Lesson 1-2-3, and 4: States of Matter, Physical and Chemical Change, Elements, Compounds, and Mixtures, & Atomic Structure.
Lesson 1: States of Matter
States of Matter includes: Solid, Liquid, and Gas.
Solid - packed closely infixed positions and cannot move anywhere. It cannot be compressed or squished. They are rigid. No kinetic energy. (Computer, Phone, Ice, etc.)
Liquid - (water) particles are closely packed but not so close that they can move with each other it can be compressed. No fixed shape, fixed volume. It takes up its container.
Gas - (Helium) particles are moving they have a strong kinetic energy. They are farm apart. They can be compressed. No fixed shape and volume. They take up the container or place they are in.
Lesson 2: Physical and Chemical Properties/Change
Physical Properties are properties that are observed without changing the composition of the substance, though their form may change. color, viscosity, transparency, melting point, boiling point, hardness, density, specific heat, and atomic or molecular diameter involved in a physical change.
Examples of Physical Change:
Melting Ice
Boiling Water
Tearing Paper
Breaking Glass
Chopping Wood
Dissolving Sugar in Water
Perspiration, etc. (sweating)
Purple iodine vapor appears when solid iodine is
warmed.
Meanwhile, Chemical Properties or Changes are observed when a property of the material is changed into a new material substance concluding that there must have been a chemical change that happened by process. This property is related to the composition of the material. chemical change takes place to form completely different substances It results in the formation of new chemical substances. A chemical reaction is usually detected when one of the following is observed: formation of an insoluble product (precipitate), evolution of gas (bubbles), or change in color.
Examples of Chemical Changes:
Burning Wood
A cornstalk grows from a seed that is watered and
fertilized.
A match ignites to form ash and a mixture of gases
A silver fork tarnishes slowly in air.
A scab forms over an open cut.
Gasoline fumes are ignited by a spark in a car’s
engine cylinder.
Electric current decomposes water into different
substances (hydrogen and oxygen).
Lesson 3: Elements, Compounds, and Mixtures.
Matter is composed of Pure Substances and Mixtures. Under Pure Substances we have Elements- elements are the only one in a substance, meaning they do not share the space with anything. For example: Oxygen, Helium, Hydrogen, Nitrogen, and Carbon. Pure Substances also includes Compounds- compounds has two or more elements in them making it a twin of different combination of element. An example of compounds are : Water(h20), Carbon Dioxide, Sodium Chloride, and Ethanol.
On the other hand, we have mixtures- mixtures are a mixture of two or more pure substances(wether elements or compounds). We have two types of Mixtures: Homogenous and Heterogenous.
Homogenous Mixtures refers to a composition of pure substances which are uniform to each other or in simple words when you mix these substances they look exactly like one, they do not separate but rather bond with each other. (E.g: Water and Salt, Air)
Heterogenous Mixtures refers to a composition of pure substances that are not uniform to each other. Think of a couple who got arranged marriage and do not really like each other, they end up getting divorce and ruining their children’s childhoon because they are not uniform together. An example of Heterogenous Mixtures is the combination of Oil and Water. Because oil is less denser than water it settles at the top of it while water sinks.
Lesson 4: Atomic Theory
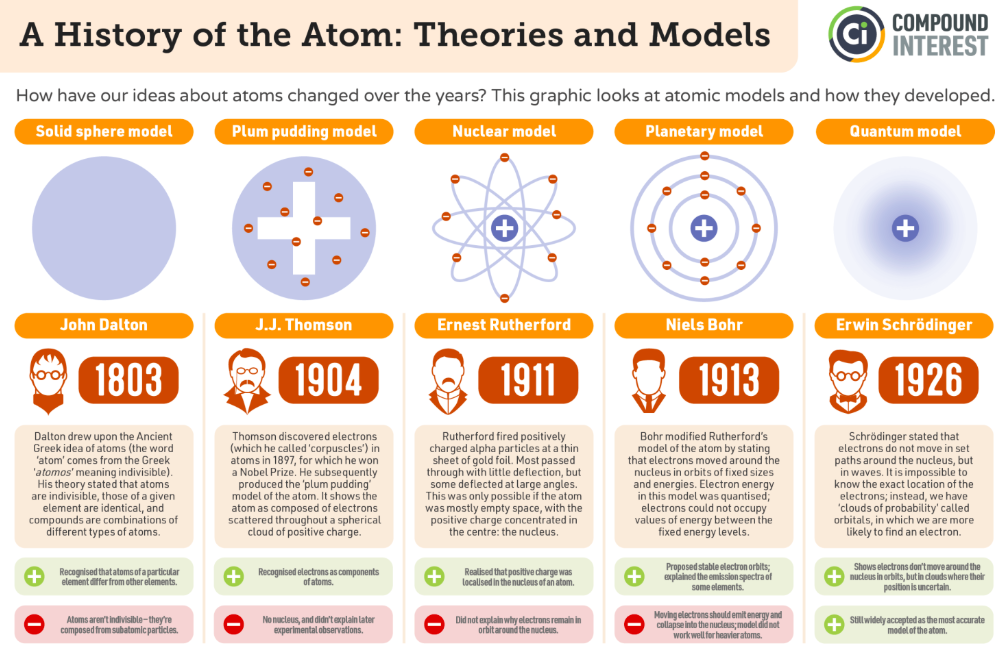
John Dalton’s atomic theory, proposed in 1804, was the first modern attempt to describe all matter in terms of atoms and their properties. He proposed that matter consists of indivisible atoms, atoms of an element are identical in mass/properties, compounds are combinations of different types of atoms, and chemical reactions involve the rearrangement of these atoms.
Matter Composition: All matter is composed of extremely small, indivisible, and indestructible particles called atoms.
Element Properties: All atoms of a specific element are identical in mass, size, and other properties.
Differentiation: Atoms of different elements differ in size, mass, and other properties.
Compound Formation: Atoms of different elements can combine in fixed, simple whole-number ratios to form compounds.
Chemical Reactions: Chemical reactions involve the separation, combination, or rearrangement of atoms; atoms are not created or destroyed in the process.
Significance: This theory established the foundation of modern chemistry, introducing the concept of atomic weights for elements.
Atom - came from greek word which literally means “uncuttable” or “indestructible”. It is the smallest unit of matter the retains the properties of a substance. Smallest particle of a given element.
Subatomic Particles (inside the atom) - Proton(+), Neutron (Neutral), and Electron(-).
Proton - It is inside the nucleus of an atom with neutron. Its mass in grams is: . If they are in stable state and are neutral: The number of proton and electron in an element is equal. Atomic Number equals Proton. It determine the element's identity (atomic number), provide positive charge to attract electrons, and contribute to the nucleus's mass and stability. As positively charged particles (1+ charge), they define the element’s chemical properties and hold the nucleus together through strong forces, acting as the fundamental building block. (Protons and Neutrons together are called: Nucleons.)
Neutron - Inside the nucleus with Proton. Its mass in grams is: . Neutrons are essential subatomic particles found in atomic nuclei that stabilize the nucleus by counteracting proton repulsion via the nuclear force, allowing for the existence of elements heavier than hydrogen. They contribute to nuclear mass, facilitate isotope formation, drive nuclear fission, and serve as non-destructive tools for material analysis and imaging.
Electron - outside the nucleus of an atom. It is a negatively charged particle. Its mass in grams is: . Drive chemical bonding, electricity, and atomic structure. They determine chemical reactivity through valence shell interactions, create electricity via movement in conductors, and maintain atomic stability by balancing nuclear positive charges.
The nucleus accounts for the majority of mass of the atom.
Atomic Models
Joseph John Thompson- Plum Pudding Model. He discovered that there were negatively charged particles. He proposed that these negatively charged particles were embedded in a positively charged clouds.
Ernest Rutherford, Johannes “Hans” Willhelm Geiger and Ernest Marsden tested Thompson’s model by bombarding and very thin sheet of gold foil with positively charged alpha particles - alpha particle scattering.
Rutherford proposed: the existence of nucleus.