Diels-Alder reaction
Pericyclic reaction - reaction with no intermediates and no charges
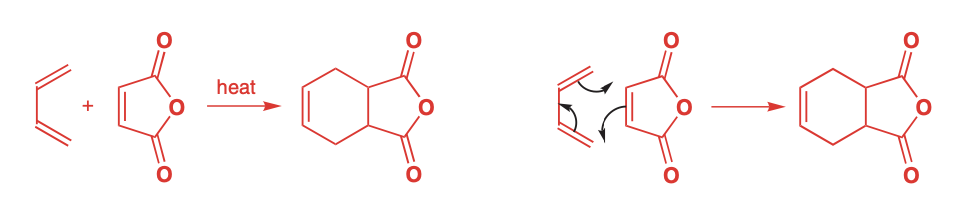
Cycloaddition - two or more molecules form to create a cyclic structure
Diels-Alder reaction - Diene + Dienophile = Product
Geometry is retained through reaction
will create a racemic mixture
Diene - the 4 pi system
electron rich
electron donor groups
R groups must be cis
can be open chain or cyclic
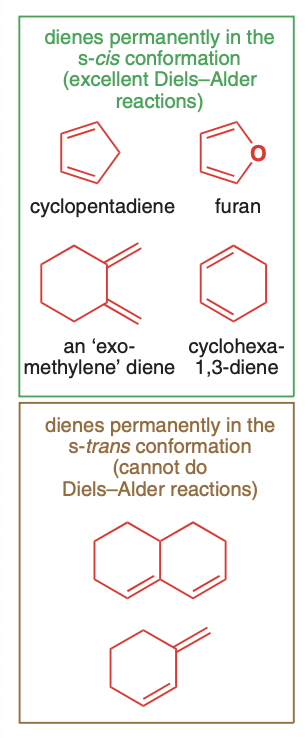
Dienophile - the 2 pi system
electron poor
electron-withdrawing groups
the minimum for reaction is a phenyl group or a chlorine atom
R groups can be either cis or trans
Simple alkenes that work
conjugated carbonyl compounds
nitro compounds
nitriles, sulfones
aryl alkenes
vinyl ethers and esters
haloalkenes
dienes
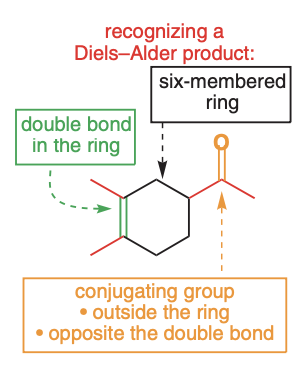
Product - the final product
Six-member ring with a double bond inside the ring and a conjugating group outside of the ring, opposite to the alkene
Temperature should be around 100 - 150 °C to work
Sterioselectivity
Geometry is retained
Endo rule for Diels-Alder reaction
Exo product - more stable thermodynamic product
less sterics
Endo product - less stable kenetic product
more sterics
If reaction is reversible
Exo product formed
If reaction is irreversible
Endo product formed
Easiest way to figure out final sterochemistry
Draw the overall mechanism
Draw both molecules in the plane of the paper with the diene over the denophile
Add all hydrogen atoms to the carbons with stereogenic centers
Draw out the final product unfolded to show the geometry of the hydrogens
Add the stereochemistry of the additional substituents
This is the endo product
Dimerization of dienes
Due to high LUMO and low HOMO, dienes are susceptible to dimerization
Solvent - contrary to expectation for organic molecules, water works well as an “anti-solvent” due to it forcing organics to close proximity
Intermolecular Diels-Alder
Sterics are more important than endo rule
Regioselectivity
Attack the less substituted carbon first as it will create a more stable cation as an intermediate
The carbons with the largest coefficient of HOMO will attach first
Summary and tips
Diels-Alder reaction will lead to an aromatic which will direct ortho and para