AP Biology
Unit 1 - Chemistry of Life
Topic 1.1: Structure of Water and Hydrogen Bonding
Form follows function: The structure of biological molecules and their sequence determine the properties of that molecule.
Polarity
In a water molecule, an oxygen atom is polar-covalently bonded to two hydrogen atoms.
Polar covalent: unequal sharing
Non-polar covalent: equal sharing
Here, covalent is the sharing of electrons between atoms. Polar molecules occur when one atom is more electronegative (attracts electrons stronger) than the other less electronegative atom. In this case, Oxygen attracts more electrons than hydrogen.
Hydrogen Bonding
Definition: When a hydrogen atom in one molecule interacts with another highly electronegative atom such as F, O, or N, in another molecule.
When two molecules are attracted to each other, this is intermolecular forces (like an interstate between states). Intramolecular forces occur between bonds within a molecule, which is what connects Oxygen and Hydrogen.
Cohesion
Definition: When two of the same molecules form hydrogen bonds.
For example: Water — Water.
Adhesion
Definition: When two different molecules form hydrogen bonds.
For example: Water — Other
Capillary Action & Transpiration
Life:
Transpiration: Movement of H2O up plants which leaves plants as water vapor into the atmosphere (like sweating).
Capillary Action: H2O clings to each other by cohesion; clinging to Xylem tubes by Adhesion.
Surface Tension
Increased Hydrogen bonding forces at the surface of water.
High Specific Heat
Absorbs a lot of thermal energy before changing state → resists sudden changes in temperature.
Life:
Creates a stable marine/land environment.
High heat of vaporization → allows for evaporative cooling while sweating.
Water as a Solvent
Water is a solvent: a dissolving liquid. It can dissolve many solutes (what gets dissolved) and is considered a universal solvent.
Hydrophilic
Water is hydrophilic (water loving). Characteristics of hydrophilic include:
Affinity for H2O
Polar, Ions
Cellulose, sugar, salt
Blood
Hydrophobic
Characteristics of hydrophobic include:
Repel H2O
Nonpolar
Oils, lipids
Tails of plasma membrane
Expansion Upon Freezing
Water molecules slow down with a decrease of temperature and bonds between water molecules become more stable and spread out. This forms a crystalline structure.
Ice is less dense than water and will float due to water’s cohesion and unique hydrogen bonding.
Life:
This creates a “blanket” for organisms that live underwater, where heat will be trapped underneath the ice. This ice blanket reduces the heat loss so they can survive.
Topic 1.2 & 1.3: Elements of Life & Introduction to Biological Macromolecules
Energy
Law of conservation of energy: Energy cannot be created nor destroyed, only transformed.
All living things need a constant input of energy.
Eating = Energy: We must access atoms and molecules from our environment (biological macromolecules).
Diversity of Carbon
Molecules can be chains, ring-shaped, or branched. Carbon is tetravalence (can have 4 bonding partners).
Elements of Life
Carbon
Hydrogen
Oxygen
Nitrogen
Phosphorus
Sulfur
Biological Macromolecules & Atoms Contained
Carbohydrates → Carbon, Hydrogen, Oxygen (CHO)
Lipids → Carbon, Hydrogen, Oxygen, sometimes Phosphorous (CHO[P])
Proteins → Carbon, Hydrogen, Oxygen, Nitrogen, sometimes Sulfur (CHON[S])
Nucleic Acids → Carbon, Hydrogen, Oxygen, Nitrogen, Phosphorous (CHONP)
Building Molecules
Monomers (subunits) link together to form polymers.
Mono = One
Poly = Many
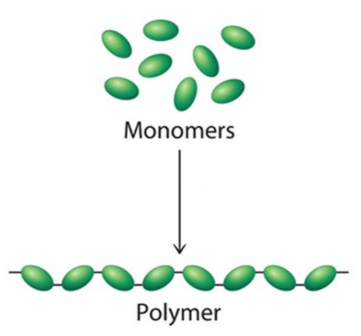
Monomers of the 4 Biological Macromolecules
Carbs = Monosaccharides
Proteins = Amino Acids
Nucleic Acids = Nucleotide
Lipids = No true monomers
Dehydration Synthesis
H2O is lost and polymers form.
Hydrolysis
H2O added to break polymers → forms monomers.
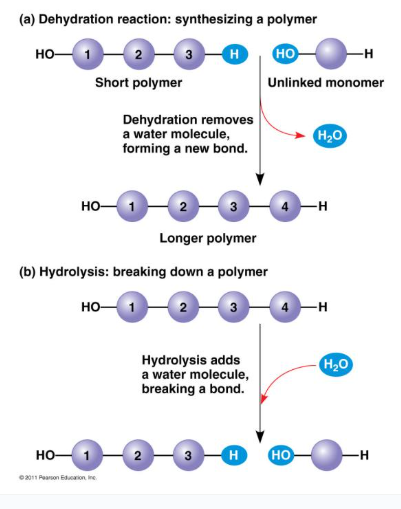
Topic 1.4: Carbohydrates
→Carbs have a 1:2:1 ratio.
Monosaccharides (monomers): Glucose and Ribose.
Polysaccharides (polymers): long chains of monosaccharides (starch and cellulose).
Function: Fuel and Building material.
Two forms of glucose:
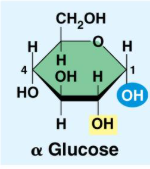
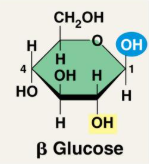
Storage Polysaccharides: Plants (starch) and Animals (glycogen)
Structure Polysaccharides: Cellulose and Chitin
Topic 1.5: Lipids
*Lipids do not have true monomers
Structure: Lots of C,H, as well as some O. Also P (Phospholipids).
There are three major types of lipids:
Fats
Triglycerides (store energy long term) and adipose (insulates).
Triglyceride = Glycerol + 3 fatty acids
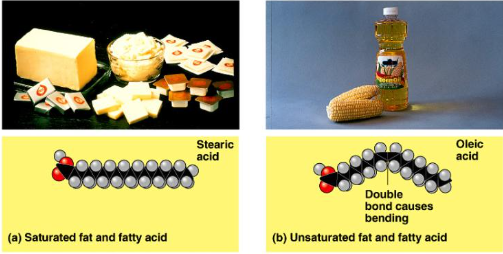
Saturated, unsaturated, polyunsaturated fatty acids.
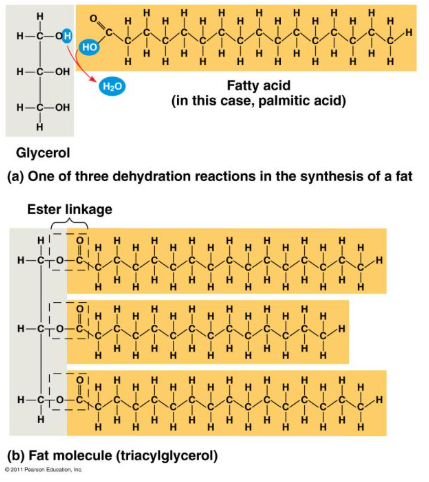
Saturated
Saturated with Hydrogen.
Solid at room temperature.
In animals.
Ex/ butter and lard.
Unsaturated and Polyunsaturated
Have Carbon double bond Carbon, resulting in kinks. Kinks = can’t tightly pack, liquid.
Liquid at room temperature.
In plants.
Ex/ Olive oil, corn oil.
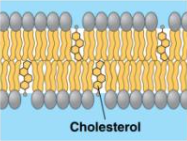
Steroids
Cholesterol (helps create plasma membrane) and hormones (chemical messengers).
Role in plasma membrane: Provides stability during temperature fluctuations.
Phospholipids
Lipid bilayer of plasma membrane.
Hydrophilic (water-loving) head.
Hydrophobic (water-hating) tails.
Amphipathic
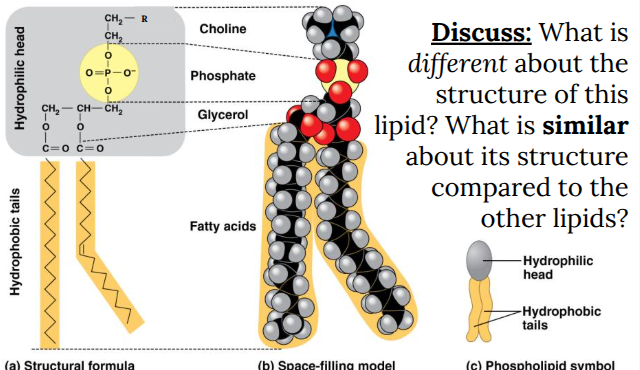
Phospholipid Bilayer: Surrounding membrane of a cell.
Topic 1.2: Nucleic Acids
Monomers: Nucleotides
Structure: C, H, O, N, P
Function: Store and transmit genetic information
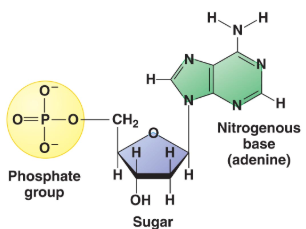
Nitrogenous bases:
Purines: Adenine and Guanine
Pyrimidines: Cytosine, Thymine, Uracil
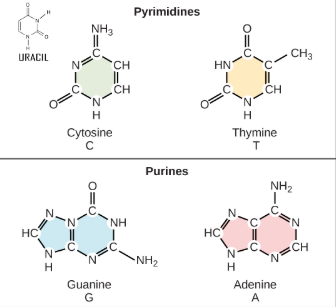
*Purines and Pyrimidines bond VIA HYDROGEN BONDS*
A double bonds to T/U
C triple bonded to G (most stable)
Backbone
Sugar (deoxyribose or ribose) = 3’
Phosphate(PO4-) = 5’
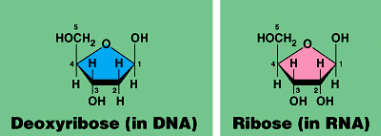
DNA is anti-parallel: You build of the 3’ end.

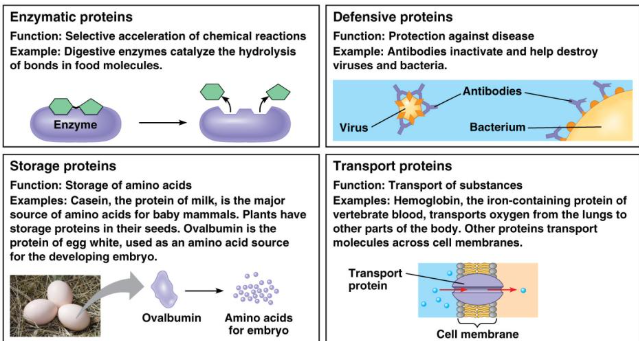
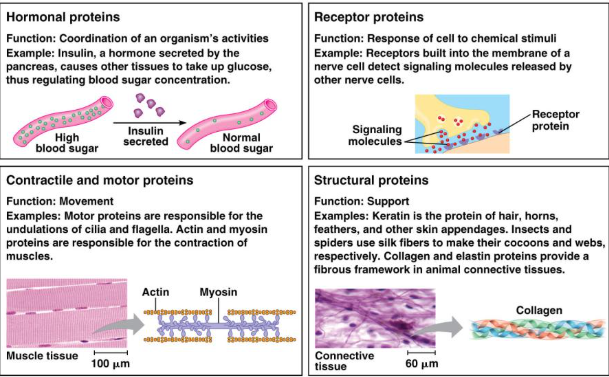
Topic 1.7: Proteins
Monomers: Amino Acids
Structure: C, H, O, N, S
4 Levels of Protein Structure
Primary Structure
Secondary Structure (contains a=helix and b=pleated sheet)
Teritary Structure
Quatenary structure
Primary Structure
Amino acid sequence = polypeptide chain.
Covalent peptide bonds link the amino acids together.
Always add to carboxyl end.
Secondary Structure
Gains 3D Shape VIA HYDROGEN BONDING.
Alpha a helix and Beta B pleated sheet.
Teritary Structure
Bonding between “R” groups of Amino Acids.
Hydrogen bonds, ionic bonds, disulfide bridges, van der waals interactions.
Quaternary
2+ poly peptides bond together.
Structure and Function
Proteins are very sensitive and can denature if pH or Temperature are not optimal.
Change in structure=change in function.