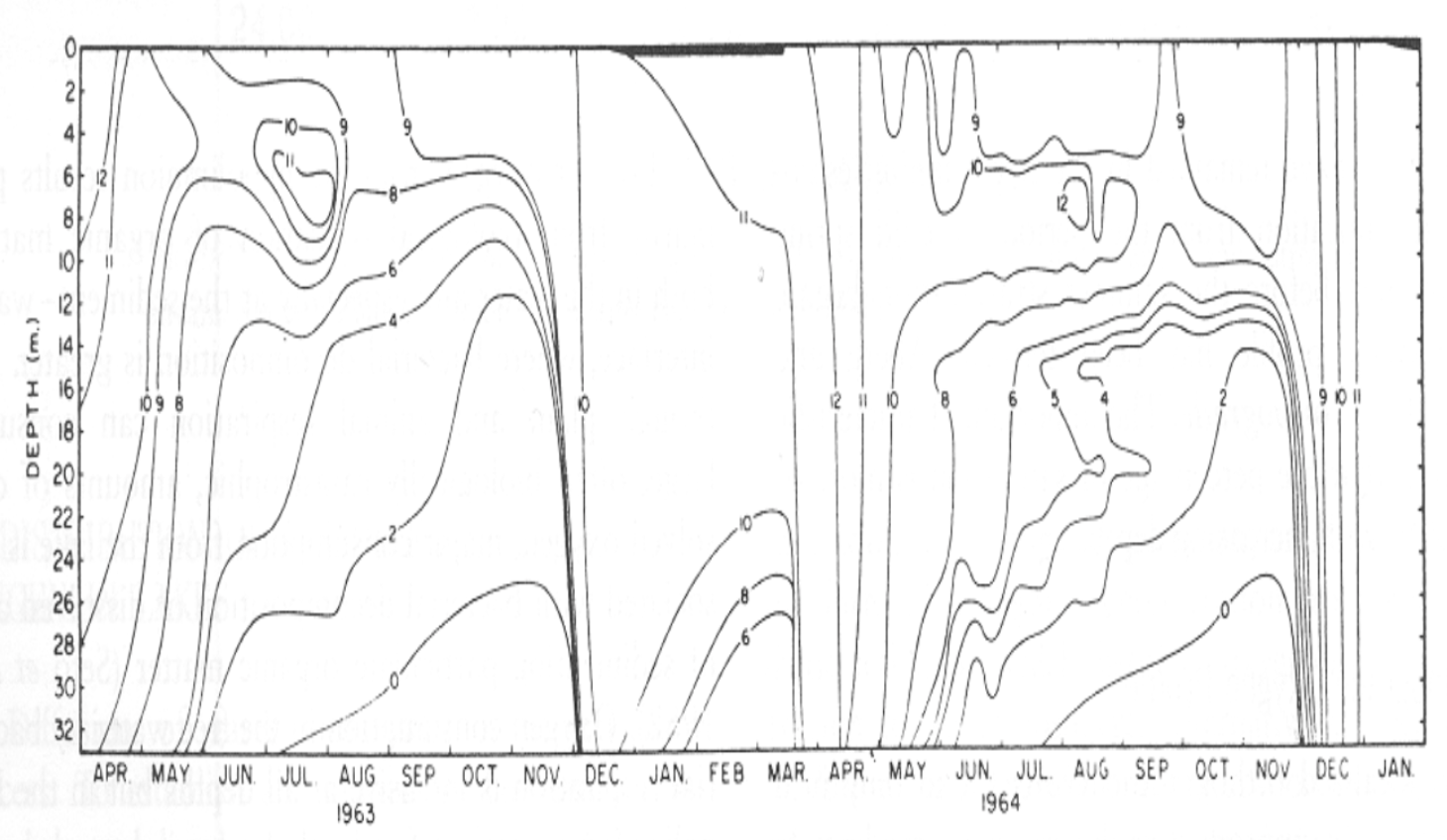
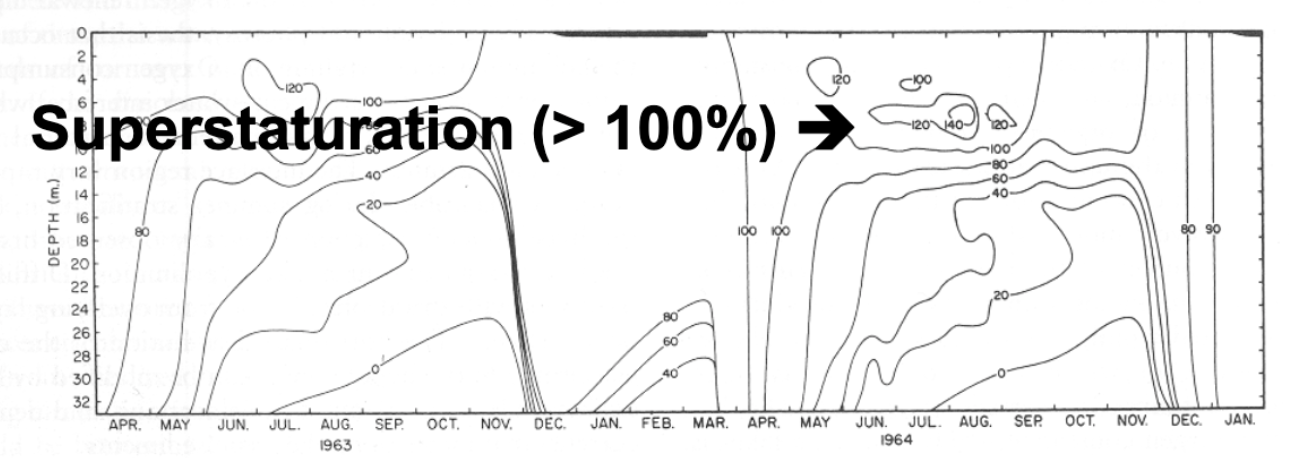
2) Percent saturation (%)
Measuring Devices for Dissolved Oxygen in a Lake
Dissolved oxygen meter
Wikler titrations
standard method
Variations in Oxygen Profiles
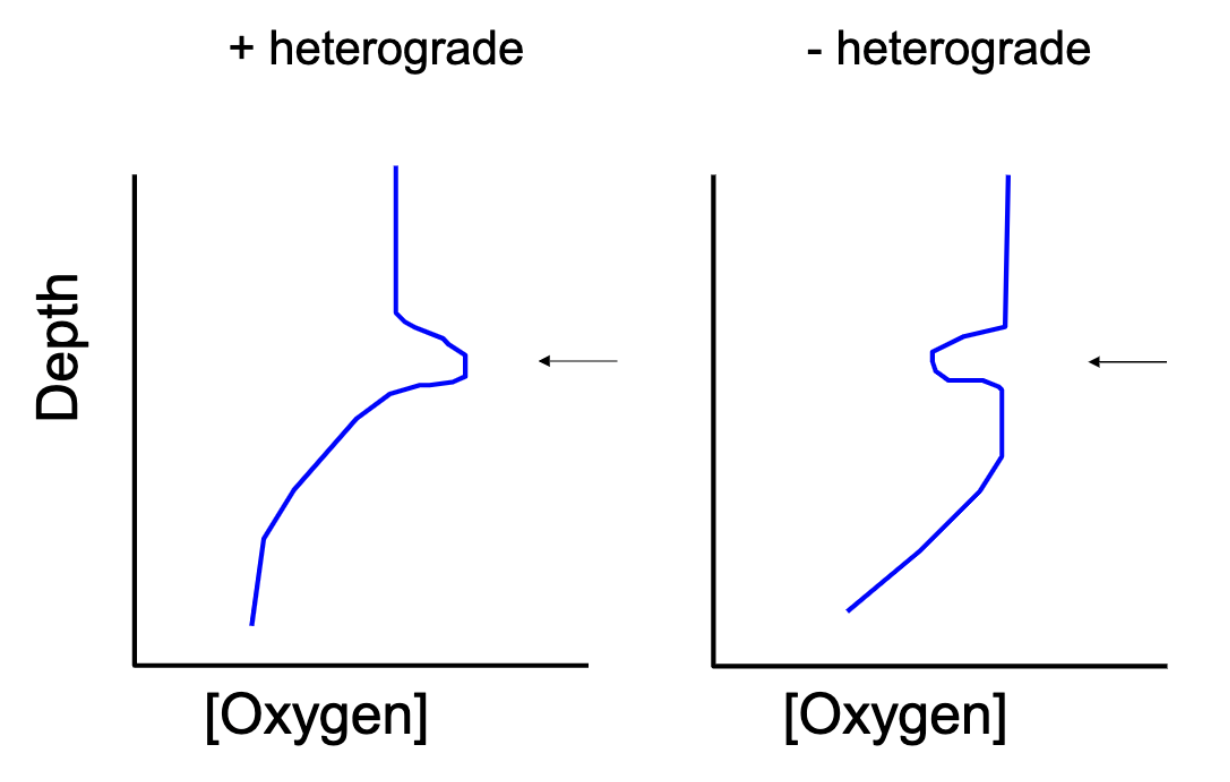
(+) heterograde
bump in (+) oygen
created by an abundance of algae bloom at a specific depth → producing an excess amount of oxygen
(-) heterograde
(-) bump in oxygen
caused by respiration and decomposition
usually by an abundance of zooplankton
Example:
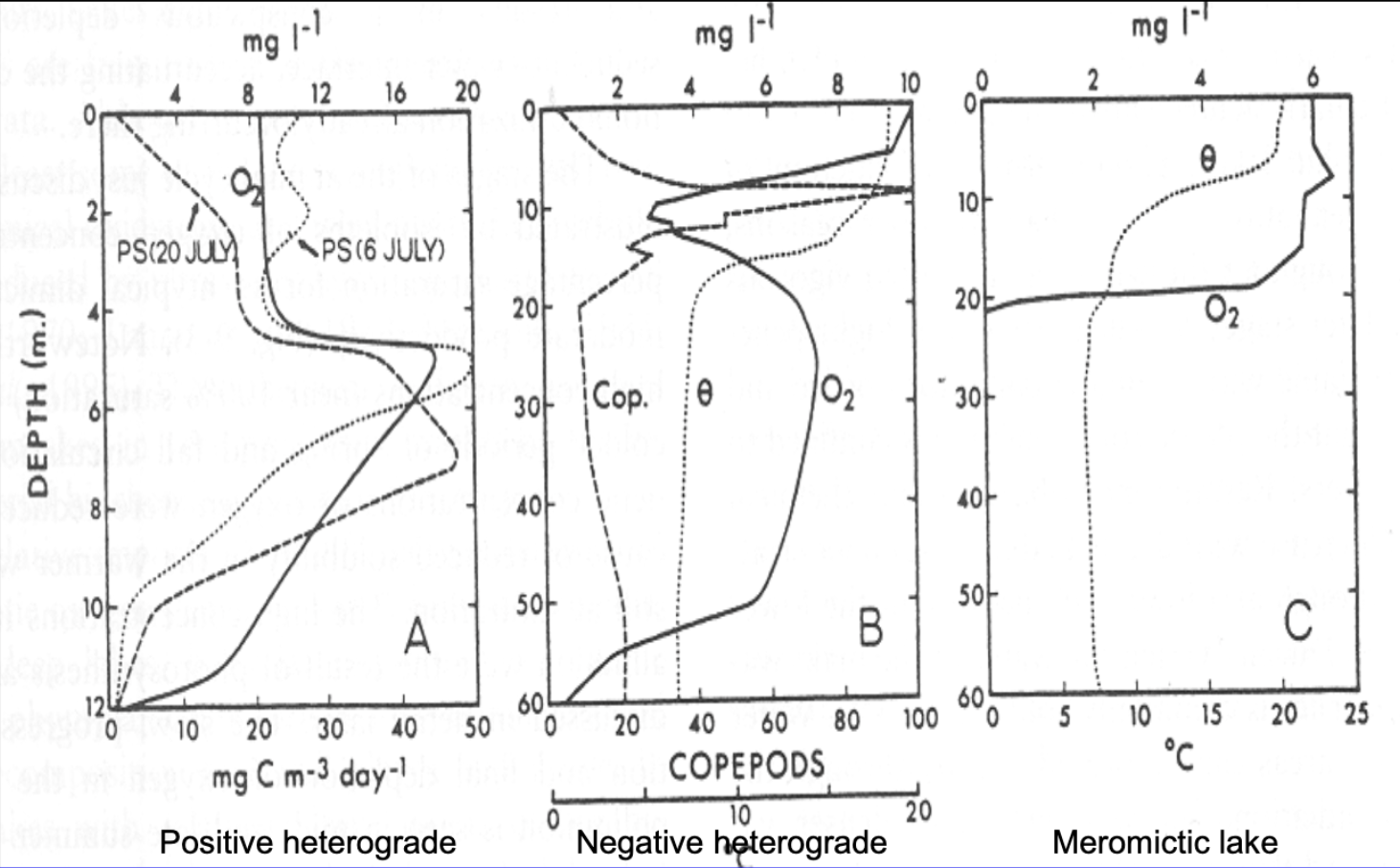
Changes in a Lake
Diurnal Changes = changes over the course of a 24h day
e.g. there might an abundance of photosynthesis during the day, and nothing during the night (high oxygen during the day and low oxygen at night)
Horizontal Differences in O2
Between basin differences
variations in depth, temperature stratification, and nutrient availability, leading to distinct oxygen concentrations in different areas of the lake
Aquatic macrophytes
higher plants that photosynthesize a lot creating super-stratification
Organic pollution from point sources (e.g. incoming river)
Ice cover
creates a barrier on the lake, decreasing oxygen coming from the atmosphere and decreases photosynthesis
there might be ice covering one side of the lake whereas the other side has no ice covering