Hydrogen Fuel Cell
What is a Hydrogen Fuel Cell?
A hydrogen fuel cell generates electricity through an electrochemical reaction between hydrogen and oxygen, producing only water and heat as byproducts.
Diagram of a Hydrogen Fuel Cell
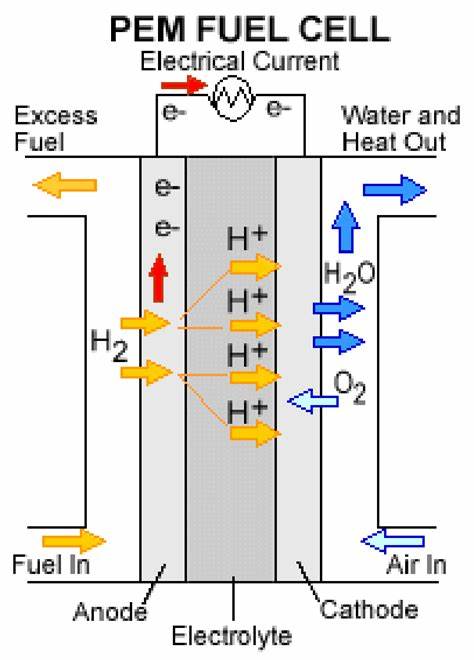
Principles of a Hydrogen Fuel Cell:
The basic principles include:
Electrolyte: The cell contains an electrolyte that facilitates the movement of ions between electrodes.
Anode Reaction: Hydrogen gas (H₂) is introduced to the anode, where a catalyst splits it into protons (H⁺) and electrons (e⁻).
H2 → 2H+ + 2e−
Cathode Reaction: Oxygen gas (O₂) is introduced to the cathode, where it reacts with the protons and electrons, forming water.
O2 + 4H+ + 4e− → 2H2O
Electron Flow: The electrons flow through an external circuit from the anode to the cathode, creating an electric current that can power devices
Benefits of Hydrogen Fuel Cells:
Environmentally Friendly: The primary byproducts are water and heat, making them a clean energy source with no greenhouse gas emissions during operation.
Efficient: Hydrogen fuel cells can achieve higher efficiencies than traditional combustion engines, especially in combined heat and power applications.
Renewable Potential: Hydrogen can be produced from renewable energy sources, such as solar and wind, via electrolysis.
Quiet Operation: Fuel cells operate silently, making them suitable for various applications, including residential and transportation.
Energy Density: Hydrogen has a high energy-to-weight ratio, allowing for lightweight, energy-dense storage.
Drawbacks of Hydrogen Fuel Cells:
Hydrogen Production: Most hydrogen is produced from fossil fuels (e.g., natural gas) emitting carbon dioxide. Electrolysis using renewable energy is not yet widespread due to cost and efficiency challenges.
Cost: Fuel cells are expensive due to the use of precious metals like platinum in catalysts and the complexities of hydrogen storage and infrastructure.
Storage and Transport: Hydrogen is difficult to store and transport because it is a low-density gas requiring high-pressure tanks, cryogenic temperatures, or chemical bonding.
Infrastructure Gaps: There is limited refuelling infrastructure for hydrogen vehicles, which hampers widespread adoption.
Durability: Fuel cells have a limited lifespan and can degrade over time, particularly when exposed to impurities in hydrogen fuel.